* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Population Before Selection
Transgenerational epigenetic inheritance wikipedia , lookup
Genetic testing wikipedia , lookup
History of genetic engineering wikipedia , lookup
Public health genomics wikipedia , lookup
Hardy–Weinberg principle wikipedia , lookup
Dominance (genetics) wikipedia , lookup
Designer baby wikipedia , lookup
Dual inheritance theory wikipedia , lookup
Koinophilia wikipedia , lookup
Medical genetics wikipedia , lookup
Human genetic variation wikipedia , lookup
Polymorphism (biology) wikipedia , lookup
Genetic drift wikipedia , lookup
Group selection wikipedia , lookup
Behavioural genetics wikipedia , lookup
Microevolution wikipedia , lookup
Population genetics wikipedia , lookup
Lecture 12:
Quantitative
Genetics
12. Thurs., 12 Feb. 2015 - Quantitative Genetics
Vitzthum, V. J. 2003. A number no greater than the sum of its parts:
the use and abuse of heritability. Human Biology 75:539-558.
1
"A central question in biology is whether observed
variation in a particular trait is due to
environmental factors or biological factors —
sometimes expressed as the nature–nurture
debate.
Heritability is a concept which summarizes how
heritable a phenotype of interest is, in particular
with reference to the resemblance of offspring and
parents.
Heritability is both a word that is used in common
speech and a technical term in genetics, thereby
causing confusion."
Visscher, P. M., W. G. Hill, and N. R. Wray. 2008. Heritability in the genomics era —
concepts and misconceptions. Nature Reviews Genetics 9:255-266.
2
Now let's work towards the definition of
"heritability" used in quantitative genetics …
3
Mendel first sorted out the "particulate" or
discrete nature of inheritance.
He was lucky and insightful to study traits that
allowed him to see this.
4
Mendel worked with traits that came in discrete
forms, either/or traits:
e.g., round/wrinkled, yellow/green
Different alleles gave clearly distinguishable
phenotypes. "Heritability" was clear.
5
But variation in most phenotypic traits is continuous
or quantitative, not discrete like Mendel's peas.
Height distribution of 486 children
measured at age 5 and 8 years.
http://adc.bmj.com/content/79/4/318.full
6
We can reconcile "Mendelian inheritance" with
quantitative trait variation if we assume that many
different genes affect the trait
(and also that variation in environmental factors
may cause "smear" among the genetic categories
of trait variation).
7
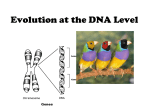
Let's imagine that 3 loci (A,B,C), each with 2 alleles,
affects the quantitative trait of human skin color.
Assuming
completely
independent
assortment
(no linkage)
Purely additive effects of alleles,
i.e., heterozygotes are exactly
intermediate between the two
homozygotes at every locus
(no dominance or epistasis)
"Punnett square"
7 different categories that grade into each other
http://www.tokresource.org/tok_classes/biobiobio/biomenu/polygenic_inheritence/index.htm
8
Let's imagine that 3 loci (A,B,C), each with 2 alleles,
affects the quantitative trait of human skin color.
Assuming
completely
independent
assortment
(no linkage)
Purely additive
effects of alleles
(no dominance
or epistasis)
7 different categories that grade into each other
http://www.tokresource.org/tok_classes/biobiobio/biomenu/polygenic_inheritence/index.htm
9
For quantitative or continuous-valued traits, like
skin color, variation among individuals within a
population is caused by:
1. genetic differences at multiple loci
2. environmental differences experienced since
fertilization of egg (or even before)
Phenotypic variance of a population is calculated
as:
(Xi - Xmean)2
-------------------------------
N-1
Standard deviation is square root of variance
10
Although standard deviations are more intuitive,
because they have the same units as the mean
(average), quantitative genetics works with
variances because variances can be added.
11
Phenotypic variance of a population can be
partitioned into various components.
Simplest partitioning:
VP = VG + VE
12
Phenotypic variance of a population can be
partitioned into various components.
Simplest partitioning:
VP = VG + VE
May also have Genotype-Environment interaction
a. which genotype has the higher phenotype
depends on the environment in which
rearing occurs
b. "reaction norm" or "norm of reaction"
= set of phenotypes produced by a
given genotype across a range of
environments (phenotypic plasticity)
13
Phenotypic variance of a population can be
partitioned into various components.
