* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Received June 19, 1964.
Survey
Document related concepts
Polyadenylation wikipedia , lookup
Metalloprotein wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Deoxyribozyme wikipedia , lookup
Peptide synthesis wikipedia , lookup
Citric acid cycle wikipedia , lookup
RNA silencing wikipedia , lookup
Genetic code wikipedia , lookup
Epitranscriptome wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Fatty acid synthesis wikipedia , lookup
Biochemistry wikipedia , lookup
15-Hydroxyeicosatetraenoic acid wikipedia , lookup
Butyric acid wikipedia , lookup
Specialized pro-resolving mediators wikipedia , lookup
Nucleic acid analogue wikipedia , lookup
Transcript
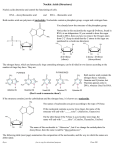
Comparison of Incorporation and Metabolism of RNA Pyrimidine Nucleotide Precursors in Leaf Tissues 1. 2 Cleon Ross Department of Botany and Plant Pathology, Colorado State University, Fort Collins carbamylase, and demonstrated several of its properties. Buchowicz and Reifer (7) found orotic acid to be converted to UMP, CMP, uridine, and uracil in wheat seedling leaves. Carbamyl aspartate also labelled these compounds and, in addition, orotic acid (30). This work suggests a pathway of nucleotide synthesis similar to that in other organisms. However, the latter workers reported that the conversion of orotic acid to UMP involved uridine as an intermediate product (9), and were unable to detect any OMP in their wheat leaves. Buchowicz (8) also reported that uracil could be incorporated directly into the nucleotides of RNA in wheat seedling homogenates without first labelling UMP and CMP. A direct binding of uracil by a polynucleotide acceptor was suggested to explain this surprising result. The present experiments were undertaken to provide more evidence as to some of the reactions involved in synthesis of uridylic and cytidylic acids in higher plants and to extend the results to plants other than wheat. Quantitative comparisons of the rate of incorporation of certain precursors into RNA were made and several metabolites were identified, including OMP. The pathway of synthesis of cytidylic and uridylic acids has been tentatively established in certain microbial and animal cells according to recent reviews (14, 35, 28). The sequence of reactions is probably -as follows: CO, + ATP + NH3 carbamyl -H^O aspartate diphosphate ~ carbamyl aspartate -2 H PRPP3 hydroorotic acid orotic acid > oro-tidine-5'-phosphate UMP + CO2. In yeast, UMP was further converted to UTP by kinases requiring ATP (23). Amination of uridine nucleotides to form cytidine nucleoticles involved participa-tion of glutamine in mammalian cells (18, 34) and NH3 in Escherichia coli (21). Neither uracil or uridine, cytosine or cytidine, are normal intermediates in synthesis of uridylic and cy-tidylic acids. The only pyrimidine involved is orotic acid, according to this pathway. In higher plants, uracil, uridine, and cytidine are incorporated into RNA (2, 8, 15), although incorporation of cytosine has apparently not been reported. Anabolism of uracil may occur either by addition of ribose from ribose-1-phosphate to produce uridine (11) or by -conversion directly to UMP upon reaction with PRPP (11, 13). Uridine was converted to UMP in the presence of ATP and a uridine kinase (11). The enzymes responsible for cytidine utilization have apparently not been extensively studied. Considerably less is known of the pathway of pyrimidine nucleotide synthesis in higher plants. In .a preliminary communication Kapoor and Waygood (19) reported in wheat embryos the presence of the -enzyme converting orotic acid to OMP, orotidine-5'-phosphate pyrophosphorylase, and of dihydroortic de-hydrogenase, which forms orotate from dihydroorotate. In a similar brief report, King and Wang (20) showed that C14 from both uracil and aspartic acid labelled pyrimidines of wheat leaves. Neumann an1ld Jones (25) recently studied aspartic acid trans1 Materials and Methods Plants Used. In most of the experiments bean plants, Phaseolus vulgaris var. Idaho 111, were used. Leaf discs (1.2 cm diameter) were cut with a cork borer from the leaflets of the first trifoliate leaves when these leaflets were 2.5 to 3.8 cm long. Discs were also cut from the most rapidly growing leaves (7-8 cm long) of the cocklebur, Xanthium pennsylvcanicum Wall., for use in some of the studies. Chemicals. Except for uridine-2-C14 and DLcarbamyl-C14-aspartic acid which were obtained from the New England Nuclear Corporation, all compounds were of the best grade supplied by the California Corporation for Biochemical Research. All radioactive compounds were chromatographed 2-dimensionally in the BAW and t-BKW solvents described further below. Significant impurities were not present in any except perhaps carbamyl aspartic acid. Here we found other compounds present after chromatography to the extent of 4.7 % of the total C14. Unlabelled carbamyl aspartic acid and 8-ureidopropionic acids were synthesized by alkaline treatment of dihydrouracil and dihydroorotate, respectively. 'Received June 19, 1964. GB-907 from the National Science Supported by grant Foundation. 3 Abbreviations: PRPP, 5-phosplhoribosyl-l-pyrophos-phate; UMP, UDP, UTP are 5'-mono, -di, and -triphosphates of uridine; CMP, CDP, CTP, cytidine nucleotides; uridylic and cytidylic acids refer to the products of alkaline hydrolysis of RNA; OMP, orotidine-5'-phosphate; 2 UDPG, uridine diphosphate glucose; PUP, 0-ureidopro- Pionic acid. 65 Downloaded from on June 18, 2017 - Published by www.plantphysiol.org Copyright © 1965 American Society of Plant Biologists. All rights reserved. 