* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download 1 Pathophysiology Name Chapter 24: Alterations of Cardiovascular
Cardiovascular disease wikipedia , lookup
Cardiac contractility modulation wikipedia , lookup
Electrocardiography wikipedia , lookup
Management of acute coronary syndrome wikipedia , lookup
Jatene procedure wikipedia , lookup
Infective endocarditis wikipedia , lookup
Coronary artery disease wikipedia , lookup
Aortic stenosis wikipedia , lookup
Hypertrophic cardiomyopathy wikipedia , lookup
Heart failure wikipedia , lookup
Antihypertensive drug wikipedia , lookup
Artificial heart valve wikipedia , lookup
Cardiac surgery wikipedia , lookup
Myocardial infarction wikipedia , lookup
Arrhythmogenic right ventricular dysplasia wikipedia , lookup
Lutembacher's syndrome wikipedia , lookup
Dextro-Transposition of the great arteries wikipedia , lookup
Rheumatic fever wikipedia , lookup
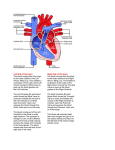
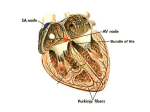
PATHOPHYSIOLOGY Name Chapter 24: Alterations of Cardiovascular Function – Part 2 Diseases of the Heart Wall, Heart Disease and Shock I. Disorders of the Heart Wall A. Disorders of the Pericardium 1. Acute pericarditis Acute inflammation of the pericardium whereby membranes become inflamed and roughened, often producing exudates. May result from infection, drug therapy, or tumors. Clinical presentation: Sudden onset of severe anterior chest pain that worsens with respiratory movement or lying down; low-grade fever; sinus tachycardia; friction rub. 2. Pericardial effusion Accumulation of fluid in the pericardial sac (cavity), usually secondary to acute pericarditis. Clinical presentation: Often asymptomatic unless cardiac compression results. Cardiac tamponade – compression of the heart by fluid in the pericardial sac, which interferes with filling of the heart. o Cardiac function may be severely impaired if the accumulation of fluid occurs rapidly and involves a large volume. o Clinical signs: increased venous pressure and congestion resulting in distention of the jugular veins, edema, and hepatomegaly; distant or muffled heart sounds, poorly palpable apical pulse, dyspnea on exertion, and dull chest pain. 3. Constrictive pericarditis Fibrous scarring and calcification of the pericardium causes the visceral and parietal pericardial layers to adhere, obliterating the pericardial cavity, resulting in encasement of the heart in a rigid shell over time. Cause: Often idiopathic but may result from infection (tuberculosis, gram positive bacteria), radiation exposure, rheumatoid arthritis, or post-surgical scarring. Clinical presentation: Asymptomatic in early stages; exercise intolerance, fatigue, dyspnea on exertion develop and worsen over time; symptoms of venous congestion; eventually compresses heart, preventing ventricular filling. B. Disorders of the Myocardium Cardiomyopathies- a diverse group of primary myocardial disorders. Usually due to underlying cardiac disease, such as hypertension or ischemic heart disease, but can be caused by infections, toxin exposure, and a variety of other disorders. Classified according to their effects on the heart wall. 2 1. Dilated cardiomyopathy (congestive cardiomyopathy) – the ventricular walls dilate and become thin, resulting in a decreased strength on contraction. 2. Hypertrophic cardiomyopathy - the ventricular myocardium thickens (hypertrophies). o Asymmetrical septal hypertrophy – genetic disorder causing enlargement of the interventricular septum. o Hypertensive (valvular hypertrophic) cardiomyopathy - develops secondary to hypertension or valvular stenosis. The ventricles enlarge in an attempt to compensate for the increased workload being placed on the heart. 3. Restrictive cardiomyopathy - increased rigidity (decreased compliance) of the myocardium from severe scarring (e.g., from radiation therapy) or from infiltration of abnormal substances into the myocardium (e.g., iron in hemochromatosis, or amyloid in amyloidosis). C. Disorders of the Endocardium 1. Valvular dysfunction – caused by conditions that damage the heart valves. May be congenital or acquired. Acquired valvular disease can be caused by inflammatory diseases, trauma, tissue degeneration, or infections. Most of the acquired valvular disorders involve the aortic and mitral valves. a. Valvular stenosis – narrowing of the valve opening. Results in increased pressure and hypertrophy of the myocardium in the heart chamber pumping into that valve. 