Simplest partitioning:
VP = VG + VE
May also have Genotype-Environment interaction
a. which genotype has the higher phenotype
depends on the environment in which
rearing occurs
b. "reaction norm" or "norm of reaction"
= set of phenotypes produced by a
given genotype across a range of
environments (phenotypic plasticity)
So, can add term:
VP = VG + VE + VG X E
14
Also may have genotype-environment correlation,
i.e., different genotypes tend to occur in different
environments or microenvironments:
a. genotypes might differentially select
microenvironments
b. strongest individuals obtain best territories
c. farmer takes better care of the best calves
If so, then need VP = VG + VE + VG X E + VGEcorr
This can be avoided by making measurements in
controlled environments, with little or no variation
among where individuals live, hence eliminating
sources of variance.
15
However, resulting numbers may or may not be
relevant to natural populations.
Some workers do make estimates in nature,
especially for sessile organisms, such as plants,
or animals that use nests.
Many bird populations have been studied with
artificial nest boxes.
In many cases, for practical reasons, workers
often just lump VG X E and VGEcorr into VE,
i.e., they do not try to estimate them separately.
16
Broad-sense heritability = VG/VP
But not all genetic variance can be passed on to
offspring, only the additive genetic variance.
So, a more useful partitioning for some purposes
is:
VP = VA + VD + VI + VE + VG X E + VGEcorr
VA = variance caused by additive effects of alleles
at all relevant loci
VD = variance caused by dominance deviations,
i.e., non-additive interactions between alleles
at a single locus
VI = variance caused by epistatic deviations,
i.e., non-additive interactions between alleles
at different loci
17
What do we mean by additive effects of alleles?
● Many different genetic loci affect a complex trait
● At each locus, we might have multiple alleles:
"+" alleles
"–" alleles
"o" alleles
● For body mass, + alleles might be those that
increase appetite, increase secretion of growth
hormone, reduce activity levels, etc.
●
●
– alleles would tend to do the opposite
o alleles would have no effect on these lowerlevel (subordinate) traits
18
What do we mean by additive effects of alleles?
● The effects of an allele at one locus do not
depend on what alleles are at other loci
● A large individual will have mostly + alleles at
most loci
● A small individual will have mostly – alleles at
most loci
● Next slide shows hypothetical examples
for 14 loci …
19
What do we mean by additive effects of alleles?
● Genotype of Large Individual
(genetic score = +21)
+–++o++++o++++
++o+++–+++++++
● Genotype of an "Average" Individual
(genetic score = 0)
–+ooooo–oooooo
oo+oooo–oooo+o
● Genotype of Small Individual
(genetic score = –18)
––––+o–––––o––
+–––o–––+–o–––
These would be the genetic propensities of these individuals.
Could be masked or amplified by environmental effects.
20
Narrow-sense heritability = VA/VP
Narrow-sense heritability is viewed as the single
most important descriptive statistic about the
quantitative genetics of a given trait in a given
population.
To a first approximation, it indicates the (shortterm) evolutionary potential of the trait.
How do we estimate narrow-sense heritability?
Resemblance of relatives: phenotypic similarity
will be correlated with genetic relatedness.
21
Fisher, R. A. 1918. The correlation between relatives on the
supposition of Mendelian inheritance. Transactions of the Royal
Society of Edinburgh 52:399-433.
Fisher_Young_Man_Photograph courtesy of Professor A W F Edwards by kind permission of Joan Fisher Box.jpg
22
Fraction of Shared Influence
Different
kinds of
relatives
share
different
kinds of
genetic
effects to
varying
degrees.
Relatives
VA
VD
VI
clones
1
1
1
full sibs
1/2
1/4
1/4
half sibs
1/4
0
1/16
first cousins
1/8
0
1/64
double first cousins
1/4
1/4
1/16
one parent-offspring
1/2
0
1/4
grandparent-grandchild
1/4
0
1/16
aunt-niece
1/4
0
1/16
uncle-nephew
1/4
0
1/16
23
Fraction of Shared Influence
By comparing
how strongly
different kinds of
relatives
resemble each
other for some
trait, you can, by
subtraction,
estimate other
variance
components,
such as
dominance.