66 PLANT PHYSIOLOGY Methods of Supplying Labelled Compounds to the Tissues. Unless otherwise noted, 10 leaf discs (about 275 mg fresh weight for cocklebur and 225 mg for bean tissues) were added to 125 ml Erlenmeyer flasks .containing 5.0 ml 0.01 M phosphate buffer at pH 5.8. These flasks had a 1.5 cm diameter by 2.5 cm long well glued to the inside wall about 3 cm above the bottom. To this well 1.0 ml 3 % KOH was added to absorb CO2 released in respiration. Solutions of the radioactive compounds at equal total and specific radioactivities were added to the buffer solutions. Usually about 2.0 ,uc of each were used. After stoppering the flasks they were kept in darkness on a constant temperature water bathshaker, usually at 28° for 4 hours. Fractionation of Radioactivity in the Tissues. Analysis of C14 in respired CO, was performed as described previously (32). The leaf discs were washed with water to remove unabsorbed compounds, then killed in 50 ml hot 80 % ethanol and boiled until chlorophyll free. This solution was decanted and the discs were rinsed with enough 80 % ethanol so that the combined volume was 100 ml. Aliquots were plated on stainless steel planchets and C14 measured. The extract was combined in a separatory funnel with 2 volumes ethyl ether, 1 volume water, and 0.5 volume cyclohexane. After shaking, the lower aqueous phase was chlorophyll free. It was evaporated at room temperature with air or at 40° in a Rinco rotating evaporator under reduced pressure. The residue was redissolved in water for chromatography. The decolorized leaf discs were next homogenized in 20 ml of 50 % ethanol (previously adjusted to pH 4 with acetic acid) using an Omni mixer. After centrifugation and rinsing, aliquots were plated and C14 determined. The residues were further extracted twice with cold 0.2 N perchloric acid. The extracts were neutralized with KOH, chilled to precipitate KClO4, and aliquots plated to measure radioactivity. Corrections for self-absorption were required and made. The remainder of the 50 % ethanol and perchloric acid extracts were evaporated as above, redissolved in water, and combined with the concentrated 80 % alcohol extracts for chromatography. RNA was extracted with 0.3 M KOH and measured spectrophotometrically as described previously (31). Material insoluble in KOH and that precipitated from the KOH extracts by adjustment to pH 1 (including DNA) were analyzed for radioactivity in dry powder form. Approximate self-absorption corrections were applied. In a few cases these powders were converted to CO2 by a wet combustion technique described previously (36). After conversion to BaCO3, radioactivity was measured at infinite thickness as above. Radioactivity Measurements. These were made using a Nuclear Chicago Corporation gas flow geiger tube covered by a micromil window, and connected to an automatic sample changer. All samples except the BaCO3 were prepared on identical stainless steel planchets to insure uniform counting geometry. Self-absorption corrections were required for all except the ethanol soluble extracts. Counting efficiency was about 32 %. Chromatography, Electrophoresis, and Autoradiography. Whatman 3 MM papers, 46 X 57 cm, were used for chromatography of the combined alcohol and neutralized perchloric acid extracts. Initial separations were usually made with n-butanol-acetic acidlwater (BAW, 2: 1: 1 v/v) followed by t-butanolmethyl ethyl ketone-water-NH4OH (t-BKW, 4: 3: 2: 1 v/v) (16). For rechromatography, to aid in identification of the compounds thus resolved, the following additional solvents were employed: isopropanol-formic acid-water (IFW, 7: 1: 2), 95 % ethanol-i at ammonium acetate (7: 5: 3, pH 3.8 or 7.5, see ref. 26), n-propyl acetate-formic acid-water (11: 5: 3) and n-propanol-NH4OH-water (6: 3: 1, see ref. 5). and Pabst solvent III (1). Various 2-dimensional combinations of these were used as found appropriate. All were run descendingly. Paper electrophoresis was performed on 3.8 X 5/7 cm strips of paper, using 0.03 M citrate buffer at pH 3.5 or 5.2. A voltage gradient of about 20 v/cm was maintained. Strips were immersed in a large bath of carbon tetrachloride to avoid overheating. C14 on these papers was determined using a Vanguard Model 800 strip scanner. Autoradiograms of the 2-dimensional chromatograms were made with Kodak Blue Brand X-ray film. Exposure periods of 2 to 6 weeks were used. Spots thus detected were cut out, placed in planchets, and counted directly. Radioactivity in each metabolite was expressed as a per cent of the total detectable C14 present, uncorrected for absorption by the paper. For determination of the specific activities of RNA cytidylic and uridylic acids, chromatography of the nucleotides was performed in isopropanol-HCl-water (170: 41: 39 v/v). Nucleotides were detected with a Mineralight lamp, eluted with 0.1 N HCO and their concentrations determined in a Beckman DU spectrophotometer. C14 was measured in these same solutions. Identification of Metabolites. UDPG was identified as follows. Its ultraviolet absorption spectrum matched that of commercial UDPG or other uridine nucleotides. It cochromatographed with commercial UDPG in 5 solvent systems and the 2 moved together upon electrophoresis at both pH 3.5 and 5.2. In IFW and BAW partial hydrolysis occurred and radioactive compounds subsequently chromatographing with UDP, UMP, and less often, uridine were formed. By these methods the possibility that the spot contained appreciable amounts of UDP-acetylglucosamine was eliminated, but the presence of UDP-galactose or other closely related sugar derivative of UDP is possible. UMP was identified by its absorption spectrum in acid and alkaline solution and by cochromatography in several solvents. When 2-dimensional cochromatography was performed in conjunction with auto- Downloaded from on June 18, 2017 - Published by www.plantphysiol.org Copyright © 1965 American Society of Plant Biologists. All rights reserved. ROSS-PYRIAIIDINE NUCLEOTIDE SYNTHESIS radiography, the ultraviolet absorbing spot on the paper (detected with a Mineralight lamp) matched both in shape and position the exposed area on the film. UDP was found to cochromatograph with the known compound 1-dimensionally in 4 solvents and to have the same movement upon electrophoresis at pH 3.5. The latter method eliminated the chance that a significant amount of labelled CDP was present in the UDP area. UDP was also found to hydrolyze when chromatographed in acid solvents. Under these conditions