* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Functional Characterization of Two Human Olfactory Receptors
Survey
Document related concepts
Transcript
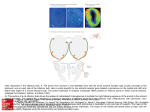
Chem. Senses 30: 195–207, 2005 doi:10.1093/chemse/bji015 Functional Characterization of Two Human Olfactory Receptors Expressed in the Baculovirus Sf9 Insect Cell System Valéry Matarazzo1*, Olivier Clot-Faybesse1*, Brice Marcet1*, Gaëlle Guiraudie-Capraz1, Boriana Atanasova2, Gérard Devauchelle3, Martine Cerutti3, Patrick Etiévant2 and Catherine Ronin1 1 UMR 6149 et GDR 2590 CNRS et Université de Provence, IFR du Cerveau, 31 Chemin J. Aiguier, F-13402 Marseille Cedex 20, France, 2Laboratoire de Recherches sur les Arômes, INRA, 17 rue Sully, BP 86510, 21065 Dijon Cedex, France and 3Station de Recherches de Pathologie Comparée, CNRS- INRA et GDR 2590, St Christol-Lez-Alès, France Correspondence to be sent to: Pr. Catherine Ronin, UMR 6149, CNRS, 31 Chemin J. Aiguier, F-13402 Marseille Cedex 20, France. Email: [email protected] *Contributed equally to this paper and should be considered as first coauthors. Abstract Olfactory receptors (ORs) are the largest member of the G-protein-coupled receptors which mediate early olfactory perception in discriminating among thousands of odorant molecules. Assigning odorous ligands to ORs is a prerequisite to gaining an understanding of the mechanisms of odorant recognition. The functional expression of ORs represents a critical step in addressing this issue. Due to limitations in heterologous expression, very few mammal ORs have been characterized, and so far only one is from human origin. Consequently, OR function still remains poorly understood, especially in humans, whose genome encodes a restricted chemosensory repertoire compared with most mammal species. In this study, we have designed cassette baculovirus vectors to coexpress human OR 17-209 or OR 17-210 with either Gaolf or Ga16 proteins in Sf9 cells. Each OR was found to be expressed at the cell surface and colocalized with both Ga proteins. Using Ca2+ imaging, we showed that OR 17-209 and OR 17210 proteins are activated by esters and ketones respectively. Odorant-induced calcium response was increased when ORs were coexpressed with Ga16 protein ,whereas coexpression with Gaolf abolished calcium signaling. This strategy has been found to overcome most of the limitations encountered when expressing an OR protein and has permitted odorant screening of functional ORs. Our approach could thus be of interest for further expression and ligand assignment of other orphan receptor proteins. Key words: baculovirus, calcium imaging, confocal microscopy, GPCR, insect cell system, olfactory receptor Introduction Early olfactory discrimination of higher vertebrates is accomplished via the recognition of odorous ligands by olfactory receptors (ORs) which belong to the G-protein-coupled receptor (GPCR) superfamily (for review, see Breer, 1994). A single OR could bind different odorants sharing the same odotope while one odorant molecule could be recognized by multiple ORs, suggesting the existence of a combinatorial receptor scheme to encode odorous entities (Malnic et al., 1999). Odorant discrimination has been shown not only to proceed through the recognition of numerous molecules of widely different structures but also to markedly vary upon odorant concentration (Kajiya et al., 2001). Over the last decade, thousand of sequences of putative ORs have been made available in the Olfactory Receptor Data Bank (http://senselab.med.yale.edu/senselab/ORDB/ default.asp), which currently contains the largest family of heptahelical receptors, covering up to 3% of the genome superfamily (for review, see Mombaerts, 2001). The human OR genome includes numerous dysfunctional sequences, or pseudogenes (Rouquier et al., 1998; Menashe et al., 2003), since mutations disrupting OR coding region have been accumulated fourfold faster than any other species sampled (Gilad et al., 2003). About 350 genes in our olfactory repertoire are potential candidates for full-length OR sequences (Zozulya et al., 2001; Malnic et al., 2004), while almost 50% are pseudogenes (Malnic et al., 2004). Such a limited repertoire is of particular biological relevance since the human ortholog corresponding to an identified mammal OR could be nonfunctional (Sharon et al., 1999; Gaillard et al., 2002). Furthermore, not all ORs play a role in perception since human OR genes are also expressed in nonolfactory tissues, like testicular tissue, where they might be Chemical Senses vol. 30 no. 3 ª The Author 2005. Published by Oxford University Press. All rights reserved. For permissions, please e-mail: [email protected] 196 V. Matarazzo et al. involved in the fertilization process (Vanti et al., 2003; Volz et al., 2003). In this respect, we have recently shown that the expression of two human OR genes, OR 17-209 and OR 17-210, was restricted to the olfactory neuroepithelium (Matarazzo et al., 2002). Both ORs are located in chromosome 17 OR gene cluster, a region of special interest as it originates from a copy of an ancestral OR repertoire (Ben-Arie et al., 1994). With their rather restricted chemosensory repertoire, humans represent a good model for understanding olfaction because of the intensive information available on olfactory faculties in Homo sapiens, gathered from earlier work (Beets, 1971) to the more recent modelling of human olfactory stimulus–response function (Chastrette et al., 1998), including data on phylogenic evolution of human ORs (see http://bioportal.weizmann.ac.il/HORDE/). Starting from gene sequences provided by genome analysis, identifying OR proteins and assigning their ligands represent a crucial step in the elucidation of the combinatorial olfactory code. However, functional properties of ORs still remain poorly understood since no more than a few dozen mammal OR proteins have been successfully obtained so far, including only one receptor of human origin. This receptor, OR 17-40, has been expressed in mammalian cells and its ligand assigned by screening of odorant libraries (Hatt et al., 1999; Wetzel et al., 1999). It was found to discriminate an aldehydic molecule, helional, between structurally related odorants. Limitation in heterologous expression is due to impaired OR protein translocation to the cell surface (Ivic et al., 2002; Lu et al., 2003) and largely accounts for the limited functional data on OR obtained so far. Among the heterologous systems available for gene expression, the Sf9 cell system has been established