Relatives
VA
VD
VI
clones
1
1
1
full sibs
1/2
1/4
1/4
half sibs
1/4
0
1/16
first cousins
1/8
0
1/64
double first cousins
1/4
1/4
1/16
one parent-offspring
1/2
0
1/4
grandparent-grandchild
1/4
0
1/16
aunt-niece
1/4
0
1/16
uncle-nephew
1/4
0
1/16
24
Fraction of Shared Influence
A common way
to estimate
additive genetic
effects is to
compare
parents with
their offspring
(measured at
the same age).
Relatives
VA
VD
VI
clones
1
1
1
full sibs
1/2
1/4
1/4
half sibs
1/4
0
1/16
first cousins
1/8
0
1/64
double first cousins
1/4
1/4
1/16
one parent-offspring
1/2
0
1/4
grandparent-grandchild
1/4
0
1/16
aunt-niece
1/4
0
1/16
uncle-nephew
1/4
0
1/16
25
Average Value of Offspring
Least-squares linear regression
slope = estimate of narrow-sense
heritability = 0.5615 in this example
3
N = 50
2
If have no epistasis,
and also no maternal
effects or commonfamily environmental
effects.
1
0
-1
Y = 0.5615*X - 0.0424
2
R = 0.2676
-2
-3
-3
-2
-1
0
1
2
3
Average Value of Parents
26
Average Value of Offspring
Repeatability Generally Sets an Upper Limit to Heritability
3
Low repeatability
would add a lot of
"jitter" to the data
points and reduce
the slope.
2
1
0
-1
-2
-3
-3
-2
-1
0
1
2
Low repeatability
can be caused by
high measurement
error per se, as
well as biological
factors that cause
a trait to fluctuate
over time (e.g.,
3 blood pressure).
Average Value of Parents
27
Narrow-sense heritability would be
estimated as 0.18 (assuming no maternal
effects or common-family environmental
effects, and no epistasis). 95% confidence
interval ranges from 0.057 to 0.311.
Swallow, J. G., P. A. Carter, and T. Garland, Jr. 1998. Artificial selection for increased wheel-running behavior in house mice.
Behavior Genetics 28:227-237.
28
Heritability of beak size in a Darwin's Finch (Geospiza fortis)
Boag, P. T. 1983. The heritability
of external morphology in
Darwin's ground finches
(Geospiza) on Isla Daphne Major,
Galapagos. Evolution 37:877-894.
Darwin's Finches in the Galapagos islands include Geospiza fortis (nutcracker finch), which has evolved a deep bill for
cracking seeds. Each point shows the mean offspring bill depth and its corresonding midparent value (the average of
the two parents). The relation between between these measures in 1976 (red circles) had a slope of 0.82 (red line). A
drought in 1978 produced tougher seeds with lower water content: finches with larger beaks (hence greater cracking
strength) were more likely to survive. Heritability remains quite similar in 1978 (slope of blue line = 0.74, which is
approximately parallel to red line): the mean beak size increased (blue line displaced upward ~0.5 mm) and no birds with
beaks <9 mm survived (blue circles). Although beak size has constant high heritability, this does not mean that the trait
is constant: beak size in any one year is highly variable (note ranges of axes), and varies when the environment changes
29
(upward displacement of slope beween years). [Modified from text material © 2010 by Steven M. Carr]
In principle, can use any relatives,
just need to know the expected causes of
resemblance.
For example, in an organism that had no paternal
care, might measure offspring and only the
fathers:
double the regression slope to estimate
narrow-sense heritability.
30
Another common "breeding design" is to mate
each father (sire) with multiple mothers (dams)
and measure trait of interest in the offspring only.
This half-sib, full-sib breeding design allows
estimation of narrow-sense heritability.
In particular, the among-sire component of
variance is proportional to additive genetic effects
(if have no non-genetic paternal effects).
31
Current state-of-the-art analytical techniques allow use
of pedigrees to determine the expected resemblances
for any sorts of relatives ("animal model"). The dairy cattle
industry has
millions of
animals in
pedigrees, along
with measures of
milk produciton.
http://1.bp.blogspot.com/-KZtTHgTZhSA/UZG73jom5_I/AAAAAAAABrM/tte0aTmq7zE/s1600/DarwinPedigree.gif
http://phylonetworks.blogspot.com/2013/05/charles-darwins-family-pedigree-network.html
32
Narrow-sense heritability can also be
estimated from a selective breeding
experiment.