1) Aortic stenosis – narrowing of aortic semilunar valve between left ventricle and aorta. Causes: congenital (children), rheumatic heart disease (RHD) (children and adults), or due to degeneration and calcification of the valve with aging (elderly). Incomplete emptying of left ventricle results in its hypertrophy and eventual failure. 2) Mitral stenosis – narrowing of mitral valve between left atrium and left ventricle. Causes: congenital (children); RHD or bacterial endocarditis. Results in hypertrophy and dilation of left atrium due to incomplete emptying; blood backs up into pulmonary system; systemic outflow is decreased; atrial fibrillations and pulmonary edema occur. b. Valvular regurgitation – incomplete closure of a valve. Allows blood to leak backward into the heart chamber pumping into that valve. Results in an increase in volume in the heart chambers and dilation of the myocardium. 1) Aortic regurgitation – aortic valve fails to close properly; blood leaks back to left ventricle. Causes: associated with Marfan syndrome in youths; in adults caused by RHD or bacterial endocarditis, syphilis, hypertension, or atherosclerosis. Left ventricle dilates and hypertrophies, with eventual failure of left ventricle. 3 2) Mitral regurgitation – mitral valve fails to close properly; blood leaks back into left atrium. Causes: associated with Marfan syndrome in youths; in adults caused by mitral valve prolapse, RHD or bacterial endocarditis, congestive cardiomyopathy, or atherosclerosis. Left atrium dilates; left ventricle dilates (to accept larger volume from left atrium) and hypertrophies (to pump enough blood into aorta, despite leakage through mitral valve). Blood backs up into pulmonary system; atrial fibrillations and pulmonary edema occur. Left ventricle eventually fails. 3) Tricuspid regurgitation – tricuspid valve fails to close properly: blood leaks back into right atrium. Causes: due to failure and dilation of right ventricle, RHD or infective endocarditis. Right atrium dilates; right ventricle dilates and hypertrophies; eventually right ventricle fails. c. Mitral valve prolapse syndrome (MVPS)- one or both of the cusps of the mitral valve billow upward (prolapse) into the left atrium during systole. Most common valve disorder in the United States, especially in young women. Mitral valve leaflets do not position themselves properly during systole. MVPS may be completely asymptomatic or can result in unpredictable symptoms. Afflicted valves are at greater risk for developing infective endocarditis. 2. Acute Rheumatic Fever and Rheumatic Heart Disease a. Rheumatic fever (RF) – a diffuse, inflammatory disease caused by a delayed immune response to pharyngeal infection by the group A beta-hemolytic streptococci. Antibiotic therapy given within first 9 days of strep infection usually prevents RF. Acute RF – fever and inflammation of the joints, skin, nervous system, and heart o Caused by cross reaction of antibodies against strep with self-antigens on heart, muscle, and brain and joints, resulting in an autoimmune response that inflames and potentially scars these tissues. If left untreated, rheumatic fever may lead to rheumatic heart disease. b. Rheumatic Heart Disease (RHD) 10% of individuals with rheumatic fever develop rheumatic heart disease (RHD). RHD refers to cardiac complications of rheumatic fever, primarily endocarditis and damage to the heart valves, although myocarditis may also occur. Damage occurs because antibodies bind to the lining of the heart, surface of valves, and connective tissue within the myocardium and trigger an autoimmune response. 4 3. Infective Endocarditis - infection and inflammation of the endocardium and often cardiac valves. Causes: bacteria (most common); sometimes viruses, fungi, rickettsiae, and parasites. Pathophysiology: o The endocardium is “prepared,” usually by endothelial damage, for microorganism colonization. o Blood-borne microorganisms adhere to the damaged endocardial surface. o These proliferate and promote the propagation of infective endocardial vegetations. o Vegetations are clumps of microorganisms surrounded by fibrin, which prevents inflammatory cells from reaching them. In the mildest cases, valvular function may be slightly impaired by vegetations that collect on the valve leaflets. If left unchecked, severe valve abnormalities, chronic bacteremia, and systemic emboli may occur as vegetations break off the valve surface and travel through the bloodstream. Antibiotic therapy can limit the extension of this disease. II. Manifestations of Heart Disease A. Dysrhythmias Disturbance of the heart rhythm. Range from occasional “missed” or rapid beats to severe disturbances