as a useful intact cell setting to reconstitute GPCRs for ligand binding studies (Butkerait et al., 1995; Barr et al., 1997). The choice of such a system is of particular interest for human protein expression since Sf9 cells carry out a variety of co- and post-translational processing events (Butkerait et al., 1995; Poul et al., 1995). These processing events support viable targeting of receptors and G-protein subunits to the cell membrane. In distinction of mammalian cells, the Sf9 cell model is free of potential interference with endogeneous GPCRs and the low level of endogeneous G proteins permits to test the interaction of mammalian subunits with the coexpressed receptor. Also, the insect cells can be used for the functional expression of mammalian receptors linked to changes in Ca2+ homeostasis (Hu et al., 1994). To provide tools for OR deorphanization, we have thus developed a strategy based on the expression of human OR protein in Sf9 cells together with Ga protein in order to transduce a measurable biological signal in response to odorant stimulation. We designed recombinant baculoviruses to coexpress OR 17-209 or OR 17-210 with either Gaolf protein, a subunit involved in olfactory transduction through cAMP production (Jones and Reed, 1989), or Ga16 protein, an ubiquitous subunit of the phosphoinositide pathway. Quantitative pharmacological odorant screening of both ORs has been then performed using a Ca2+ imaging method to monitor change in intracellular Ca2+ concentration in Sf9 cells. Material and methods Chimera constructs The cDNA encoding rat Gaolf protein sequence was cloned by RT-PCR using a whole-brain total RNA preparation, kindly provided by Dr. Gilles Toumaniantz (IPMC, Nice, France). RNAs (4 lg) were primed (5 min at 70C and 5 min at 4C) with 1 lg of oligo dT primer (Invitrogen, Netherlands) in a final volume of 15 ll. A 10 ll aliquot of RNA was used as a template for RT (42C for 70 min) to cDNA using the cDNA cycle kit from Invitrogen. The reaction was then inactived at 94C for 5 min. Fisrt-strand cDNA synthesis was used for further PCR amplification. The HA sequence was also introduced by PCR in frame with the methionine of the Gaolf gene. The following oligonucleotic sense (S) and antisense (AS) primers were used: Gaolf /BglII HA S: 5#TTACGATATCAGATCTGCCACCATGTACCCCTACGACGTCCCTGACTCGCCATGGGGTGTTTG-GGCAAC3#; Gaolf /HindIII AS: 5#-CTATAAGCTTTCACAAGAGTTCGTACTGCTTGAG-3#. The cDNA encoding human Ga16 gene was obtained from the Human Leukemia, promyelocytic HL60 cDNA library (Clontech, France). The HA sequence was introduced by PCR in frame with the methionine at the 5# end of cDNA. The following oligonucleotidic sense (S) and antisense (AS) primers were used (Eurogentec, Belgium): Ga16/BglII HA S: 5#-TTACGATATCAGATCTGCCACCAT-GTACCCCT ACGACGTCCCT-GACTACGCCA-TGGCCCGCTCGC TGACC-3#; Ga16/HindIII AS: 5#-CTATA-AGCTTTCACAGCA-GGTTGATCTCGTCCAG-3# (restriction sites are underlined and HA sequence is double underlined). The intronless genes encoding OR 17-209 and OR 17-210 were amplified by PCR using the cosmid no. ICR F105cF06137 (GenBank HSU53583) as described previously (Matarazzo et al., 2002). The following oligonucleotidic S and AS primers (Eurogentec) were used: OR 17-209/NcoI S: 5#-TAAGAAGCTTGCCACCATGGAGGGGAAAAATCTG-3#; OR-209/KpnI AS: 5#-TAACGGTACCGCGGCCGCCTAAGGGGAATGAATTTTCCG-3#; OR 17-210/NcoI S: 5#-CAATAAGCTTCCATGGCTATGTAT-TTGTGTCTCAGCAAC-3#; OR-210/KpnI AS: 5#TAACGGTACCG-CGGCCGCTTAAGCCACTGATTTAGAGTG-3#. In all PCR experiments, 10 ng of cDNA, 10 mM Tris–HCl (pH 8.3), 50 mM KCl, 2.5 mM MgCl2, 0.001% gelatin, 0.2 mM dNTP, 1.5 U of high-fidelity Expand mix of polymerases (Roche Molecular Biochemicals) and 100 pmol of each primer were mixed according to the following schedule: 94C (60 s, 1 cycle), 94C (45 s), 58C (45 s), 72C (90 s, 35 cycles) and 72C (120 s, 1 cycle). PCR products were Two Human ORs Expressed in a Baculovirus Insect Cell System 197 then digested and subcloned into the transfer vectors p119 and pGmAc 217T respectively. Transfer vectors The unique BglII and HindIII cloning sites of the p119 transfer vector (Marchal et al., 2001), located downstream of the baculovirus p10 promoter, were used to insert either the HA-Gaolf cDNA or HA-Ga16 cDNA. Two plasmids containing the p10 promoter, namely p119-HA-Gaolf and p119-HA-Ga16, were thus obtained. An 108 pb oligonucleotide, corresponding to the EGT (ecdysteroid UDPS-glycosyltransferase) signal peptide (Riegel et al., 1994) from AcMNPV followed by the FLAG epitope tagging sequence and two unique restriction sites (NcoI and KpnI), was inserted into the BglII/SacI sites of the pGmAc217T transfer vector (Gaymard et al., 1996). The OR 17-209 or OR 17-210 sequences were then inserted into the NcoI and KpnI sites. Two plasmids containing the polyhedrin promoter were thus obtained: pGmAc217T-FLAG-OR 17-209 and pGmAc-FLAG-OR 17-210. was assessed by EcoRI digestion and Southern blot analysis. Finally, in order to obtain double-recombinant baculo viruses, AcSLP10GaolfPh or AcSLP10Ga16Ph DNA were cotransfected with pGmAc 217T-transfer vector into Sf9 cells and selected as obÿ virus phenotype. The final four doublerecombinant baculoviruses were designated as AcGaolf-209, AcGa16–209, AcGaolf-210 and AcGa16–210. Western blot analysis Lysates (TE buffer: 10 mM Tris–HCl, pH 8.0, 1 mM EDTA) of 36–48 h post-infected cells were electrophoresed on 12.5% SDS–polyacrylamide gel and transferred to nitrocellulose filter papers. After a blocking step (30 min in TS buffer, 5% dry milk, 0,05% Tween 20), blots were incubated overnight at 4C with an 1:50 dilution of anti-HA monoclonal antibody 3F10 (Roche) or 1:1000 dilution of anti-FLAG M2 monoclonal antibody (Sigma) and revealed with alkaline phosphataseconjugated goat anti-mouse IgG antibody (Promega) at 1:500 dilution. Peroxidase-conjugated antibody (Sigma) was used to probe the in-house anti-polyhedrin antibody. Recombinant baculoviruses Immunofluorescence and confocal microscopy Sf9 insect cells (ATCCCRL 1711), maintained at 28C in medium containing 5% serum, were used for virus propagation, transfection, plaque assays and limiting dilutions. Baculoviruses DNA from AcMNPV wild type and AcSLP10 (Blanc et al., 1993) were prepared from budding particles. Transfections were performed as previously described (Poul et al., 1995) with minor modifications. Sf9 cells were cotransfected with AcMNPV baculovirus and then with pGmAc217T (see Figure 1B). This recombination procedure permitted to obtain two single baculoviruses expressing either the FLAG-OR 17-209 or the FLAG-OR 17-210 