some C14 was detected in a position corresponding to the movement of UMP. The following compounds were identified only by 2-dimensional chromatography, but in all cases matching of the exposed area on the film with the shape and position of the known compound on the paper left little doubt as to their identities: orotic acid, uracil, uridine, dihydrouracil, ,6-ureidopropionic acid, and carbamyl aspartic acid. The latter 3 compounds were located after cochromatography with the knowns by first opening the rings (if needed) by spraying with 0.5 N NaGH. After the papers dried they were sprayed with p-dimethylanminobenzaldehyde. Yellow colors resulted. The presence of labelled sucrose wvas confirmed by chromatography and autoradiography as above, and, in addition, it gave a yellow-green reaction characteristic for sucrose when sprayed and heated with aniline diphenylanmine (Sigma Chemical Company). Glucose gave a blue-grey color in this reaction. Two unknown compounds were believed to be oroti(line and orotidine-5'-phosphate because they contained C14 when the leaves absorbed orotic acilcarboxyl-C14. They were chromatographed 2-dimensionally with synthetic orotidine and OMP and then autoradiographs were made. Solvent systems were: A) BAW followed by t-BKW, B) IFW followved by Pabst III, C) BAW followed by the pH 3.8 ethanol-ammonium acetate solvent. In all cases, coincidence of the exposed area on the films with the spots on the paper detected with the Mineralight lamp appeared to confirm identities. Finally they were subjected to electrophoresis with commercial orotidine and OMP. The movement of C14 exactly matched the movement of the known compounds. Results and Discussion Coniparison of Incorporation of RNA Precursors. The distribution of C14 in various bean leaf tissue fractions after addition of several pyrimidine nucleotide precursors is shown in table I. The results sunmmarize 6 separate experiments in which 2 to 4 labelled precursors were studied at a time under the same experimental conditions. About 7.8 % of the added cytosine was absorbed. Absorption was less for aspartic acid, uridine, uracil, and orotic acid (5.74.2 % of that added), while only 1.9 % of the carbamyl aspartic acid was taken up. For all compounds, the greatest recovery of C14 was in the combined ethanol extracts. \With the exception of cyto- 67 sine all the compounds were extensively metabolized, and incorporation into RNA was easily measurable. Only traces of C14 were found in CO., and in RNA after feeding cytosine-2-C14, and amounts in RNA were too low to determine whether cytidylic or uridylic acid, or both, contained the label. This result supports those of others who found cytosine to be poorly converted to RNA of rat tissues (4). The present data also show that the poor anabolism is not due to a rapid breakdown to CO,. The label from DL-aspartic acid-3-C14 was incorporated into the RNA in relatively low amounts. Most of it was recovered in the aqueous ethanol extracts and in CO.,. Chromatograms of the nucleotides resulting from KOH hydrolysis of the RNA showed that the majority of the C14 was present in uridylic acid. Amounts in cytidylic acid were not detectable by our methods. However, DL-carbamyl-C'4-aspartic acid was a more efficient RNA precursor than aspartic acid and was far less rapidly catabolized to CO., Only the pyrimidine nucleotides of RNA were detectably labelled; the specific activity ratio of cytidylic acid/uridylic acid (C/U) averaged 0.63. Reifer et al. (30) found L-carbamyl aspartic-C14-acid to be incorporated into these nucleotides in excised wheat leaves and had previously concluded (29) that it was metabolized in plants along the same path as in animals. Orotic acid-2-C14 was very efficiently utilized in RNA synthesis and, like carbaryl aspartate, was no, rapidly catabolized to CO.}. Cytidylic and uridylic acids contained all the detectable C14 in RNA and the C/U ratio averaged 0.60. Conversion of uracil-2-C'4 to RNA was approximately half as efficient as orotic acid conversion. Both cytidylic and uridylic acids were labelled. Part of this relatively poor utilization of uracil for RNA was apparently due to competing catabolic reactions, since one-third of the C14 absorbed in this compound was recovered as CO.,. Uridine-2C14 was catabolized to a greater extent than either carbamyl aspartate or orotate, but only about half as rapidly as uracil, based on radioactivity of the CO.. Incorporation of uridine to RNA was very extensive, nearly equal to that of orotic acid. This suggests the adequate presence of a kinase to convert uridine to UMP. Other workers who studied both plants (15) anI animals (10) have found a rapid catabolism of uracil. Canellakis (10) showed that the relative effectiveness of uracil, orotic acid, and UMP depended on the amounts added. At high levels of each, uracil was almost as efficiently used as the others, while the percentage of uracil breakdown was much less than at low substrate levels. Thus, our results in table I might be expected to vary if different precursor concentrations were provided. However, we have found almost identical distribution of C14 among various tissue fractions when cytosine, uracil, and aspartic and orotic acids were supplied at one-fifth the amounts of table I, but have no data for larger differences in concentrations of added substrates. Downloaded from on June 18, 2017 - Published by www.plantphysiol.org Copyright © 1965 American Society of Plant Biologists. All rights reserved. 