* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Amino Acids, Proteins and Enzymes
Survey
Document related concepts
Western blot wikipedia , lookup
Interactome wikipedia , lookup
Fatty acid synthesis wikipedia , lookup
Two-hybrid screening wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Nucleic acid analogue wikipedia , lookup
Protein–protein interaction wikipedia , lookup
Point mutation wikipedia , lookup
Metalloprotein wikipedia , lookup
Ribosomally synthesized and post-translationally modified peptides wikipedia , lookup
Peptide synthesis wikipedia , lookup
Genetic code wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Proteolysis wikipedia , lookup
Transcript
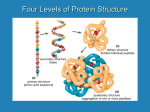
Chapter 18 Amino Acids, Proteins and Enzymes Amino Acids • 20 amino acids; all are α-amino acids: 20 Amino Acids • Essential AA’s – – – – – – – – – – Isoleucine Leucine Lysine Methionine Phenylalanine Threonine Tryptophan Valine Arginine Histidine • Non-Essential AA’s – – – – – – – – – – Alanine Asparagine Aspartate Cysteine Glutamate Glutamine Glycine Proline Serine Tyrosine • Non-protein significance of AA’s – Tryptophan precursor for serotonin, melatonin and niacin Serotonin Melatonin Tryptophan Niacin – Glycine is a precursor for porphyrins such as heme Glycine Porphyrin Heme B ‘Non-Protein’ Amino Acids • β-Alanine – Carnosine • β-alanyl-L-histidine – Anserine • β-alanyl-N1-methyl-L-histidine – Glutathione • γ-L-glutamyl-L-cysteinylglycine Classification of AA R groups (Pg. 549) • Nonpolar Side Chains – Proline: a secondary amine – Methionine: contains S – Tryptophan & Phenylalanine: aromatic rings • Polar, Neutral Side Chains – Cysteine: S-H group – Tyrosine: aromatic ring • Acidic Side Chains • Basic Side Chains Zwitterions Although -amino acids are commonly written in the unionized form, they are more properly written in the zwitterion (internal salt) form. O R-CH-COH O R-CH-CO- NH2 Un-ionized form NH3 + Zwitterion COOH donates H to NH2 - Unionized form doesn’t exist in aqueous soln. or solid state - All have high melting pts - All are soluble in water 21Isoelectric Point • Isoelectric point, pI: the pH at which the majority of molecules of a compound in solution have no net charge © 2003 Thomson Learning, Inc. All rights reserved N onpolar & p olar side chains alanine asp aragine cys teine glutamin e glycin e is oleucine leucin e methionine p henylalanine p roline serine th reonine tyrosine tryptop han valine pI 6.02 5.41 5.02 5.65 5.97 6.02 6.02 5.74 5.48 6.30 5.68 6.53 5.63 5.89 5.97 Acid ic Side Chain s pI aspartic acid 2.98 glutamic acid 3.08 Basic Side Chains arginine histid ine lysin e pI 10.76 7.64 9.74 21-8 Handedness or Chirality - - COO H NH3 + COO + H3 N CH3 D-A lanine H OH CH2 OH D -Glycerald ehyde H CH3 L-Alan ine CHO the n aturally occu rring form the n aturally occu rring form “Left Handed” CHO HO H CH2 OH Stereocenter L-Glycerald ehyde All but one of the AA’s are of the L configuration, which means that their configuration around the central Carbon is similar to that of Lglyceraldehyde. Enantiomers • Enantiomers (optical isomers): the 2 mirror image forms of a chiral molecule – Same formula but different arrangement • ALL enantiomers are stereoisomers – Same formula, same chemical groups (COOH, NH3, etc.) but different spatial arrangement (like cis-trans) • Enantiomeric pairs differ in effect on polarized light. Blue represents which atom? Red? White? Grey? Which AA is this? (Note: this is not the zwitterionic form) Peptides and Polypeptides • Peptide bond - Review • Naming Conventions – Amino end (N-terminal)Carboxyl end (CTerminal) – ‘yl’ added to name except to C-Terminal AA – Ser-gly-ala not the same as Ala-gly-ser • Primary Structure = AA sequence 21Primary Structure • Primary structure: the sequence of amino acids in a polypeptide chain • The number peptides derived from the 20 proteinderived amino acids is enormous • there are 20 x 20 = 400 dipeptides possible • there are 20 x 20 x 20 = 8000 tripeptides possible • the number of peptides possible for a chain of n amino acids is 20n • for a small protein of 60 amino acids, the number of proteins possible is 2060 = 1078, which is possibly