under control of the polyhedrin promoter, which were plaque-purified as an occlusion-bodies negative (obÿ) virus phenotype and designated as AcMNPV209 and AcMNPV210 respectively. A three-step recombination procedure (see Figure 1C) was carried out to construct four double-recombinant baculoviruses (FLAG-OR 17-209 with either HA-Gaolf or HA-Ga16 and FLAG-OR 17-210 with either HA-Gaolf or HA-Ga16). The first step consisted of cotransfecting Sf9 cells with AcSLP10 baculovirus and p119 transfer vector. Recombinant baculoviruses expressing either HA-Gaolf or HA-Ga16, under the control of the p10 promoter, were plaque-purified as obÿ virus phenotype and designated as AcSLP10Gaolf and AcSLP10Ga16. In a second step, AcSLP10Gaolf or AcSLP10Ga16 DNA were cotransfected with the pGmAcI50 transfer vector (including the polyhedrin sequence and its promoter, as illustrated in Figure 1A) into Sf9 cells. Recombinant baculoviruses expressing either Ga protein and the polyhedrin were plaque-purified ob+ virus phenotype and designated as AcSLP10GaolfPh and AcSLP10Ga16Ph. Insertion of polyhedrin gene in the recombinated viral genome All fixation steps were made on ice to prevent receptor internalization. Post-infected cells (36 h) were fixed in 3% of PFA buffer for 20 min and washed with a 0.05 M NH4Cl solution for 10 min, then twice with PBS for 5 min. After a blocking step (30 min with 10% inactivated goat serum buffer), cells were incubated for 1 h with anti-FLAG (1:500 dilution) then with FITC-coupled monoclonal anti-mouse (Sigma) at 1:128 dilution. For HA-tagged G-proteins, a 1:20 dilution of the HA-rho conjugated rhodhamine (Roche) was used after a permeabilization step with 0.1% of Triton X-100. For the double-staining procedure, after anti-FLAG labelling, cells were fixed and blocked again before permeabilization. Coverslips were mounted on slides with Mowiol (Calbiochem), air-dried for 24 h prior to of fluorescent analysis with a Leica TCS 4D confocal microscope with an oil-X63 lens. OR cell surface expression An indirect ELISA protocol was applied to quantify the expression of FLAG epitope-tagged OR constructs. Briefly, after a 36, 48 or 72 h transfection period, Sf9 cells were fixed, blocked and incubated with anti-FLAG antibody (1:800 dilution). Plates were washed and incubated with a 1:500 dilution of alkaline phosphatase-conjugated goat antimouse IgG antibody (Promega) for 1h at room temperature. After three washings, the p-nitrophenyl phosphate substrate (2 mg/ml) diluted in ethanolamine (1 M, pH 9.8) was added and further incubated in the dark for up to 1 h. Absorbance was measured at 405 nm. Calibration was made using commercial preparation of bovine alkaline phospatase–FLAG (Sigma). 198 V. Matarazzo et al. A polyhedrin promoter (pph) 5’ EGT FLAG Gα subunit OR HA polyhedrin promoter polyhedrin P10 promoter (pP10) 3’ p119 pGmAc217T pGmAcI50 C B pph pph AcSLP10 baculovirus polyhedrin polyhedrin AcMNPV baculovirus pP10 p119 pP10 P10 pph AcSLP10Gα16 (Gαolf) baculovirus pGmAc217T HA Gα subunit pP10 pGmAcI50 pph EGT FLAG OR pph polyhedrin AcSLP10Gα16 (Gαolf) Ph baculovirus AcMNPV209 (210) baculovirus Gα subunit pP10 P10 pP10 pGmAc217T pph cell infection HA EGT FLAG OR AcGα16-209 (210) / AcGαolf-209 (210) baculoviruses Gα subunit HA pP10 cell infection OR 17-209 or OR 17-210 OR 17-209 or OR 17-210 FLAG FLAG Gαolf or Gα16 HA Figure 1 Schematic representation of obtention of recombinant baculoviruses. (A) Transfer vectors used for recombination with baculovirus, obtention of (B) monorecombinant baculovirus OR 17-209 or OR 17-210 and (C) double-recombinant baculovirus OR 17-209 or OR 17-210 with either Ga16 or Gaolf protein subtype. Measurement of intracellular calcium concentration 2+ Fura-2 fluorescence Ca imaging was used as previously described (Marcet et al., 2003) to measure [Ca2+]i in noninfected or 24 h post-infected cells. Briefly, Sf9 cells, cultured on glass coverslips and loaded (1 h at room temperature) with 4 lM fura-2-AM (Molecular Probes), were placed into a microperfusion chamber (200 ll bath volume) and gravity superfused with test solutions (1 ml/min). A new glass coverslip was used for each odorant mixture. Each test solution was applied in the syringe linked to the perfusion chamber with a manifold. Using a TILLvisION system (Germany), [Ca2+]i was calculated from the formula proposed by Grynkiewicz et al. (1985). Minimal and maximal experimental ratio values (Rmin and Rmax) were calculated for Sf9 cells. Rmax and Rmin are the values of R under saturating (5 lM A23187, a Ca2+-ionophore) and Ca2+-free conditions (4 mM EGTA combined with 5 lM A23187), respectively. The [Ca2+]i of individual cells was thus calculated every 5 s Two Human ORs Expressed in a Baculovirus Insect Cell System 199 and values were plotted versus time. Resting [Ca2+]i values of individual Sf9 cells oscillated between 80 and 100 nM. For comparison purposes, we determined the DCa2+ for each cell, defined as the value of the [Ca2+]i increase after ligand application minus the value of [Ca2+]i at the resting state. For each experiment, the most responsive cells (5–10 cells out of 20–30 in the camera field of view) were selected. Data were expressed as the mean ± SEM. Student’s t-test was used to determine statistical significance. Differences were considered significant if P < 0.05. Odorant screening All odorants were tested at a concentration of 1% (w/w). Stock solutions were stored at 4C and diluted (10ÿ6 M) in MBS medium (Mes-buffered saline) before use. Odotope screening was performed using successive sets of odorants. First a mixture of 100 odorant or Henkel 100 mix, as detailed by Wetzel et al. (1999), and a set of six odorants (camphor, limonene, isoamyl acetate, acetophenone, 2-heptanone and anisole), named the minimal set, were used. Next, the following sets of chemically defined groups were prepared: lactones (pentadecanolide, whisky lactone), ketones (acetophenone, camphor, b-ionone), esters (benzylacetate, 8-cineole, isobornylacetate, hexyl acetate, benzyl benzoate, isoamyle acetate, ethyl butyrate, 2-phenyl ethyl acetate), phenols (thymol, eugenol, guaiacol), alcohols (menthol, citronellol, geraniol, linalool, octanol, benzyl alcohol, cinnamyl alcohol, phenylethanol, 1-octanol) and aldehydes (citral, amyl cinnamaldehyde, cinnamaldehyde, piperonal, C7/C8/C11/C12/C13 aldehydes, octanal aldehyde, vanillin, para-anisalhedyde). When a set was positively identified, individual odorant solutions of isoamyle acetate (from the ester set) and acetophenone (from the ketone set) were further prepared. In