'co PLANT PHYSIOLOGY Table I. Distribution of C14 from Suspected RNA Precursors Among Tissue Fractions of Bean Leaf Discs Ten 1.2 cm diameter leaf discs (225 mg fresh tissue) were incubated in 5.0 ml 0.01 x potassium phosphate (pH 5.8) -for 4.0 hr at 27 to 280. Labelled substrates were added with a 100 MAl pipette after adjusting each to 4.5 mc/mmole. Ethanol extracts include the sum of cpm in hot 80 % and 50 % fractions. The residue is the KOH insoluble and the KOH soluble material precipitated at pH 1 (including DNA). Added cpm 1,490,000 1,530,000 1,470,000 1,510,000 Absorption 116,600 65,270 81,380 84,330 C14 recovery (cpm/flask) Ethanol extracts CO2 114,950 320 50,000 3,980 27,400 48,800 14,200 60,800 HC104 Cytosine Erotic acid Uracil Uridine DL-carbamyl 28,700 2,780 aspartate 34,390 1,490,000 55,380 DL-aspartate 23,100 80,940 1,427,000 * C/U is the specific activity of RNA cytidylic/uridylic acid. Nevertheless, these results are quite consistent with those expected if plants synthesize pyrimidine nucleotides by the pathway proposed in the Introduction. The observed greater conversion of orotic acid than either aspartic or carbamyl aspartic acids to -RNA is predicted by its position in the metabolic sequence, although comparisons are somewhat difficult because only the DL-forms of the latter 2 were available for studies. The reactions discussed also indicate that uridylic acid should be labelled prior to cytidylic acid upon addition of any previous precursor. Table I shows that in all cases where their labelling was measurable, the specific activity of uridylic acid was higher than that of cytidylic acid. In other unpublished experiments with cocklebur, we found that this C/U ratio increased with time after addition of orotic acid. In several instances the C/U -ratios were as high as 1.1 to 1.2 after 16 hours of exposure. To determine whether a similar situation occurs in bean leaves, a kinetic study with orotic-2-C14 acid was made. Figure 1 shows the time course for labelling of both RNA pyrimidine nucleotides over an 11-hour period. The increase in specific activity of uridylic acid appears to be linear, but the rate of cytidylic acid labelling became greater with time. As a result the C/U ratio increased from 0.43 after 3 hours to 1.0 after 11 hours. The data suggest that if the experiment had progressed longer, C/U ratios significantly greater than 1.0 would have resulted. Ratios as high as 2.0 were observed in rat mammary glands, where the transport of a high specific activity cytidine synthesized in another organ to the gland was suggested as a possible explanation for the unexpected labelling (38). Certain reasons to explain ratios greater than 1.0 could also be postulated for our plant tissues, but the exact mechanism is not known. Metabolic Products of RNA Precursors. If the proposed pathway indeed occurs in plants it should be possible to isolate some of the intermediate products. Two-dimensional paper chromatograms of the aqueous ethanol and neutralized perchloric acid extracts were prepared with this goal in mind. In various ex- soluble 870 2,410 1,100 1,620 Residue 90 380 275 500 440 420 70 820 RNA 380 8,500 3,800 7,210 2,400 1,220 C/U* 0.60 0.63 0.66 0.63 9 11 0 10 7 a TIME4 (NH)10 4 INcIJUTIO FIG. 1. Incorporation of orotic acid-2-C14 into RNA cytidylic acid (C) and uridylic acid (U) in bean leaf discs, and increase in the C/U ratio as a function of time. Initially, 2.0 ,uc C14 were added to 230 mg tissue in a final volume of 5.1 ml 0.01 M pH 5.8 potassium phosphate buffer. O I a 3 I periments both bean and cocklebur leaf tissues were studied, resulting in very similar patterns for metabolites of uracil, cytosine, and orotic acid. As yet, cocklebur studies with other precursors have not been made. Figure 2 shows drawings of the autoradiograms made after chromatography of extracts from bean tissues fed DL-carbamyl-C14-aspartic acid and 2-C'4-labelled uracil, uridine, and orotic acid. The spots are numbered to correspond to the identifications in table II. No metabolic products of cytosine2-C14 were observed in either cocklebur or bean, after film exposures comparable to those with the other metabolites. Only a very dense spot on the films caused by unchanged cytosine was present. This result substantiates the very poor metabolism of cytosine shown in table I. It apparently eliminates any chance that conversion of uracil or other metabolites labelling RNA cytidylic acid proceeded through cytosine as an intermediate. Thus, amination must occur at the Downloaded from on June 18, 2017 - Published by www.plantphysiol.org Copyright © 1965 American Society of Plant Biologists. All rights reserved. 69 ROSS-PYRI M IDI NE NUCLEOTIDE SYNTHESIS A. CARSAMYL-C" ASPARTATE B. URACIL-2-C"4 0 0 To 21 Id C) s 0 | UK-I I 35 UK-K 0 D. OROTIC ACID-2-C'1 C. URIDINE-2-C' 10r. 7 tn *6 6 UN-4 30 U5-4 09 QUz~K-s t.5> t 09 , 0 0 0.25 BAW-_ 0.50 Rt 0.75 0 0.25 UK-a 0.50 I0 0.75 FIG. 2. Chromatography of metabolites formed in bean leaf discs from RNA precursors. BAW was run first in the long direction of the paper. Compounds were detected by autoradiography and are numbered to correspond to those of table II. nucleoside or nucleotide level. It is interesting that the presence of an amino group on cytosine instead of a keto oxygen as on uracil would lead to such striking differences in utilization by these plants. Aspartic acid was converted primarily into other amino or Krebs cycle acids. Although the data are not shown, the distribution pattern was rather similar to that reported by Naylor et al. for wheat and barley leaves (24). Malic acid contained about half of the total radioactivity detectable on the chromatograms. Glutamic and citric acids were other important metabolites. Another ninhydrin positive compound was not identified, but was probably threonine. The important observation from these chromatographic studies of aspartic acid metabolites is that no nucleotides were detected. It is likely that they were formed but in such low quantities as to escape detection. Competing reactions involving the added aspartic acid were apparently much more extensive. Nucleotides were formed when carbamyl aspartic acid was added to the leaf discs. Spot 1 contained principally UDPG, although small amounts of UTP were also present, as evidenced by chromatography in other solvents and by paper electrophoresis at pH 3.5 and 5.2. By these methods it was found that any C14 present in CTP was so low as to escape detection. The area described by spot 2 was sometimes streaked between spots 1 and 3, perhaps due to partial hydrolysis in the acidic BAW solvent used in the first dimension. This area