* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Chapter 23 - Simpson County Schools
Physical organic chemistry wikipedia , lookup
Homoaromaticity wikipedia , lookup
Discodermolide wikipedia , lookup
Elias James Corey wikipedia , lookup
Organosulfur compounds wikipedia , lookup
Petasis reaction wikipedia , lookup
Kinetic resolution wikipedia , lookup
Aldol reaction wikipedia , lookup
Baylis–Hillman reaction wikipedia , lookup
Strychnine total synthesis wikipedia , lookup
Asymmetric induction wikipedia , lookup
Wolff rearrangement wikipedia , lookup
Nucleophilic acyl substitution wikipedia , lookup
Carbohydrate wikipedia , lookup
Hydroformylation wikipedia , lookup
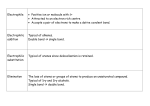
CHAPTER 23 Aldehydes & Ketones 23.1 STRUCTURES OF ALDEHYDES & KETONES Both contain the carbonyl group (C=O) Aldehyde, written linearly as CHO Ketone written linearly as CO 23.2 NAMING ALDEHYDES & KETONES IUPAC Naming of Aldehydes: Select the longest continuous chain of carbon atoms that contains the aldehyde group. The carbons of the parent chain are numbered starting with the aldehyde group. Form the parent aldehyde name by dropping the –e from the corresponding alkane and adding the suffix –al. Other groups attached to the parent chain are named and numbered as previously done. Aromatic aldehydes are named after the corresponding carboxylic acid. Thus the name benzaldehyde is from benzoic acid IUPAC Naming of Ketones: Select the longest continuous chain of carbon atoms that contains the ketone group. Form the parent name by dropping the –e from the corresponding alkane name and adding the suffix –one. If the chain is longer than four carbons, it is numbered so that the carbonyl group has the smallest possible number; this number is prefixed to the parent name of the ketone. Other groups attached to the parent chain are named and numbered as previously done. Draw the structural formulas for: 4-methylhexanal Butanedial 2-pentanone 4 methyl-3-hexanone: Draw the structural formulas for: 4-methylhexanal 1, 4-Butanedial 2-pentanone 4 methyl-3-hexanone: NAME THE FOLLOWING: NAME THE FOLLOWING: MORE NAMING: MORE NAMING: 1-phenyl-1-butanone 5-methyl-3-hexanone 23.3 BONDING & PHYSICAL PROPETIES Low MW aldehydes & ketones (<5 C) soluble in both organic solvents & water. Higher MW aldehydes & ketones (>5C) soluble in organic solvents, insoluble in water. Boiling point comparison of aldehydes, ketones & alcohols: In general…if 4 or fewer C atoms: alcohols have higher boiling points than ketones, ketones have higher boiling points than aldehydes. Rate the following molecules based on boiling points from lowest to highest: 2,2-dimethylpropane (MW 72 g/mol) 1,2-propanediol (MW 76 g/mol) 1-butanol (MW 74 g/mol) butanal (MW 72 g/mol) 23.4 CHEMICAL PROPERTIES OF ALDEHYDES & KETONES Reactions of Aldehydes and Ketones: Products of alcohol oxidation. See alcohol notes! Reduced to alcohols. Aldehydes yield primary alcohols Ketones yield secondary alcohols. Easily undergo addition due to double bond (C=O) Aldehydes oxidize readily to carboxylic acids. Ketones can’t be oxidized as readily. PRODUCTS OF ALCOHOL OXIDATION. SEE ALCOHOL NOTES! ALDEHYDES OXIDIZE READILY TO CARBOXYLIC ACIDS. Ketones can’t be oxidized as readily. Tollens, Fehling, and Benedict solutions do not give a positive test for ketones; therefore, they are used to distinguish between ketones and aldehydes…you will do this in lab! What are the IUPAC names for these compounds? ALDEHYDES OXIDIZE READILY TO CARBOXYLIC ACIDS. Ketones can’t be oxidized as readily. Tollens, Fehling, and Benedict solutions do not give a positive test for ketones; therefore, they are used to distinguish between ketones and aldehydes…you will do this in lab! What are the IUPAC names for these compounds? Ethanal ethanoic acid NAME AND DRAW THE PRIMARY OXIDATION PRODUCT FOR THE FOLLOWING REACTION: NAME AND DRAW THE PRIMARY OXIDATION PRODUCT FOR THE FOLLOWING REACTION: Cyclohexanal Cyclohexanoic acid (or cyclohexanecarboxylic acid) REDUCED TO ALCOHOLS Aldehydes yield primary alcohols, whereas ketones yield secondary alcohols. In biochemistry, aldehyde/ketone reduction is reversible! Ketosis – a metabolic condition caused by starvation or a low-carbohydrate/high protein diet. During ketosis, the liver produces low-molar-mass ketones that are water-soluble and easily dissolve in the bloodstream to be transported to tissues. At the same time, some of these ketones are reduced in the liver and the alcohol product is released in to the blood. DRAW THE ALCOHOL THAT IS THE KETONE REDUCTION PRODUCT. (NOTE 1O OR 2O) DRAW THE ALCOHOL THAT IS THE KETONE REDUCTION PRODUCT. (NOTE 1O OR 2O) EASILY UNDERGO ADDITION WITH ALCOHOLS TO FORM: hemiacetals & hemiketals – compounds that contain an alkoxy and a hydroxy group on the same carbon, and acetals & ketals- compounds that contain 2 alkoxy groups on the same carbon. Aldehydes react with alcohols to form hemiacetals: (ketones react to form hemiketals) Hemiacetals react with a second molecule of the alcohol to give an acetal: If the alcohol and carbonyl group are within the same molecule, the result is the formation of a cyclic hemiacetal (or hemiketal): Extremely important to carbohydrate chemistry due to nearly all carbohydrates are found as ringed molecules because they form intramolecular hemiacetals or hemiketals. The acetal/ketal reaction provides the means by which carbohydrates link together to form the wellknown carbohydrate polymers such as sucrose (table sugar), lactose (milk sugar) and starch THE FOLLOWING THREE SUGARS ARE CYCLIC HEMIACETALS OR HEMIKETALS Circle the hemiacetal or hemiketal carbon in each. Can we determine which are the hemiacetals/hemiketals? 23.5 COMMON ALDEHYDES & KETONES What other functional groups do you see? Formaldehyde (methanal) – highly soluble, poisonous, useful in manufacturer of polymers, biological specimen preservative. Acetaldehyde (ethanal) – produced by the liver after alcohol consumption as part one of a 2-step detoxifying process. Polymers of ethanal, such as metaldehyde is poisonous to slugs & snails and sold as an active ingredient in pesticides. Acetone (propanone) & methyl ethyl ketone (2butanone) – widely used as solvents HOMEWORK Review Questions: 1, 4, 6-8, 11 Paired Exercises 2-22, 28 (even)