Let's see how …
33
The breeder's equation says adaptive
phenotypic evolution consists of two
parts:
r = h2 s
r = response to selection
= evolution from one generation to
the next
= change in the phenotypic mean of
a population from one generation
to the next
34
r = h2 s
h2 = narrow-sense heritability
= how much of the phenotypic
variation in a population is
caused by genetic effects that
can be passed on from parents
to their offspring
additive genetic variance
= -------------------------------------total phenotypic variance
35
r = h2 s
s = directional selection differential
= difference in mean phenotype
between the original whole
population before selection and
the mean of the individuals who
actually breed to produce the next
generation
36
r = h2 s
Now let's look at this whole equation
graphically …
37
Population Before Selection
Consider the
distribution of a
phenotypic trait
within a population
MeanBefore
38
Population Before Selection
MeanBefore
Population After Selection
MeanAfter
Now imagine that a
selective event occurs,
e.g., a winter ice storm that
kills most of the individuals
in a bird population
(Bumpus 1899). In this
example, the larger birds
survive.
s = MeanAfter - MeanBefore
39
Population Before Selection
MeanBefore
Population After Selection
MeanAfter
This subset of
"selected"
individuals
survives to breed
40
Population Before Selection
MeanBefore
Population After Selection
MeanAfter
Next Generation
MeanNext Generation
The distribution of
the trait in their
offspring might
look like this
Exactly where depends
on inheritance because
r = h2 s
41
Population Before Selection
r = h2 s
MeanBefore
Population After Selection
MeanAfter
Next Generation
s = MeanAfter - MeanBefore
r = MeanNext Generation MeanBefore
h2 = r/s
So this is one way to estimate
the narrow-sense heritability.
MeanNext Generation
Usually called the
realized heritability.
42
Realized heritability
estimated from a selective
breeding experiment for
wheel running in mice
Swallow, J. G., P. A. Carter, and T. Garland, Jr.
1998. Artificial selection for increased wheelrunning behavior in house mice.
Behavior Genetics 28:227-237.
We will go into this later.
43
Can we see any general
patterns in estimates of
heritabilities for different
types of traits?
44
Life History
(lowest)
"The data set comprises
1,120 narrow sense
heritability estimates
collected from 140
sources, representing 75
species. … estimates for
the Drosophila genus were
excluded from the present
study as they are treated
elsewhere (Roff and
Mousseau, 1987)."
Morphology
(highest)
"The results indicate that [heritabilities of] life history traits are
generally much lower than morphological traits, and that
behavioural and physiological traits tend to fall in the middle."
Mousseau, T. A., and D. A. Roff. 1987. Natural selection and the heritability of fitness components. Heredity 59:181-197.
45
Why might
morphological
traits tend to
have high
heritabilities?
Life History
(lowest)
Morphology
(highest)
More genetic
influence (maybe
because they are not
so selectively
important)
Less day-to-day
fluctuation caused
by biological factors
Less measurement
error
Mousseau, T. A., and D. A. Roff. 1987. Natural selection and the heritability of fitness components. Heredity 59:181-197.
46
"on average, heritability estimates
are larger for morphological traits
than for fitness-related traits, and
that heritability tends to be larger in
better environments when compared
with poorer environments."
Visscher, P. M., W. G. Hill, and N. R. Wray. 2008. Heritability in the genomics era — concepts and misconceptions.
Nature Reviews Genetics 9:255-266.
47
Box 4 | The heritability of IQ controversy
Nowhere has the debate about nature and nurture been so
controversial as in the study of mental ability in humans.
In one meta-analysis of a number of twin studies the modelling
of maternal effects implied a narrow-sense heritability of only
0.3 and an estimate of broad-sense heritability of 0.5.
Therefore, we can conclude from the wealth of empirical data
currently available that the resemblance between relatives is
large and consistent with the hypothesis that
a large proportion of the variation in IQ between individuals
within a population is associated with
additive genetic factors.
So, about half of the among-individual variation in IQ in a given human population is
probably related to non-genetic factors. Thus, potentially we could do a lot to improve
IQ by manipulating relevant environmental factors (e.g., early-life nutrition, education).
Visscher, P. M., W. G. Hill, and N. R. Wray. 2008. Heritability in the genomics era — concepts and misconceptions.