all experiments, octopamine (Sigma) was used to stimulate an endogenous GPCR (Hu et al., 1994) to control the intactness of infected Sf9 cells. Thapsigargin (Sigma) was also used to assess genuine intracellular calcium release response upon odorant application. Results designated as AcMNPV209 or AcMNPV210 (Figure 1B). To construct double-recombinant viruses, AcSLP10 baculovirus was recombined with transfer vectors p119, pGmAcI50 and pGmAc217T sequentially (Figure 1C). Four doublerecombinant baculoviruses, AcGaolf-209, and AcGaolf-210, AcGa16-209 and AcGa16-210, were thereby designed. PCR product analysis of OR and Ga protein DNAs is represented in Figure 2A. Southern blot analysis of the modified AcSLP10 baculovirus DNA revealed that the polyhedrin gene has been re-integrated in its proper locus, as compared with the wild type baculovirus (Figure 2B), allowing proper selection of recombinant viruses and subsequent OR cloning. Western blot analysis of cell lysates showed that polyhedrin, Ga subunits and OR recombinant proteins were found at the expected size for each recombinated baculovirus (Figure 2C). Expression and localization of ORs and Ga proteins Cellular localization of OR and Ga proteins were assessed by immunofluorescence using anti-FLAG and anti-HA mAbs as presented in Figure 3. Confocal microscopy of Sf9 cells infected with monorecombinant viruses AcMNPV209 or AcMNPV210 showed a remarkably typical punctuated localization at the cell surface in absence of permeabilization (Figure 3A, left panels), while cells expressing only Gaolf or Ga16 subunits exhibited an intense cytoplasmic labelling after a required permeabilization step (Figure 3A, right panels). When the two labelling procedures were carried out sequentially on infected cells with any of the four doublerecombinant viruses, a significant portion of the Ga protein labelling was found to colocalized with OR staining (Figure 3B, right panel). No significant differences in OR staining were noticed whether OR proteins were expressed alone (Figure 3A, left panels) or together with a Ga subunit (Figure 3B, left panel). Using an in-house immunoassay with commercial anti-FLAG antibodies and a FLAG-protein calibrator, up to of 250 ng of recombinant tagged protein per 106 cells were estimated for each OR over a 96 h post-infection period. Both the intensity of cell surface labelling and the estimation of OR production indicated a satisfactory level of OR expression in the baculovirus/Sf9 cell system. Human olfactory receptor expression Baculovirus transfer vector pGmAc217T has been designed for cloning PCR-amplified OR 17-209 or OR 17-210 cDNAs. Each full-length OR coding sequence was included in a plasmid cassette containing the FLAG-tagged OR cloned downstream from an EGT signal sequence under the polyhedrin promoter (Figure 1A, left). Both Gaolf and Ga16 sequences were tagged with an HA sequence and cloned separately in transfer vector p119 under the P10 promoter (Figure 1A, middle). Transfer vector pGmAcI50 (Figure 1A, right) was used for double recombinating steps as described below. Recombination of transfer vector pGmAc217T with AcMNPV baculovirus enabled monorecombinant OR baculoviruses Functional characterization of OR 17-209 and OR 17-210 To assess the functional expression of OR 17-209 and OR 17210, we used fura-2 fluorescence Ca2+ imaging. Ratiometric fluorescence (340/380) was used to calculate [Ca2+]i. For each experiments, the resting [Ca2+]i in unstimulated cells was determined before odorant application (90 ± 10 nM, n = 100). Pharmacological screening was based on the measurement of an increase of [Ca2+]i in OR expressing cells upon application of various sets of odorants. For each cell response to a given odorant, the change in intracellular Ca2+ concentration (D[Ca2+]i) was calculated by subtracting the resting value of [Ca2+]i from the maximal value of [Ca2+]i induced by 200 V. Matarazzo et al. A 09 -2 OR MWM 17 10 -2 OR 17 MWM 16 Gα MWM olf Gα 1 Kb 1 Kb + + + - Gαolf 62 47.5 47.5 Gα olf Gα OR 62 16 ol ntr Kda Co lf αo G 6 ol ntr α1 G Co Kda MW M C + + olf + - OR 17210 -G OR α16 17210 -G α AcG α16 -2 09(21 0) Gα16 17209 OR 17209 -G α1 6 OR 17209 -G αolf OR 17210 AcSL P10G αo Polyhedrin lf AcSL P10G + - α16 AcMN PV B AcG αol f20 9(210 ) 1 Kb 45 Kda 32.5 32.5 25 25 Figure 2 Analysis of recombinant materials. (A) PCR products of human OR 17-210 and OR-17-209 obtained from cosmid DNA (left). RT-PCR product analysis of Ga16 (middle) and Gaolf (right) obtained respectively from human HL60 cDNA bank and a total cDNA brain preparation. (B) EcoRI enzymatic restriction and Southern blot mapping of the incorporated polyedrin gene in each recombinated baculovirus and in the wild type baculovirus ACMNPV. (C) Western blot analysis of polyhedrin protein expression (left), G-protein expression (middle) and OR expression (right). The control lane corresponds to Sf9 cells infected by wild type baculovirus. Polyhedrin, Ga16 and Gaolf proteins were detected similarly in Sf9 cells infected with any of the four double-recombinant baculovirus (AcGa16209, AcGa16-210, AcGaolf-209, AcGaolf-210). For simplification purpose only two lanes, Ga16 or Gaolf, are represented in the left and middle Western blot experiments. the odorant application. In all experiments, stimulation of an endogenous GPCR by octopamine has permitted to control the intactness of infected Sf9 cells (Figures 4–6). In a first step, Henkel 100 mixture (50 lM) was used to assess large-scale screening of OR 17-209 or OR 17-210 expressed in Sf9 cells. We observed that Henkel 100 elicited an increase in [Ca2+]i for both ORs (data not shown). Screening was then carried out by using selected chemical members of qualitative groups, consisting in a minimal set of six odorants (camphor, limonene, isoamyl acetate, acetophenone, 2heptanone, anisole). Application of the minimal set evoked a transient [Ca2+]i rise in cells expressing either OR 17-209 (D[Ca2+]i = 40 ± 5 nM, n = 4) or OR 17-210 (D[Ca2+]i = 50 ± 5 nM, n = 4), as illustrated in Figure 4A. To assess whether odorant application induced a genuine calcium release signal, experiments were performed with thapsigarginpretreated Sf9 cells. Preincubation with thapsigargin (5 lM, 20 min) prior to odorant stimulation inhibited the calcium response of Sf9 cells (Figure 4B). Also, when cells were incubated in free Ca2+ MBS medium, responses were still observed upon minimal set application (Figure 4B), supporting the hypothesis that the measured [Ca2+] increase originated from intracellular stores. The identification