that affect the pumping ability of the heart. Diagnosed and evaluated by doing an electrocardiogram. Different dysrhythmias are characteristic of specific problems in the myocardium and conduction system. Can be caused by an abnormal rate of impulse generation or abnormal impulse conduction. B. Heart Failure General term used to describe several types of cardiac dysfunction that result in inadequate perfusion of tissues with oxygen and nutrients. Cardiac output (CO) = stroke volume x heart rate. Stroke volume is determined by: o Contractility – the strength of cardiac contraction. o Reduced by disease processes that affect muscle cells, including ischemia. Preload (or left ventricular end-diastolic volume, LVEDV) – the amount of blood in the ventricle just before it contracts. o Reduced by poor venous return; increased when fluid volume goes up. Afterload- pressure the heart must work against to push blood out of the heart. Primarily determined by peripheral vascular resistance (blood pressure). 5 1. Left heart failure Also call congestive heart failure (CHF) The most common pattern of heart failure is for one ventricle, usually the left, to fail first. o Causes: o The left ventricle has a heavier workload, as well as a greater requirement for oxygen. Myocardial ischemia or infarction, cardiomyopathies, valve disorders, hypertension, etc. Pathophysiology: o Left ventricle is faced with a high workload due to hypertension or valve disorders, or is weakened due to disease. In response: o Heart muscle undergoes myocardial hypertrophy and ventricular remodeling. o Results in decreased contractility which causes lower stroke volume and higher LVEDV. o The heart dilates to accommodate the higher volume so left ventricular preload increases. o Higher preload stretches the wall and causes further dilation and decreased contractility. o As the myocardium weakens, cardiac output falls, reducing systemic BP. In response: Sympathetic nervous system (SNS) releases epinephrine causing vasoconstriction and increased heart rate which raises blood pressure (and afterload). Decreased kidney perfusion causes activation of renin-angiotensin-aldosterone (RAA) system, which also causes vasoconstriction and increases fluid volume. Together these raise vascular resistance and blood pressure. Decreased cardiac output causes ischemia in all organs, as well as exertional fatigue. Pulmonary congestion and edema occur as the weakened left ventricle pumps out less and less blood, producing dyspnea and cough. 2. Right heart failure Causes: o Chronic pulmonary hypertension caused by left heart failure. o Chronic lung disease that increases the afterload of the right ventricle. o Less frequently, cardiomyopathies or MIs of the right ventricle or disorders of the tricuspid or pulmonary semiluner valves. Blood backs up into the systemic veins leading into the right atrium, causing venous congestion. Clinical manifestations: o Jugular venous distension, hepatomegaly, and peripheral edema. 3. High-output failure Inability of the heart to supply the body with oxygen and nutrients, despite adequate blood volume and normal or elevated myocardial contractility. Causes – extreme exertion, anemia, septicemia, hyperthyroidism, and thiamine deficiency. 6 4. Treatments for heart failure Prevention - dietary changes to limit sodium intake and reduce risk factors for coronary artery disease and hypertension, exercise, and weight loss. Pharmacologic therapy (just like in acute heart failure) is aimed at: o Reducing preload with diuretics (including furosemide [Lasix] and spironolactone) o Reducing afterload with vasodilators (ACE inhibitors, beta blockers) o Increasing contractility in selected patients (digoxin, pacemaker placement, myoplasty surgery) Address underlying cause of heart failure - lipid-lowering agents and percutaneous coronary intervention (PCI) (dilation of narrowed coronary arteries with a catheter). III. Shock In shock the cardiovascular system fails to perfuse the tissues adequately. This results in widespread impairment of cellular metabolism, involving positive feedback loops, that places the individual on a downward physiologic spiral leading to multiple organ dysfunction syndrome (MODS). A. Impairment of Cellular Metabolism 1. Impaired oxygen use Underperfusion of vital tissues impairs cellular metabolism due to a lack of oxygen. When this occurs, cells shift to anaerobic metabolism, which results in: o A reduction in adenosine triphosphate (ATP) production o An inability of the cells to maintain their normal membrane electrolyte pumps causing sodium, chloride, and calcium to accumulate in cells, resulting in cellular lysis. Ischemia activates the inflammatory response, causing release of cytokines, lysosomal enzymes, and activators of the clotting cascade. Anaerobic metabolism produces lactic acid, which lowers the blood and tissue pH. o Acidosis interferes with cellular metabolism and electrolyte balance. o If the shock state persists, multiple organ dysfunction develops. 