Nature Reviews Genetics 9:255-266.
48
When selection acts on a trait, it will tend to:
1. increase frequency of alleles with additive
effects in the favored direction
2. decrease frequency of alleles with additive
effects in the "wrong" direction
3. increase frequency of alleles with dominance
effects in the favored direction
This leads to predictions about the genetic
architecture of traits ...
49
Traits that have been under (strong)
directional selection should exhibit
relatively low narrow-sense heritabilities.
This could explain why life-history traits
tend to have low heritabilities.
If we cross a population that has been under
selection for high values of a particular
trait with a population that has not been
under selection, then the F1 offspring
should tend to resemble the parents from
the selected population.
50
It is presumed that wild house mice have generally been under directional selection for
high activity levels. A cross of wild house mice with laboratory house mice found
that, after the first few days, wheel running of the F1 animals was the same as that of
the wild mice, thus indicating net dominance in the direction of high wheel running.
Dohm, M. R., C. S. Richardson, and T. Garland, Jr. 1994. Exercise physiology of wild and random-bred laboratory house mice and their reciprocal hybrids.
American Journal of Physiology 267 (Regulatory Integrative Comp. Physiol. 36):R1098-R1108.
51
VO2max of the F1 animals was also the same as that of the wild mice, indicating
net dominance in the direction of high VO2max. That makes sense, as it would go
along with high activity levels. It is not clear what body size selection may have favored
in wild house mice, but the population studied was smaller in size than the lab house
mice with which they were crossed. Body mass of the F1 animals was intermediate
between wild and lab mice,
indicating
no net dominance.
Dohm, M. R., C. S. Richardson, and T. Garland, Jr. 1994. Exercise physiology of wild and random-bred laboratory house mice and their reciprocal hybrids.
American Journal of Physiology 267 (Regulatory Integrative Comp. Physiol. 36):R1098-R1108.
52
This equation describes the response to
directional selection of a single
phenotypic trait, given its narrow-sense
heritability and the intensity of selection:
r = h2 s
However, organisms comprise many
phenotypic traits, and they may be
correlated.
Therefore, selection that affects one may
affect another.
53
3
Imagine two
traits that are
positively
correlated in a
population
before selection.
2
Trait B
1
0
-1
-2
-3
-3
-2
-1
0
1
2
3
Trait A
54
3
Die
Imagine that
selection
eliminates all
individuals with
values < 1.0 for
Trait A.
Survive
2
Trait B
1
0
-1
-2
-3
-3
-2
-1
0
1
2
3
Trait A
55
3
Die
Imagine that
selection
eliminates all
individuals with
values < 1.0 for
Trait A.
Survive
2
Trait B
1
These survivors
will also have
values for trait B
that tend to be
greater than the
mean for the
whole population
before selection.
0
-1
-2
-3
-3
-2
-1
0
1
2
3
Trait A
56
Trait B in Offspring
In addition, traits may be genetically correlated,
3
i.e., tend to run together in families
Therefore, to
understand
2
phenotypic
evolution, we
1
need to
consider both
phenotypic
0
and additive
genetic
-1
correlations
between traits.
-2
We need a
multi-trait
version of the
-3
-3
-2
-1
0
1
2
3 breeder's
equation.
Trait A in Parents
57
We will discuss the multivariate version of the breeder's
equation when we talk about Measuring Selection
in the Wild (174-15-Winter_17_3-March_Selection_in_the_Wild.ppt).
For now, just be aware that it includes genetic
correlations between traits instead of just the heritability
of a single trait.
Stopped here 12
Feb. 2015 = end
of material for
2nd Exam.
58
Genetic correlations can either slow (constrain) or
accelerate (facilitate) phenotypic evolution, depending on
whether they are in the same direction as selection.
Genetic correlations are caused by pleiotropy (one gene
affects > 1 trait) and/or by linkage disequilibrium (nonrandom associations among alleles at different loci during
the gametic phase), one cause of which is physical
linkage on chromosomes.
Pleiotropy is generally thought to be the more important
cause.
Genetic correlations can themselves evolve in response
to selection because alleles with different pleiotropic
effects may be favored.
Thus, a genetic correlation between two traits might
indicate the action of past correlated selection on the two
traits (http://www.bio.tamu.edu/users/ajones/gmatrixonline/gmatrixonline.html).