of the odotope family was then pursued by constructing from the composition of the Henkel 100 and the minimal set six distinct chemical families of purified Two Human ORs Expressed in a Baculovirus Insect Cell System 201 A B OR 209 OR OR 210 G-protein Gαolf Gα16 OR+G-protein OR 209/Gα16 OR 209/Gαolf OR 210/Gα16 OR 210/Gαolf Figure 3 Confocal microscopy images of Sf9 cells infected by recombinant baculoviruses. Cells were infected with either (A) monorecombinant or (B) doublerecombinant baculoviruses as indicated, prior to immunostaining procedure. Cells expressing OR 17-209 or OR 17-210 are visualized using a anti-FLAG mAb (green fluorescence) while expressed Ga16 or Gaolf proteins are visualized with the anti-HA-rhodamine-conjugated mAb (red fluorescence). For both ORs, 202 V. Matarazzo et al. A 200 octopamine 180 minimal set [Ca2+]i (nM) 160 140 120 OR 17-210 expressing cell OR 17-209 expressing cell control cell 100 80 60 0 2 6 4 8 10 Time (min) B 170 minimal set 160 150 [Ca2+]i (nM) 140 130 MBS buffer 120 110 100 Ca2+ free MBS buffer 90 preincubation with thapsigargin 80 0 0,5 1 1,5 2 2,5 Time (min) Figure 4 Calcium response of Sf9 cells after minimal set stimulation. (A) A typical recording of the Ca2+ response of a single control cell (uninfected or wild type baculovirus infected) after minimal set application (50 lM to 1 mM) is represented by the red line. For comparison purposes, the representative response of a cell infected with OR 17-209 recombinant baculovirus (green line) and one infected with OR 17-210 recombinant baculovirus (blue line) after minimal set application (50 lM) are superimposed. In all experiments, cell responsiveness was assessing by a second stimulation with octopamine (50 lM). (B) Comparison of OR expressing cell responses to minimal set application (50 lM). Cells were incubated in different conditions: standard buffer (MBS medium, green line), in the absence of extracellular Ca2+ (free Ca2+ MBS medium, blue line) and after depletion of intracellular calcium stores prior to odorant stimulation (20 min pre-treatment with 5 lM thapsigargin in MBS medium, red line). odorants sharing the same functional group (ketone, ester, lactone, alcohol, aldehyde and phenyl). Non-infected cells did not respond to any application of each of the six mixtures (Figure 5A). Tested sequentially, we found that esters induced an elevation of [Ca2+]i only in OR 17-209 expressing cells (D[Ca2+]i = 45 ± 5 nM, n = 10) whereas ketones provoked a Ca2+ response (D[Ca2+]i = 55 ± 5 nM, n = 10) only in OR 17-210 expressing cells (Figure 5B,C). Finally, single odorants from the sets positively identified were tested. Isoamyl acetate (from the ester set) and acetophenone (from the ketone set) induced a calcium response in OR 17-209 or OR 17-210 expressing cells respectively (Figure 5B,C, right). Application of 50 lM of each odorant induced an elevation of [Ca2+]i (D[Ca2+]i = 40 ± 5 nM, n = 4, for isoamyl acetate; D[Ca2+]i = 50 ± 5 nM, n = 4, for acetophenone). Application of 10 lM of odorant also induced a response in OR 17-209 or OR 17-210 expressing cells, but with a slight calcium increase for both isoamyl acetate (D[Ca2+]i = 7 ± 3 nM, n = 4) and acetophenone (D[Ca2+]i = 12 ± 5 nM, n = 4). Importantly, no response was observed when any of the odorant (single or in mixture) was applied at concentration ranging from micromolar to millimolar to control cells, i.e. non-infected cells or cells infected with wild type baculovirus (as illustrated in Figures 4A and 5A). Also, no odorant stimulation was observed when cells were infected with p119 baculovirus containing Gaolf or Ga16 protein (data not shown). These results indicate that both ORs are functionally expressed and could be efficiently activated by application of a restricted combination of odorants or individual odorous molecule. The calcium reponses are odorant specific since only esters (single or set) activated OR 17-209 expressing cells while only ketones activated OR 17-210 expressing cells. It is thus concluded that each OR specifically binds to chemically distinct odorants. Ongoing study with structurally related molecules will further help to determine the fine specificity of each OR protein for odorants. As shown in Figure 6, esters (A) and ketones (B) induced a significant increased Ca2+ responses (t-test; P < 0.05) in cells coexpressing the Ga16 subunit with OR 17-209 (D[Ca2+]i = 56 ± 3 nM, n = 8 for esters) or OR 17-210 (D[Ca2+]i = 72 ± 5 nM, n = 8 for ketones) respectively, compared with those observed in cells expressing OR alone (D[Ca2+]i = 46 ± 3 nM, n = 10 for esters; D[Ca2+]i = 55 ± 5 nM, n = 10 for ketones). The coexpression of the Ga16 subunit with OR 17-209 or OR 17-210 improved the Ca2+ response induced by esters or ketones by 22 ± 1.5 and 31 ± 3% respectively. Similarly, Ga16 coexpression induced also an improved calcium response of 20 ± 5% upon application of isoamyl acetate (Figure 6A) and 30 ± 2% upon application of acetophenone (Figure 6B). Calcium response to the minimal set (Figure 6A,B) was also improved by ;20% in cells coexpressing Ga16 with either OR. These findings therefore indicate that the coexpression and coupling of the Ga16 subunit with OR increase the odorant calcium response in Sf9 cells. Alternatively, in Sf9 cells coexpressing OR 17-209 or OR 17-210 together with the Gaolf subunit, no change in [Ca2+]i was observed when any ligand (50 lM to 1 mM) was applied to both ORs, while the octopamine (50 lM)-induced Ca2+ response was unaffected (Figure 6B,C, left). This observation suggests that both ORs may have been preferentially coupled to the recombinant human Gaolf protein than to immunostaining is visualized at the plasma membrane and shows typical punctuated localization at the cell surface when OR is expressed alone (A, left panels) or coexpressed with either Ga16 or Gaolf protein (B, left panels). Similarly, either Ga subtype staining shows intense cytoplasm labelling (A, right panels and B, middle panels). Superposition of immunostaining (yellow fluorescence) of cells infected with double-recombinant baculovirus reveals colocalization of OR and Ga protein at the plasma membrane (B, right panels). Two Human ORs Expressed in a Baculovirus Insect Cell System 203 A Control p to oc e in am 100 90 70 60 50 40 30 on ct al im la in m 20 es t se 10 e at et ac yl m ne oa is no he op et s ac ne to ke rs te es s de hy de al ls ho co al s ol en ph ∆[Ca2+]i (nM) 80 0 p to oc B OR 17-209 in am 100 e 90 t e at et ac (50µM) se 50 yl rs te es al im in 60 m oa is 70 m ∆[Ca2+]i (nM) 80 40 30 (10 µM) s s ne de to hy s ol s ol en h co ke de al al es 10 ph on ct la 20 0 to oc C OR 17-210 m pa 100 e in 90 al ne t se (50µM) no s he op et ne im ∆[Ca2+]i (nM) ac to in 60 m 70 ke 80 50 40 30 rs te es s de hy de al ls ho co al s ol en ph s e on ct 10 la 20 (10 µM) 0 Figure 5 Odorant-induced calcium responses of Sf9 cells expressing OR 17-209 or OR 17-210. (A) Control cells (uninfected or wild type baculovirus infected) or (B) cells expressing OR 17-209 or (C) cells expressing OR 17-210 were stimulated by different odorant sets (50 lM) or individual molecules (50 lM or 10 lM) as indicated. For each Sf9 cell, the value of D[Ca2+]i was calculated. It represents the value of the maximal [Ca2+]i increase obtained upon odorant or octopamine application (50 lM) minus the value of [Ca2+]i at the resting state. Data are means ± SEM for n independent experiments (n = 4–10; total number of cells = 30–120). endogeneous Ga subunits of Sf9 cells, inducing a shift of the Ca2+/IP3 signaling pathway to the cAMP pathway. Consequently no odorant-induced calcium response could be detected in these conditions. Taken together, the present results demonstrate that the baculovirus/insect cell system coupled to Ca2+ imaging represents a valuable method to express functional human ORs and investigate their odorant recognition properties. Ligand screening was successfully performed and has permitted to identify the respective odorant families of the human OR 17-209 and OR 17-210. Coexpression and coupling of OR with Ga16 subunit of the same human origin may also significantly improved the odorant-induced Ca2+ response in the Sf9 cell system. Discussion Difficulties in expressing OR in heterologous systems are caused by impaired intracellular traffic to the surface of the recombinant protein (McClintock et al., 1997; Lu et al., 204 V. Matarazzo et al. Figure 6 Odorant-induced calcium responses of Sf9 cells coexpressing OR together with Gaolf or Ga16 subunits. For OR 17-209 (A) or OR 17-210 (B), cells expressing OR alone (left) and cells coexpressing OR with Ga16 subunit (middle) or Gaolf (right) are represented. The value of D[Ca2+]i of each cell was determined upon odorant (50 lM) or octopamine application (50 lM). Data are means ± SEM for n independent experiments (n = 8–10; total number of cells = 60–100). For ester and ketone molecules (single or set), t-tests between OR and OR-Ga protein responses were calculated (ns, not significant; *P < 0.05). 2003). These GPCRs have specific requirements for maturation and targeting to the plasma membrane, as hypothesized by McClintock et al. (1997). Indeed, poor surface expression of OR in heterologous cells is caused by a combination of ER retention due to inefficient folding and ER export, aggregation and degradation (Lu et al., 2003). As a result, functional properties of ORs have remained poorly understood, especially in the human, whose genome encodes a restricted chemosensory repertoire compared with other mammal species. While the mechanisms responsible for inefficient translocation and surface expression of OR in heterologous cells are now in part elucidated, it still remains a technological bottleneck in the field of OR production and deorphanization. Alternatively, performing odorant screening on OR expressed in olfactory receptor neurons may suffer from several other disadvantages. The presence of non-neuronal surrogate cells, high level of background activity and lack of response reproducibility may prevent odorant identification (Reed et al., 1998; Wetzel et al., 1999). The insect cell system has been established as a useful intact cell setting for GPCR expression. Co- and post-translational processing events permit appropriate targeting of receptors and G subunits to the cell membrane. The variety of protein processing events performed by the Sf9 cell has provided valuable examples of reconstituted GPCRs, in particular for receptors of human origin (Parker et al., 1991; Butkerait et al., 1995; Barr et al., 1997). The fact that folding of helical secondary structure is better promoted at lower tempera- tures (Sf9 cells are cultivated at 27C) is also of relevant importance when dealing with seven transmembrane segment receptors. Nevertheless, since the successful expression of rodent OR5 (Raming et al., 1993), no other OR has been produced in this system so far. Mammalian cell lines have rather been used as expression system with variable success, as reported from our previous work (Matarazzo et al., 2002) and in the literature. Based on these considerations, we aimed at developing an alternate expression strategy and used the baculovirus/Sf9 cell system to express our human ORs. As described in this study, we have designed cassette baculovirus vectors containing tagged OR gene fused to an insect signal sequence. This specific leader sequence was selected to facilitate the addressing of the protein to the secretory pathway while the inherent properties of Sf9 cell allow appropriate folding of the recombinant product. Confocal microscopy associated to immunodetection experiments showed that human ORs are well expressed at the membrane of Sf9 cell together with a high level of receptor protein. Our genetically engineered baculovirus cassette is not restricted to human protein expression since OR sequences from any origin could also be expressed with the proper inserts. Ligand identification of engineered ORs requires the detection of an efficient coupling second messenger system that produces a measurable response upon application of odorant molecules. Some inherent disadvantages in using calcium imaging for odorant response measurement have been Two Human ORs Expressed in a Baculovirus Insect Cell System 205 reported, e.g. lack of reversibility, saturation and low transfection rate preventing calcium quantification (Wetzel et al., 1999). However, we did not encounter such limitations in our Sf9 system. Specific calcium responses were measured with cells expressing either OR, indicating that the necessary use of small epitope tag (FLAG) at the N-terminus of the expressed protein did not affect ligand binding or downstream signaling, as also demonstrated in the case of other mammalian ORs (Ivic et al., 2002). Indeed, ester or ketone application generated a rapid and quantitative [Ca2+]i increase, with fully reversible signals in OR 17-209 or OR 17-210 expressing cells, respectively. Earlier work on human OR 17-40 indicated that odorant recognition was not affected by the choice of a given OR expression system (Wetzel et al., 1999). Recently, however, it