greater than the number of atoms in the universe! © 2003 Thomson Learning, Inc. All rights reserved 21-13 21Primary Structure • Just how important is the exact amino acid sequence? • human insulin consists of two polypeptide chains having a total of 51 amino acids; the two chains are connected by disulfide bonds • in the table are differences between four types of insulin Human Cow Hog Sh eep © 2003 Thomson Learning, Inc. All rights reserved A Chain p ositions 8-9-10 B Chain p os ition 30 -Thr-Ser-Ile-A la-Ser-Val-Thr-Ser-Ile-Ala-Gly-Val- -Thr -Ala -Ala -Ala 21-14 1o Structure Crucial to Function • Hemoglobin and Sickle Cell Anemia – Normal Hb: -Thr-Pro-Glu-Glu-Lys-Ala Position: 4 5 6 7 8 9 – Sickle Cell: -Thr-Pro-Val-Glu-Lys-Ala 21Primary Structure • vasopressin and oxytocin are both nonapeptides but have quite different biological functions • vasopressin is an antidiuretic hormone • oxytocin affects contractions of the uterus in childbirth and the muscles of the breast that aid in the secretion of milk Cys S S Cys Pro Gly NH2 Tyr A sn Ph e Gln Vas op res sin © 2003 Thomson Learning, Inc. All rights reserved Cys S S Cys Pro Leu NH2 Tyr A sn Ile Gln Oxytocin 21-16 Primary Structure • The ‘linear’ AA sequence • Protein ‘Backbone’ consists of the alpha C, amino N and Carboxyl C. The 6 atoms, from alpha-C to alpha-C are arranged within an imaginary plane. – Important in 2o structure Shape-Determining Interactions in Proteins • H-bonding along backbone • R-group interactions – H-bonding – Hydrophobic interactions – Salt bridges – Covalent bonds • S-S 21Secondary Structure • Secondary structure: conformations of amino acids in localized regions of a polypeptide chain • the most common types of secondary structure are helix and b-pleated sheet • -helix: a type of secondary structure in which a section of polypeptide chain coils into a spiral, most commonly a right-handed spiral • b-pleated sheet: a type of secondary structure in which two polypeptide chains or sections of the same polypeptide chain align parallel to each other; the chains may be parallel or antiparallel Interaction: H-bonding along backbone © 2003 Thomson Learning, Inc. All rights reserved 21-19 21-Helix Grey = C Blue = N Red = O Yellow = Rgroup White = H Hydrogen bonds are between the C=O of peptide bond and the H-N of another peptide linkage 4 AA’s further along the chain. © 2003 Thomson Learning, Inc. All rights reserved 21-20 21b-Pleated Sheet Grey = C Blue = N Red = O Yellow = Rgroup White = H © 2003 Thomson Learning, Inc. All rights reserved 21-21 Beta-pleated sheet can be parallel or anti-parallel Fibrous Proteins • Fibrous Proteins (water-insoluble) – Water Insoluble – Secondary Structure determines nature • Ex. Alpha-keratins – Hair, fingernails, wool – Composition of alpha-helixes; multiple strands twist together • Ex. Fibroin – Silk – Composed of b-sheets 21Collagen Triple Helix © 2003 Thomson Learning, Inc. All rights reserved 21-24 Tertiary Structure • Overall Conformation of a Peptide chain – How it folds, interactions between various AA’s along the chain. • Determined by interactions of R-groups – – – – Hydrophobic Salt-bridges H-bonds disulfide Example of a Globular Protein • Myoglobin – Conjugated Globular Protein • Contains a heme group 21Quaternary Structure • Quaternary structure: the arrangement of polypeptide chains into a noncovalently bonded aggregation • the individual chains are held in together by hydrogen bonds, salt bridges, and hydrophobic interactions • Hemoglobin • adult hemoglobin: two alpha chains of 141 amino acids each, and two beta chains of 146 amino acids each • each chain surrounds an iron-containing heme unit • fetal hemoglobin: two alpha chains and two gamma chains; fetal hemoglobin has a greater affinity for oxygen than does adult hemoglobin © 2003 Thomson Learning, Inc. All rights reserved 21-28 21Hemoglobin © 2003 Thomson Learning, Inc. All rights reserved 21-29 Chemical Properties of Proteins • Hydrolysis – breaking of peptide bonds – By enzymes – By acid • Denaturation – disruption of non-covalent bonds – – – – Heat Mechanical agitation Detergents pH change • Disrupt salt-bridges – Solvents • Disrupt hydrophobic interactions