contained principally UDP with perhaps very small amounts of CDP present. It is possible that at least part of these compounds were formed by destruction of the corresponding triphosphates. Spot 3 contained UMP. Although CMP was not separated from UMP in these solvents, the 2 were easily resolved by use of IFW in 1 dimension. No CMP has ever been detected in the UMP area. Spot 4 contained unmetabolized carbamyl aspartate. The spot labelled UK-1 immediately above carbamyl aspartate is probably dihydroorotic acid, but since this compound was also present in the added solution of labelled carbamyl aspartate, or was formed from it during chromatography, it is not possible to say whether it is a true metabolite of the leaves. Reifer Table II. Per Cent Distribution of Radioactivity in Compounds Formed from Pyrimidine Nucleotide Precursors in Bean Leaf Discs Total radioactivity present in extracts is given in table I. After autoradiograms were made the spots thus detected were cut out, placed in planchets, and C14 measured. Precursor Compound 1. UDPG + UTP 2. UDP 3. UMP 4. Carbamyl aspartate Orotic acid 51.2 15.1 14.7 ... Uracil 34.5 7.6 14.2 Uridine 34.1 3.0 14.9 ... OL-carbamyl aspartate 17.9 3.0 9.5 68.0 ... 1.0 5.9 0.4. 0.6 2.9 0.6 7.4 30.1 45.4 ... 0.5 1.2 ... 1.3 6.2 UK-1 .. . .. ]6 0.1 UK-2* 42 0.2 ... UK-3 0.1 2.4 UK-4* ... G2 * We have not established that UK-2 is the same compound on chromatograms of both orotic acid and uracil metabolites. UK-4 may similarly not be identical in orotic acid and uridine experiments. Only the chromatographic behavior is known to be similar. 5. O-Ureidopropionate 6. Sucrose 7. Uracil 8. Dihydrouracil 9. Uridine 10. Orotic acid Downloaded from on June 18, 2017 - Published by www.plantphysiol.org Copyright © 1965 American Society of Plant Biologists. All rights reserved. 70 PLANT PHYSIOLOGY et al. (30) detected labelled UMP, CMP, uracil, uri- dine, and orotic acid after feeding carbamyl-C14aspartate to excised wheat leaves. We might also have found the latter 3 compounds had larger quantities of C14 been added, but their amounts relative to the others would certainly be very low. Leaf discs incubated with uracil, uridine, and orotic acid formed the same labelled nucleotides as those fed carbamyl aspartate (spots 1 to 3 of fig 2 A-D). The relative C14 content of each labelled area is given in table II. Since partial hydrolysis of the di- and triphosphates and of UDPG occurred during elution and/or subsequent concentration and rechromatography, it was not possible to report the exact radioactivity in UTP and UDPG, each of which was present in spot 1. These data are, however, consistent with results of others (38,17) who have found C14 in these nucleotides after adding 1 or more of the same labelled substrates. The low content of C14 in cytidine compounds relative to uridine nucleotides has been observed in various animal tissues (38,17). Bieleski and Laties (6) reported very small amounts of CTP and CDP present in potato tubers and were unable to detect CMP. Buchowicz and Reifer (9) found only about 10 % as much radioactivity in CMP as in UMP after adding labelled orotic acid to wheat leaves. Uracil was metabolized into several other compounds, including ,8-ureidopropionic acid, sucrose, and 2 others not identified. Unknown number 2 (UK-2) moved almost identically to carbamyl aspartate in the BAW, t-BKW solvents, but uracil-2-C14 would not be expected to label carbamyl aspartate. It has not been found on enough chromatograms to allow further studies as to its nature. UK-3 was ninhydrin positive and ran very near serine, yet it is not this amino acid. It was also found in the cocklebur extracts reported previously (32). Ureidopropionic acid is an intermediate in uracil catabolism in various organisms (35), and has been identified in cocklebur leaf discs (32), Longleaf pine tissue cultures (3), and rape seeds (15) following uracil feeding. The detection of labelled sucrose was surprising, and its mechanism of synthesis from uracil is not understood. Sucrose can be formed from urea (12). We have looked carefully for evidence of uridine formation from uracil, since uridine was an intermediate in UMP formation from uracil in the Ehrlich ascites tumor (35). Such uridine has not been detected on our chromatograms of bean or cocklebur extracts. This suggests, but certainly doesn't prove, that uridine does not participate in synthesis of UMP from uracil. This problem will be considered further below. On the other hand, uridine was converted to uracil in relatively large quantities (table II), perhaps by a uridine phosphorylase. Orotic acid labelled both uracil and, to a greater extent, uridine. Conversion of erotic acid to uracil and uridine was reported in pine embryos by Barnes and Naylor (3) and in wheat leaves by Buchowicz and Reifer (7). The latter authors' evidence indicated that either or both were intermediates in formation of UMP from orotic acid. They thus proposed that formation of orotidine-5'phosphate could be at least partially bypassed in wheat blades. They were unable to detect either this compound or orotidine in the extracts. Such a bypass is considered unlikely for RNA synthesis in bean and cocklebur leaves for 4 principal reasons. Instead, the labelling of uridine and uracil by orotic acid probably followed, not preceded, synthesis of UMP. First, we have found previously (32) that RNA synthesis in cocklebur leaves from orotic acid was blocked by addition of 6-azauracil. Azauracil is known to be converted metabolically to azauridine-5'-phosphate, which then inhibits orotidine-5'-phosphate (OMP) decarboxylation (27, 33). This implicated OMP in RNA formation in this plant. Carbon dioxide release from orotic acid was inhibited to about the same degree as was its incorporation into RNA, further showing that the principal sensitive reaction in RNA synthesis is at OMP decarboxylase (32). Secondly, decarboxylation of orotic acid appears to be dependent on PRPP. Experiment 2 of table III shows nearly a 6-fold increase in CO2 release from orotic acid-carboxyl-C14 in bean