59
Definitions:
Phenotype = any measurable trait (height,
metabolic rate, I.Q.)
Genotype = genetic material, usually DNA, actual
set of genes on chromosomes
Genotype (zygote) is translated into phenotype
(adult) through development during an organism's
ontogeny, and subject to many environmental
effects
60
Definitions:
Natural Selection = individual variation in
Darwinian fitness that is correlated with variation
in one or more phenotypic traits.
Darwinian Fitness (simply) = number of offspring
left to the next generation by a given individual
(measure at same stage, zygote-to-zygote best,
difficult in practice)
Components of Darwinian Fitness, e.g.,
survivorship, fecundity, # of mates
These are often studied because it is too difficult to measure
lifetime reproductive success.
61
This lecture was too short by about 15 minutes in 2011.
I added about 6 slides, and it was too short by about 20
minutes in 2012!
This lecture was too short by about 15 minutes in 2013,
but I had not yet moved the definitions slides in here!!!!
This is end of material for Midterm Exam #2 in Spring 2012.
Extra Slides Follow
Add more on examples of genetic correlations, pleiotropy,
hormonal pleiotropy, etc. --> but that is in the next lecture …
Add modern examples with animal model analyses?
62
Updates to be Done for 2011 Spring for QG:
M:\PowerPoint\Quantitative_Genetics_from_NIU.ppt
Visscher, P. M., W. G. Hill, and N. R. Wray. 2008. Heritability in the genomics era — concepts and misconceptions. Nature Reviews
Genetics 9:255-266.
http://www.husdyr.kvl.dk/htm/kc/popgen/genetics/6/4.htm
http://www.mun.ca/biology/scarr/2900_Natural_Selection_in_the_Wild.html
George W Gilchrist and Raymond B Huey. 1999. The direct response of Drosophila melanogaster to selection on knockdown
temperature. Heredity 83:15-29. doi:10.1038/sj.hdy.6885330
http://www.nature.com/scitable/topicpage/multifactorial-inheritanceand-genetic-disease-919
Citation: Lobo, I. (2008) Multifactorial inheritance and genetic
disease. Nature Education 1(1):5
http://www.tutorvista.com/content/biology/biology-iii/heredity-andvariation/quantitative-inheritance.php
63
Nice figures:
Gary T Miller, William T Starmer and Scott Pitnick. 2001. Quantitative genetics of seminal receptacle length in Drosophila
melanogaster. Heredity 87:25-32. doi:10.1046/j.1365-2540.2001.00903.x
Fig. 1 Response in (a) the first
and (b) the second high
(squares) and low (circles) SR
selection lines, and the response
in (c) the first and (d) the second
experiments as a function of
cumulative selection differential.
The heritability is twice the
slope (m) of the regression.
Asterisks indicate the first
generation of relaxed selection
(see text for details).
64
Population Before Selection
MeanBefore
Population After Selection
These
Individuals
Breed
MeanAfter
Next Generation
MeanNext Generation
65
Midterm 1 Fall 2004
N
Mean SD
Min
Max
21 UG
82.6
11.64 58.5 98.0
8 Grads
87.5
6.84 74.5 94.0
All 29
84.0
10.65
ANOVA of UG vs. Grads,
2-tailed P = 0.278
Levene’s test to compare
variances, 2-tailed P = 0.063
66
Boag, P. T. 1983. The heritability
of external morphology in
Darwin's ground finches
(Geospiza) on Isla Daphne Major,
Galapagos. Evolution 37:877-894.
67
Boag, P. T. 1983. The heritability of external morphology in Darwin's ground finches (Geospiza) on Isla Daphne Major, Galapagos. Evolution
37:877-894.
68
3. for higher-level phenotypic traits, many allelic variants
must be almost selectively neutral, because:
a. most phenotypic traits are affected by many genes
= polygenic characters, polygenic inheritance
b. any individual gene (and its alleles) has a small effect
c. if stabilizing selection is usually
operating, then an allele that increases body size
will be advantageous in a small individual, and
vice versa, and so neutral overall
d. environment and hence selection may fluctuate, almost
stochastically, so net selection over many generations
may be around zero
69
70
Variation in most
phenotypic traits is
continuous or
quantitative, not
discrete like Mendel's
peas.
71