2. Impaired glucose use Occurs because less glucose is delivered to the tissues, many disease states cause increased demand for glucose, and glucose uptake is impaired by many toxins and cytokines. Systemic compensation for the shock state includes the release of cortisol, thyroid hormone, and catecholamines (like epinephrine), which increase glucose production from glycogen. Once glycogen stores are used up, gluconeogenesis is activated to continue glucose production, but at the expense of protein depletion. 7 o As proteins are broken down they release alanine, which can contribute to lactic acid production as well as the release of ammonia and urea, compounds that are toxic to cellular metabolism. o Muscle dysfunction from protein degradation contributes to multiple organ dysfunction. B. Types of Shock 1. Cardiogenic shock Decreased cardiac output and evidence of tissue hypoxia in the presence of adequate intravascular volume. Causes: o Can occur whenever the heart is unable to generate an adequate cardiac output. o Severe left heart failure, severe MI (>40% of myocardium damaged), severe dysrhythmias, cardiac tamponade, tension pneumothorax, etc. Clinical Manifestations: o Pulmonary edema, which causes dyspnea and inspiratory crackles. o Hypotension and severe underperfusion of vital organs, which causes poor capillary refill, cyanosis, cold clammy extremities, and oliguria. Treatment: o Use of pressors such as dobutamine (which increase contractility), careful fluid management, and treatment of the underlying cardiac abnormality if possible. 2. Hypovolemic shock Causes: o Any condition that can result in the loss of greater than 15% of the intravascular volume. o Dehydration, hemorrhage, burns, severe diarrhea, overuse of diuretics or dialysis, or extreme hyperglycemia or hypercalcemia. Compensatory mechanisms: o Increased heart rate (tachycardia) o Activation of the renin-angiotensin-aldosterone system and release of antidiuretic hormone o Increased peripheral vascular resistance (vasoconstriction) o Movement of fluid from interstitial compartment to bloodstream If these are inadequate to maintain tissue perfusion, then shock ensues. Clinical manifestations: o Same as cardiogenic shock but without pulmonary edema. Treatment: o Reversing the underlying loss of fluid or blood o Intravenous fluid administration. 8 Distributive shock – caused by an inability to maintain an adequate peripheral vascular resistance due to vasodilation of the systemic arteries. Includes neurogenic, anaphylactic and septic shock. 3. Neurogenic shock Causes: o Loss of sympathetic stimulation due to injury to the brain or spinal cord, or certain drugs. o Overstimulation of the parasympathetic nerves due to pain, stress, drugs, or toxins. Pathophysiology: o Arterioles lose their stimulus to maintain a constant amount of vasoconstriction (loss of vascular tone) and, therefore, peripheral vascular resistance falls. o This causes the blood volume to be redistributed to nonvital organs, with a loss of blood pressure and underperfusion of tissues. Clinical manifestations: o Very low peripheral vascular resistance and bradycardia, with neurogenic insult. Treatment: o Treatment of the underlying insult. o Careful patient positioning, the administration of pressors, and appropriate fluid management. 4. Anaphylactic shock Causes: o Severe allergic reactions. Pathophysiology: o Exposure to high doses of allergen cause an immediate severe immune reaction with production of high levels of IgE. This results in: o Rapid, widespread degranulation of mast cells, with release of multiple vasoactive and inflammatory mediators, including histamine. o Profound peripheral vasodilation and increased capillary permeability cause a decrease in blood pressure and tissue perfusion. Clinical manifestations: o History of allergies, urticaria (hives), edema, bronchoconstriction causing dyspnea, decreased peripheral vascular resistance, hypotension, and increased bowel sounds. Treatment: o Removal of the allergen. o Administration of epinephrine, corticosteroids, bronchodilators, and fluids. o Mechanical ventilation may also be required. 