leaf homogenates when 10-4 M PRPP was added, and a 13-fold stimulation by 10-3 M PRPP. Formation of uracil or uridine directly from orotic acid as proposed by Buchowicz and Reifer (7) would likely not require PRPP, whereas if OMP is an intermediate, PRPP is essential (22). Thirdly, table IV shows that when azauracil or azauridine was added to inhibit OMP decarboxylation, accumulations of OMP and orotidine, as well as of orotic acid, were noted in bean and cocklebur leaf discs fed orotic acid-carboxyl-CI4. Without inhibitor only orotic acid and traces of orotidine (in beans) were found. (We previously observed only orotic acid accumulation when these inhibitors were added to cocklebur leaves (33), but less sensitive detection techniques were used). The majority of the C14 not converted to CO2 was accounted for in Table III. Stimulation of C1402 Release from Orotic Acid-Carboxyl-C'4 in Bean Leaf Homogenates by PRPP Eighteen g buds and young leaflets from 6-week-old bean plants were frozen, then homogenized 3 min in 45 ml cold 0.05 M Tris, pH 7.4, using a Servall Omni mixer at top speed. Homogenate was filtered through cheesecloth, and 4.0 ml then added to flasks with other additions to make 0.001 M MgCl,2 PRPP as indicated, and 1.0 Ac orotic acid (5.3 mc/mmole). Final volume was 5.55 ml. Flasks were incubated in darkness, with shaking, 3.8 hr at 270. C1402 collected in 3 %o KOH and measured by direct plating. All values are means of 3 flasks. Molarity PRPP None added 0.0001 0.001 C1402 recovery (cpm/flask) Expt. 1 Expt.2 150 101 450 573 ... 1300 Downloaded from on June 18, 2017 - Published by www.plantphysiol.org Copyright © 1965 American Society of Plant Biologists. All rights reserved. ROSS-PYRI MIDINE Table IV. Formiation of Orotidine and Orotidine A1onophosphate in Bean, and Cocklebutr Leaves front Orotic Acid-Carboxyl-C'4 To 290 mg fr wt of cocklebur leaf discs, 0.6 ,uc orotic acid-carboxyl-C14 was added. Tissue was incubated in 10 ml of 0.01 M potassium phosphate, pH 5.8, for 3 hr, then killed in hot 80 %o ethanol. Ethanol extracts were cochromatographed with the known compounds and autoradiographed. Bean leaf discs were incubated in 10 ml 0.05 M Tris, pH 7.4, containing 2.0 ac orotic acid. All values are means of duplicate flasks. Specific activity of orotic acid was 5.3 mc/mmole. C14 in compounds (cpm/flask) 0.003 M 0.003 Ai No inhibitor 6-azauracil 6-azauridin2 Cocklebur Orotic acid Orotidine Orotidine-5'phosphate Bean Orotic acid Orotidine Orotidine-5'phosphate 1,140 Not found 2,660 235 7,910 170 Not found Not found 730 ... 6,150 360 ... 32,900 3,970 Not found ... 625 orotic acid, perhaps because the equilibrium catalyzed by OMP pyrophosphorylase strongly favors orotic acid (22). The orotidine might have arisen from phosphatase action on OMP. Finally, the possibility of prior conversion of orotic acid to uracil before incorporation into RNA is eliminated by the nearly 2-fold greater ability of orotic acid than uracil to label the RNA. Uridine involvement cannot be disproved as convincingly by this latter argument alone, since it was almost as efficiently used for RNA synthesis as was orotic acid. Orotic acid was converted into 16-ureidopropionic acid (carbamyl 18-alanine) in both cocklebur andl bean leaf discs (table II). This compound is normally considered to be a catabolic product of uracil. The corresponding product of orotic acid catabolism is carbamyl aspartic acid (,8-ureidosuccinic acid), not identified in our orotic acid experiments, although it was apparently produced in pine embryos and tissue cultures from orotic acid (3). UK-2 could be carbamyl aspartate, based on its chromatographic movement, yet it appears to be the same compound as was found in the uracil experiments. UK-4, consistently found in orotic acid experiments and often in the uridine studies, had chromatographic properties close to those of uridine. It was not ninhydrin positive. The formation of 18-ureidopropionate suggests that catabolism of orotate may have occurred after its conversion to UMP and uracil. Evidence that this is an important pathway in cocklebur leaf tissues, at least, is that C1402 release from orotic acid-2-C1t is inhibited by 6-azauracil, known to lead to an inhibition of OMP decarboxylase (27, 32), thus blocking UMP formation. Similar reactions were proposed for orotate breakdown in rat livers (37). N 71 UCLEOTIDE SYNTHESIS Influence of Uridine on Uracil Incorporation into RNA. If uracil is initially converted to uridine prior to labelling uridine nucleotides, simultaneous addition of uridine with uracil-2-C14 should dilute the incorporation of C14 into RNA. Table V shows results of an experiment with bean leaf discs in which a concentration of uridine equal to that of added uracil-2C14 (5 X 10 6 Ai) decreased the specific activity of the RNA by 22 % while a 10-fold higher uridine concentration reduced it by 82 %. This effect was not due simply to an inhibition of uracil absorption by the uridine, although some decrease in total C14 recovered in the tissues was observed. The results suggest uridine was an intermediate in conversion of uracil into RNA and appear, therefore, contrary to the chromatographic data where no uridine was detected after addition of uracil. To clarify this problem, 5 X 10-4 AI uridine was added with labelled uracil to form a trapping pool for any uridine produced metabolically. Portions of the alcohol soluble tissue extracts were chromatographed 1-dimensionally in the neutral -ethanolammonium acetate solvent and 2-dimensionally in BAW and t-BKW. Autoradiography of the latter chromatograms did show radioactivity in the uridine. Its specific activity was 1,400 cpm/,umole. It represented only 0.57 % of the total C14 present on the chromatograms. It is assumed that this uridine arose directly from uracil although it is also possible that it was formed from dephosphorylation of UMP. Figure 3 shows the distribution of radioactivity in compounds along the 1-dimensional chromatograms. Where no uridine was added with the labelled 26 - UMP+UDPG 24 22 20 14 ~~~~~~~~~~~~~~~URACIL 0 .12 0 6 10 4~~~~~~~~~~~~~~~~~~~'u A~~~~~~~U DISTANCE FROM ORIGIN (cm) FIG. 3. Distribution of radioactivity in metabolites of uracil-2-C14 in the presence of 5 X 10-4 M uridine (dotted line) or in its absence (solid line). Chromatography was performed with the neutral ethanol-ammonium acetate solvent on paper strips. Strips were cut into 1 cm sections, eluted, and counted in the planchet counter. Values are means of 4 samples each. Downloaded from on June 18, 2017 - Published by www.plantphysiol.org Copyright © 1965 American Society of Plant Biologists. All rights reserved. 