has been reported that odotope affinity may differ according to the system used (Levasseur et al., 2003). Such contradictory findings may be explained by a low level of OR expression, thus reinforcing the need for an efficient expression system for screening and identification of the odotope. Since hundreds of different odorant molecules exist, we have developed a strategy based on chemical dichotomy in order to functionally characterize each OR. First, Henkel 100, a mixture of 100 diverse odorants from commercial mixtures (Wetzel et al. 1999), was used to assess large-scale screening of OR 17-209 and OR 17-210 expressed in Sf9 cells. Screening was then carried out by using selected chemical members of qualitative groups. These odorants, defined as the minimal set, were originally determined on a neuro-olfactory basis (Duchamp-Viret et al., 1999) and were used previously for odorant stimulation of a mammal OR (Gaillard et al., 2002). From there, identifying the odotope family was pursued by constructing six distinct chemical families of purified odorants sharing the same functional group (ketone, ester, lactone, alcohol, aldehyde and phenyl). Induced calcium responses obtained with single members of a positive odorant set (isoamyl acetate or acetophenone from the ester and ketone sets respectively) represent the first step in odotope identification of both ORs. Current investigation is now aimed at refining ligand identification with the use of other structurally related odorous molecules and, if possible, defining the odorant specificity parameters for each expressed OR. Odorant response obtained with cells expressing OR alone or together with a Ga subtype confirmed that mammalian GPCRs can couple to different Ga proteins, including endogenous Ga proteins of Sf9 cells (Leopoldt et al., 1997). Coexpression with the Ga16 subunit improves the odorantinduced calcium response (+20 to +30%), suggesting an efficient OR coupling. Such enhanced response obtained when coexpressing a human OR together with a human Ga16 subunit might help to discriminate among related odorants. Also, our observation underlines the putative role of the Ga16 subtype protein in promoting intracellular calcium response to odorants (Krautwurst et al., 1998). Alternatively, this preferential coupling may also result from a shut-off of cellular genes as a result of baculovirus gene expression which concomitantly reduced endogenous G-protein number and favoured OR association with recombinant Ga16 protein. No [Ca2+]i increase has been observed upon ligand application in Sf9 cells coexpressing either OR together with the Gaolf protein. This observation might indicate that OR could interact with the Gaolf subunit (as suggested by confocal microscopy experiments) and thus activate the cAMP pathway but not the IP3/Ca2+ pathway. Unfortunately, no increase in cAMP level can be recorded because its endogenous level is highly elevated in infected Sf9 cells and does not change upon odorant application (Raming et al., 1993). It is therefore technically difficult to explore whether or not a shift from the IP3 to the cAMP pathway occurred as a result of a putative coupling of OR to Gaolf protein. Of interest, such a potential coupling would be relevant to the physiological situation because odorant binding to ORs in olfactory neurons induced Gaolf protein activation (Jones and Reed, 1989). Further work is needed to determine whether ORs can efficiently couple to both transduction pathways and maintain a functional cross-talk in our heterologous system, as previously reported for the mouse OR-EG expressed in HEK cells (Katada et al., 2003). However, we cannot rule out that the over-expression of Gaolf subunit may alternatively play a role of ÔscavengerÕ for endogenous Gb/c subunits. In this case, the lack of available Gb/c subunits may prevent OR signaling via an endogenous Ca2+ release-mediating Ga subunit. In conclusion, our strategy for OR expression in the Sf9 model has been validated. Two ORs from distinct genetic human subfamilies have been functionally expressed and their respective odorant family identified by screening of odorous sets based on calcium imaging. It is hoped that the enhanced responses to odorants observed through efficient homologous coupling of Ga16 subunit to OR will help further to discriminate quantitatively among competitive odorants. This in turn will permit the refining of odotopes identified by deconvolution of structural odorant librairies. As our cassette strategy is adaptable to different gene constructs, this system is convenient for expressing other ORs and assigning their respective odorants. More generally, our approach may not be limited to orphan OR and could be also useful for expression of various GPCRs. Acknowledgements We thank J.M. Botto and J.C. Guillemot for technical assistance. This work was supported by a grant (ANS ’odorant perception’) from the Institut National de la Recherche Agronomique and part of the work has been patented jointly by CNRS and INRA under the accession number 0209377. References Barr, A.J., Brass, L.F. and Manning, D.R. (1997) Reconstitution of receptors and GTP-binding regulatory proteins (G proteins) in Sf9 cells. A direct 206 V. Matarazzo et al. evaluation of selectivity in receptorG protein coupling. J. Biol. Chem., 272, 2223–2229. Beets, J. (1971) Olfactory response and molecular structure. Handbook Sens. Physiol., 4, 257–321. Ben-Arie, N., Lancet, D., Taylor, C., Khen, M., Walker, N., Ledbetter, D.H., Carrozzo, R., Patel, K., Sheer, D. and Lehrach, H. (1994) Olfactory receptor gene cluster on human chromosome 17: possible duplication of an ancestral receptor repertoire. Hum. Mol. Genet., 3, 229–235. Blanc, S., Cerutti, M., Usmany, M., Vlak, J.M. and Hull, R. (1993) Biological activity of cauliflower mosaic virus aphid transmission factor expressed in a heterologous system. Virology, 192, 643–650. Breer, H. (1994) Odor recognition and second messenger signaling in olfactory receptor neurons. Semin. Cell Biol., 5, 25–32. Butkerait, P., Zheng, Y., Hallak, H., Graham, T.E., Miller, H.A., Burris, K.D., Molinoff, P.B. and Manning, D.R. (1995) Expression of the human 5-hydroxytryptamine1A receptor in Sf9 cells. Reconstitution of a coupled phenotype by co-expression of mammalian G protein subunits. J. Biol. Chem., 270, 18691–18699. Chastrette, M., Thomas-Danguin, T. and Rallet, E. (1998) Modelling the human olfactory stimulus-response function. Chem. Senses, 23, 181–196. Crowe, M.L., Perry, B.N. and Connerton, I.F. (1996) Olfactory receptorencoding genes and pseudogenes are expressed in humans. Gene, 169, 247–249. Duchamp-Viret, P., Chaput, M.A. and Duchamp, A. (1999) Odor response properties of rat olfactory receptor neurons. Science, 284, 2171–2174. Gaillard, I., Rouquier, S., Pin, J.