9 5. Septic shock Causes: o Gram-positive or gram-negative bacterial infection (or occasionally, fungi or viruses) that overwhelms the body’s defense mechanisms. Serious complication of severe trauma, immunodeficiency syndromes, or for the very young or elderly. Pathophysiology: o Bacteria must enter the bloodstream. o Gram-negative organisms release endotoxin (lipopolysaccharide), whereas gram-positive organisms release exotoxin and cell wall components. o These substances activate macrophages which release inflammatory cytokines, including tumor necrosis factor-alpha (TNF-alpha), interleukins, and nitric oxide. o These substances, especially nitric oxide, cause widespread vasodilation and hypotension. o The complement and coagulation systems are also activated. o This results in what is called the systemic inflammatory response syndrome (SIRS): o Signs: fever, tachypnea, tachycardia, and leukocytosis. Numerous cell and tissue complications result, including decreased peripheral resistance, decreased cardiac output, and increased capillary permeability, all of which cause hypotension and decreased tissue perfusion. o Lactic acidosis, widespread coagulation, and activation of other inflammatory cells occur. o Hypotension can cause disruption of the intestinal lining, allowing enteric bacteria to enter the bloodstream, and thus aggravating the bacteremia. o Multiple organ dysfunction syndrome (MODS) - organ dysfunction involving many of the vital organs can become overwhelming and cause death (see below). Clinical manifestations: o Initially: fever, tachypnea, tachycardia, generalized edema and warm, sweaty skin. o As the SIRS and organ dysfunction progress, the individual may exhibit the clinical manifestations associated with multiple organ dysfunction. o Laboratory findings include leukocytosis, lactic acidosis, hypoxemia, uremia (elevated blood urea nitrogen and creatinine), and hepatic insufficiency (with elevated liver enzymes). Treatment: o Prompt administration of antibiotics, oxygen, pressors, and careful fluid management. o Activated protein C is an anti-inflammatory and anticoagulant drug which has been shown to improve outcomes in severe sepsis. 10 E. Multiple Organ Dysfunction Syndrome (MODS) Progressive dysfunction of two or more organ systems resulting from an uncontrolled inflammatory response to severe illness or injury. Causes: o Septic shock (most common), trauma, burns, and other causes of severe shock with underperfusion of vital tissues. Pathophysiology: o Injury, infection or disease cause endothelial damage, activation of the sympathetic nervous system, release of stress hormones (cortisol, epinephrine, and norepinephrine), and release of inflammatory mediators. o Inflammatory mediators activate the complement, coagulation and kinin cascades. o These cause a massive, systemic immune/inflammatory response → SIRS. o Inflammatory and clotting processes cause maldistribution of blood flow and hypermetabolism. o Oxygen delivery to the tissues decreases because: Blood is shunted past selected regional capillary beds, due to inflammatory mediators. Interstitial edema, due to increased capillary permeability, interferes with oxygen diffusion to tissue cells. o Microvascular thrombi cause capillary obstruction. Hypermetabolism occurs to meet the body's increased demands for energy, but over time this depletes oxygen and fuel supplies. o Decreased oxygen delivery plus hypermetabolism combine to create an imbalance in oxygen supply and demand, in which the reserve supply of oxygen is exhausted and the amount that can be delivered by the circulation is less than the cells need. o Tissue hypoxia with cellular acidosis and impaired cellular function ensue and result in the multiple organ failure. Common Manifestations: o Acute respiratory distress syndrome (ARDS) – dyspnea, pulmonary crackles, and cyanosis. Note: the lungs are often the first system to fail. o Disseminated intravascular coagulation (DIC) – organ infarction and bleeding o Acute tubular necrosis (ATN) – decreased urine output, azotemia, and edema o Liver failure – jaundice, abdominal distension and tenderness, and hepatic encephalopathy o Heart failure – dyspnea, cough, cyanosis and peripheral edema o Ischemic bowel – translocation of bacteria into the bloodstream o Central nervous system (CNS) dysfunction – confusion and coma Treatment: o Reversal of the primary insult if possible. o Support for organ failure including mechanical ventilation, activated protein C, dialysis, fluid and electrolyte management, enteral (preferably) or parenteral nutrition, and antibiotics.