72 PLANT PHYSIOLOGY uracil, most of the C14 was in nucleotides (3 peaks closest to the origin) and little in residual uracil (last peak). The addition of uridine decreased incorporation of uracil into the nucleotides and caused its accumulation in the tissues. This result shows that the inhibition of incorporation of uracil into RNA by uridine (table V) did not occur simply because Table V. Inhibition of RNA Synthesis from Uracil-2-Cl4 by Uridine in Bean Leaf Discs Approximately 640,000 cpm uracil was added at a final concentration of 5 X 1-6 M to 10 ml 0.01 M potassium phosphate, pH 5.8. Ten leaf discs were incubated 4 hr in near darkness. Each value is a mean of 4 flasks. Uridine added None 5 X 10-6 M 5 X 10-5 M cpm absorbed 57,400 52,000 36,500 cpm in CO2 21,800 22,200 15,100 RNA C14 (cpm/mg) 10,200 7,940 1,840 uridine increased the total nucleotide pool, thus diluting the radioactivity in nucleotides derived from uracil. Instead, the effect is on conversion of uracil into the nucleotides. This result, combined with the above observation that uracil labelled uridine, is suggestive that uridine is a true intermediate in RNA synthesis from uracil. The necessary enzyme, uridine phosphorylase, has apparently not yet been demonstrated in plants. These reactions of uracil and uridine do not appear to be important in the normal synthetic pathway for RNA, which apparently involves only orotic acid as a free pyrimidine. Conclusions Both the experiments comparing the rate of incorporation of pyrimidine nucleotide precursors into RNA and the chromatographic studies suggest that much of the pathway proposed for other organisms is probably valid for higher plants as well. Specifically, RNA synthesis from aspartic, carbamyl aspartic, and orotic acids was consistent with their place in the proposed reaction sequence. The latter 2 were converted to the required uridine nucleotides, UMP, UDP, and UTP, in easily detectable quantities, although kinetic data for the order of their labelling was not obtained. In addition, UDPG was labelled by these 2 precursors. Cytidine nucleotides were probably formed but, if so, the quantities were very small compared to the uridine nucleotides. When orotic acid-carboxyl-C'4 was provided, the leaf discs formed small quantities of OMP and orotidine. The addition of 6-azauridine aided these findings because of a resulting inhibition of OMP decarboxylase, thus allowing some accumulation of these compounds. Even though OMP is an essential intermediate in RNA synthesis, it too is present only in small amounts, probably because of a rapid decarboxylation to UMP. This likely explains why Buchowicz and^ Reifer (7) were unable to find it in wheat leaves. As far as we are aware, this is the first demonstration of either OMP or orotidine in higher plants. The large stimulation of C140, loss from carboxyl-labelled orotic acid by PRPP is strongly suggestive of an OMP pyrophosphorylase enzyme synthesizing OMP from orotate, as was found by Lieberman et al. (22) in yeast. In the crude homogenate we used, however, other explanations for the result of PRPP addition are not ruled out. These leaf discs also converted uracil and uridine into RNA. Uracil catabolism was very rapid and this is undoubtedly one reason it was less efficiently used in RNA synthesis than was uridine. The data suggest uridine is an intermediate in uracil anabolism, as in the mammals studied (14). The failure of cytosine to significantly label the RNA was also consistent with animal results (4). In fact, cytosine was hardly metabolized by either bean or cocklebur leaves. Formation of 18-ureidopropionic acid from orotic acid as well as from uracil and uridine suggests the possibility that orotate catabolism normally occurs after conversion to the uridine nucleotide level. No detectable 8-ureidosuccinic acid (carbamyl-aspartic acid) was formed when orotate was fed. Previous evidence to support this interpretation was obtained in cocklebur leaf discs (32). It thus appears that orotate breakdown is not simply a reversal of its synthesis. Summary Leaf discs taken from beans, Phaseolus vulgaris var. Idaho 111, or cockleburs, Xanthiun pennsylvanicum Wall., converted several compounds into RNA uridylic and/or cytidylic acids. These, listed in the approximate order of efficiency as RNA precursors in the bean, were: orotic acid, uridine, carbamyl aspartic acid, uracil, and aspartic acid. Traces of C14 from cytosine also appeared in RNA but the extent was only about one-tenth that of uracil incorporation. All except aspartic acid and cytosine labelled soluble uridine nucleotides, including a compound with properties of uridine diphosphate glucose. Cytosine was very poorly metabolized. Formation of orotidine-5'-phosphate and orotidine from carboxyl-labelled orotic acid was demonstrated, especially where azauracil or azauridine was added to cause their accumulation. This result, combined with a large stimulation of C140, loss from orotic acid-carboxyl-C14 by 5phosphoribosylpyrophosphate, indicates a normal function for orotidine-5'-phosphate in plants. It was concluded that the results support much of the proposed pathway of pyrimidine nucleotide synthesis described for other organisms. Some of the results indicate orotic acid is principally catabolized after its conversion to uridine nucleotides, then by the normal pathway for uracil breakdown through ,8-ureidopropionic acid. Acknowledgments The technical assistance of Janet Thomas, Joseph Miller, and John Wolcott is appreciated. Robert Starr kindly performed the tissue combustion analyses. Downloaded from on June 18, 2017 - Published by www.plantphysiol.org Copyright © 1965 American Society of Plant Biologists. All rights reserved. ROSS-PYRIMIDINE NUCLEOTIDE SYNTHESIS Literature Cited 1. ANONYMOUS. 