-P., Mollard, P., Richard, S., Barnabé, C., Demaille, J. and Giorgi, D. (2002) A single olfactory receptor specifically binds a set of odorant molecules. Eur. J. Neurosci., 15, 409–418. Gaymard, F., Cérutti M., Horeau, C., Lemaillet, G., Urbach, S., Ravallec, M., Devauchelle, G., Sentenac, H. and Thibaud, J.B. (1996) The channel from Arabidopsis thaliana baculovirus/insect cell system as an alternative to Xenopus oocytes. First characterization of the AKT1 K+. J. Biol. Chem., 271, 22863–22870. Gilad, Y., Man, O., Paabo, S. and Lancet, D. (2003) Human specific loss of olfactory receptor genes. Proc. Natl Acad. Sci. USA, 100, 3324–3327. Grynkiewicz, G., Poenie, M. and Tsien, R.Y. (1985) A new generation of Ca2+ indicators with greatly improved fluorescence properties. J. Biol. Chem., 25, 3440–3450. Krautwurst D., Yau, K.W. and Reed R. (1998) Identification of ligands for olfactory receptors by functional expression of a receptor library. Cell, 95, 917–926. Leopoldt, D., Harteneck, C. and Nurnberg, B. (1997) G-proteins endogenously expressed in Sf9 cells:interactions with mammalian histamine receptors. Naunyn Schmiedebergs Arch. Pharmacol., 356, 216– 224. Levasseur, G., Persuy, M.A., Grebert, D., Remy, J.J., Salesse, R. and Pajot-Augy, E. (2003) Ligand-specific dose-response of heterologously expressed olfactory receptors. Eur. J. Biochem., 270, 2905– 2912. Lu, M., Echeverri, F. and Moyer, B.D. (2003) Endoplasmic reticulum retention, degradation and aggregation of olfactory G-protein coupled receptors. Traffic, 6, 416–433. Malnic, B., Hirono, J., Sato, T. and Buck, L.B. (1999) Combinatorial receptor codes for odors. Cell 96, 713–723. Malnic, B., Godfrey, P.A. and Buck, L.B. (2004) The human olfactory receptor gene family. Proc. Natl Acad. Sci. USA, 101, 2584–2589. Marcet, B., Chappe, V., Delmas, P. Gola, M. and Verrier, B. (2003) Negative regulation of CFTR activity by extracellular ATP involves P2Y2 receptors in CFTR-expressing CHO cells. J. Membr. Biol., 194, 21–32. Marchal, I., Cérutti, M., Mir, A.M., Juliant, S., Devauchelle, G., Cacan, R. and Verbert, A. (2001) Expression of a membrane-bound form of Trypanosoma cruzei trans-sialidase in baculovirus-infected cells: a potential tool for sialylation of glycoproteins produced in the baculovirus–insect cells system. Glycobiology, 11, 593–603. Matarazzo, V., Zsürger, N., Guillemot, J.C., Clot-Faybesse, O., Botto, J.M., Dal Farra, D., Crowe, M., Demaille, J., Vincent, P., Mazella, J. and Ronin, C. (2002) Porcine odorant-binding protein selectively binds to a human olfactory receptor. Chem. Senses, 27, 691–701. McClintock, T.S., Landers, T.M., Gimelbrant, A.A., Fuller, L.Z., Jackson, B.A., Jayawickreme, C.K. and Lerner, M.R. (1997) Functional expression of olfactory–adrenergic receptor chimeras and intracellular retention of heterolously expressed olfactory receptors. Brain Res. Mol. Brain Res., 48, 270–278. Menashe, I., Man, O., Lancet, D. and Gilad, Y. (2003) Different noses different people. Nat. Genet., 34, 143–144. Hatt, H., Gisselmann, G. and Wetzel, C. (1999) Cloning, functional expression and characterization of a human olfactory receptor. Cell. Mol. Biol., 45, 285–291. Mombaerts, P. (2001) How smell develops. Nat. Neurosci. Suppl., 4, 1192– 1198. Hu, Y., Rajan, L. and Schilling, W.P. (1994) Ca2+ signaling in Sf9 insect cells and the functional expression of a rat brain M5 muscarinic receptor. Am. J. Physiol., 266, 1736–1743. Parker, E.M., Kameyama, K., Higashijima, T. and Ross, E.M. (1991) Reconstitutively active G protein-coupled receptors purified from baculovirus-infected insect cells. J. Biol. Chem., 266, 519–527. Ivic, L., Zhang, C., Zhang, X., Yoon, S.O. and Firenstein, S. (2002) Intracellular trafficking of a tagged and functional mammalian olfactory receptor. J. Neurobiol., 50, 56–58. Poul, M.A., Cerutti, M., Chaabihi, H., Devauchelle, G., Kaczorek, M. and Lefranc, M.P. (1995) Design of cassette baculovirus vectors for the production of therapeutic antibodies in insect cells. Immunotechnology, 1, 189–196. Jones, D.T. and Reed, R. (1989) Golf: an olfactory neuron specific-G protein involved in odorant signal transduction. Science, 244, 790–795. Kajiya, K., Inaki, K., Tanaka, M., Haga, T., Kataoka, H. and Touhara, K. (2001) Molecular bases of odor discrimination: reconstitution of olfactory receptors that recognize overlapping set of odorants. J. Neurosci., 16, 6018–6025. Katada, S., Nakagawa, T., Katoaka, H. and Touhara, K. (2003) Odorant response assays for a heterologously expressed olfactory receptor. Biochem. Biophys. Res. Commun., 305, 964–969. Raming, K., Krieger, J., Strotmann, J., Boekhoff, I., Kubick, S., Baumstark, C. and Breer, H. (1993) Cloning and expression of odorant receptors. Nature, 361, 353–356. Reed, R. (1998) Opening the window to odor space. Science, 279, 193. Riegel, C.I., Lanner-Herrera, C. and Slavicek, J.M. (1994) Identification and characterization of the ecdysteroid UDP-glucosyltransferase gene of the Lymantria dispar multinucleocapsid nuclear polyhedrosis virus. J. Gen. Virol., 75, 829–838. Two Human ORs Expressed in a Baculovirus Insect Cell System 207 Rouquier, S., Taviaux, S., Trask, B.J., Brand-Arpon, V., Van den Engh, G., Demaille, J. and Giorgi, D. (1998) Distribution of olfactory receptor genes in the human genome. Nat. Genet., 18, 243– 250. Sharon, D., Glusman, G., Pilpel, Y., Khen, M., Gruetzner, F., Haaf, T. and Lancet, D. (1999) Primate evolution of an olfactory receptor cluster: diversification by gene conversion and recent emergence of pseudogenes. Genomics, 61, 24–36. Vanti, W.B., Nguyen, T., Cheng, R., Lynch, K.R., George, S.R. and O’Dowd B.F. (2003) Novel human G-protein-coupled receptors. Biochem. Biophys. Res. Commun., 23, 67–71. Volz, A., Ehlers, A., Younger, R., Forbes, S., Trowsdale, J., Schnorr, D., Beck, S. and Ziegler, A. (2003) Complex transcription and splicing of odorant receptor genes. J. Biol. Chem., 278, 19691–19701. Wetzel, C.H., Oles, M., Wellerdieck, C., Kuckkowiak, M., Gisselmann, G. and Hatt, H. (1999) Specificity and sensitivity of a human olfactory receptor functionally expressed in human embryonic kidney 293 cells and Xenopus laevis oocytes. J. Neurosci., 19, 7426–7433. Zozulya, S., Echeverri, F. and Nguyen, T. (2001) The human olfactory repertoire. Genome Biol., 2, 18.1–18.12. Accepted December 22, 2004