1956. Ultraviolet absorption spectra of 5'-ribonucleotides. Pabst Laboratories Circular OR-10. Pabst Brewing Company, Milwaukee. 2. BANDURSKI, R. AND S. MAHESHWARI. 1962. Nucleotide incorporation into nucleic acid by tobacco leaf homogenates. Plant Physiol. 37: 556-60. 3. BARNES, R. AND A. NAYLOR. 1962. Formation of 13-alanine by pine tissues supplied with intermediates in uracil and erotic acid metabolism. Plant Physiol. 37: 171-75. 4. BENDICH, A., H. GETLER, AND G. BROWN. 1949. A synthesis of isotopic cytosine and a study of its metabolism in the rat. J. Biol. Chem. 177: 565-70. 5. BIELESKI, R. AND R. YOUNG. 1963. Extraction and separation of phosphate esters from plant tissues. Anal. Biochem. 6: 54-68. 6. BIELESKI, R. AND G. LATIES. 1963. Turnover rates of phosphate esters in fresh and aged slices of potato tuber tissue. Plant Physiol. 38: 586-94. 7. BUCHOWICZ, J. AND I. REIFER. 1961. The conversion of erotic acid to pyrimidine derivatives in plant material. Acta Biochim. Polon. 8: 25-34. 8. BUCHOWICZ, J. 1963. Incorporation of [2-14C] uracil into polynucleotides in homogenates of wheat seedlings. Acta Biochim. Polon. 10: 301-07. 9. BUCHOWICZ, J. AND I. REIFER. 1962. The synthesis of pyrimidine nucleotide derivatives in plant material using [6-14C] erotic acid. Acta Biochim. Polon. 9: 63-70. 10. CANELLAKIS, E. 1957. Pyrimidine metabolism. III. The interaction of the catabolic and anabolic pathways of uracil metabolism. J. Biol. Chem. 227: 701-09. 11. CANELLAKIS, E. 1957. Pyrimidine metabolism. II. Enzymatic pathways of uracil anabolism. J. Biol. Chem. 227: 329-38. 12. CLoR, M., A. CRAFTS, AND S. YAMAGUCHI. 1963. Effects of high humidity on translocation of foliarapplied labelled compounds in plants. II. Translocation from starved leaves. Plant Physiol. 38: 501-07. 13. CRAWFORD, A., A. KORNBERG, AND E. SIMMS. 1957. Conversion of uracil and orotate to uridine-5'-phosphate by enzymes in Lactobacilli. J. Biol. Chem. 226: 1093-1101. 14. CROSBIF, G. 1960. Biosynthesis of pyrimidine nucleotides. In: The Nucleic Acids, Vol III. p 323-48. E. Chargaff and J. Davidson, eds. Academic Press. New York and London. 15. EVANS, W. AND B. AXELROD. 1961. Pyrimidine metabolism in germinating seedlings. Plant Physiol. 36: 9-13. 16. FINK, K., R. CLINE, R. HENDERSON, AND R. FINK. 1956. Metabolism of thymine (methyl-C'4 or -2-C14) by rat liver in vitro. J. Biol. Chem. 221: 425-33. 17. HURLBERT, R. AND V. POTTER. 1954. Nucleotide metabolism. I. The conversion of erotic acid-6C14 to uridine nucleotides. J. Biol. Chem. 209: 1-21. 18. KAMMEN, H. AND R. HURLBERT. 1958. Amination of uridine nucleotides to cytidine nucleotides by mammalian enzymes: Role of glutamine and guanosine nucleotides. Biochim. Biophys. Acta 30: 195-96. 19. KAPOOR, M. AND E. WAYGOOD. 1961. Enzymes of erotic acid metabolism in wheat embryos. Plant Physiol. suppl. 36: v. 73 20. KING, J. AND D. WANG. 1961. Biosynthesis of pyrimidines and their derivatives in the wheat plant. Plant Physiol. suppl. 36: v. 21. LIEBERMAN, I. 1956. Enzymatic amination of uridine triphosphate to cytidine triphosphate. J. Biol. Chem. 222: 765-74. 22. LIEBERMAN, I., A. KORNBERG, AND E. SimMS. 1955. Enzymatic synthesis of pyrimidine nucleotides orotidine-5'-phosphate and uridine-5'-phosphate. J. Biol. Chem. 215: 403-15. 23. LIEBERMAN, I., A. KORNBERG, AND E. SIMMS. 1955. Enzymatic synthesis of nucleoside diphosphates and triphosphates. J. Biol. Chem. 215: 429-39. 24. NAYLOR, A., R. RABSON, AND N. E. TOLBERT. 1958. Aspartic-C'4 acid metabolism in leaves, roots, and stems. Physiol. Plantarum 11: 53747. 25. NEUMANN, J. AND MARY E. JONES. 1962. Aspartic transcarbamylase from lettuce seedlings: Case of end product inhibition. Nature 195: 709-10. 26. PALADINI, A. AND L. LELOIR. 1952. Studies on uridine diphosphate glucose. Biochem. J. 51: 426-30. 27. PASTERNAK, C. AND R. HANDSCHUMACHER. 1959. The biochemical activity of 6-azauridine: interference with pyrimidine metabolism in transplantable mouse tumors. J. Biol. Chem. 234: 2992-97. 28. REICHARD, P. 1959. The enzymatic synthesis of pyrimidines. Advan. Enzymol. 21: 263-94. 29. REIFER, I., J. BUCHOWICZ, AND K. TOEZKO. 1960. The synthesis of the pyrimidine ring from L-carbamyl aspartic acid in excised blades of wheat seedlings. Acta Biochim. Polon. 7: 29-38. 30. REIFER, I., J. BUCIIOWICZ, AND K. TOEZKO. 1961. Metabolism of L-carbamyl aspartic-C14 to pyrimidine derivatives in excised wheat leaves. Acta Biochim. Polon. 8: 377. 31. Ross, C. 1962. Nucleotide composition of RNA from vegetative and flowering cocklebur shoot tips. Biochim. Biophys. Acta 55: 387-88. 32. Ross, C. 1964. Influence of 6-azauracil on pyrimidine metabolism of cocklebLr leaf discs. Biochim. Biophys. Acta 87: 564-73. 33. Ross, C. 1964. Metabolism of 6-azauracil and its incorporation into RNA in the cocklebur. Phytochem. 3: 603-07. 34. SALZMAN, N., H. EAGLE, AND E. SEBRING. 1958. The utilization of glutamine, glutamic acid, and ammonia for the biosynthesis of nucleic acid bases in mammalian cell cultures. J. Biol. Chem. 230: 1001-12. 35. SCHULMAN, M. 1961. Purines and pyrimidines. In: Metabolic Pathways, Vol. II. p 389-457. D. Greenberg, ed. Academic Press. New York and London. 36. STARR, R. AND C. Ross. 1964. A method for the determination of carbon in plant tissues. Anal. Biochem. 9: 243-46. 37. VON EULER, L., R. RUBIN, AND R. HANDSCHUMACHER. 1963. Fatty livers induced by orotic acid. II. Changes in nucleotide metabolism. J. Biol. Chem. 238: 2464-69. 38. WANG, D. AND A. GREENBAUM. 1962. The incorporation of [2-14C] glycine and [6-14C] orotic acid into the acid soluble ribonucleotides and ribonucleic acids of the rat mammary gland. Biochem. J. 83: 626-32. Downloaded from on June 18, 2017 - Published by www.plantphysiol.org Copyright © 1965 American Society of Plant Biologists. All rights reserved.