* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download • In the cell, nutrients and oxygen, have different electron affinities.
Survey
Document related concepts
Fatty acid metabolism wikipedia , lookup
Metalloprotein wikipedia , lookup
NADH:ubiquinone oxidoreductase (H+-translocating) wikipedia , lookup
Lactate dehydrogenase wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Phosphorylation wikipedia , lookup
Photosynthesis wikipedia , lookup
Mitochondrion wikipedia , lookup
Microbial metabolism wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Electron transport chain wikipedia , lookup
Biochemistry wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Light-dependent reactions wikipedia , lookup
Citric acid cycle wikipedia , lookup
Transcript
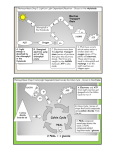
• The flow of electrons is frequently coupled to accomplishment of work. – We are most familiar with work accomplished by electric motor. – Other energy transformations are also possible, for example electron flow to light energy. • In a battery operated electric motor, the battery contains two chemicals that differ in affinity for electrons. – The electrons flow spontaneously through an electron carrier called a wire to the chemical with higher electron affinity. – Electron movement is driven by an force protional to the difference in electron affinity, measured in “volts” – When coupled to a "transducer or motor", the flow of electrons is used to accomplished mechanical work. – A transducer transforms one form of energy into another... in this case potential electric energy of the battery to mechanical energy + heat energy. • In the cell, nutrients and oxygen, have different electron affinities. – Electrons flow spontaneously towards the chemical with higher electron affinity (oxygen) through a series of electron carrier molecules rather than a wire. • The flow is driven by a force proportional to difference in electron affinity. • The flow of electrons is used by organic molecules that are energy transducers. – Enzymes (ATP Synthases) – Transport proteins (ATPase Pumps) – Motor Proteins (myosin ATPase) Energy Stored Within ATP • Mechanical work • Transport work • Chemical work Production and Use of ATP requires different enzymes • ATP production requires ATP synthases • ATP hydrolysis reactions are coupled to phosphorylation reactions by kinases or ATPases. Phosphate group from ATP is transferred to some other molecule, energizing that molecule by altering its electron configuration. Phosphorylation Via ATP synthase enzymes Hydrolysis via ATPases or Kinases ATP enz Energy from catabolism of nutrients like CHO enz Energy for cellular work (energy- consuming processes) ADP + P protein How Does ATP power cellular work? • ATP hydrolysis reactions are coupled to phosphorylation reactions: • Phosphate group from ATP is transferred to some other molecule, “energizing” that molecule. • Presence or absence of Phosphate group alters shape and activity of protein • In the mitochondria, multiple membrane bound molecules allow spontaneous flow of electrons from different nutrients to oxygen. • They couple the electron flow to production of a proton concentration difference across the inner mitochondrial membrane. • The proton concentration difference created across the membrane has potential energy. – This potenial energy is used to do chemical work: • Synthsis of ATP from ADP + Pi by an enzyme ATP SYNTHASE. – ATP synthase is a transducer – It links the spontaneous flow of protons from high to low concentration gradient across the membrane to synthesis of ATP from ADP + Pi. Fig. 2-12, p. 31 ATP Production • Sequence of steps involved in generation of ATP from CARBOHYDRATE within the cell 1. 2. 3. Glycolysis Citric acid cycle (Krebs Cycle) Electron transport chain In AIR: C6H12O6 + 6 O2 6 CO2 + 6H2O + 686 kcals HEAT In CELL: C6H12O6 + 6 O2 6 CO2 + 6H2O + HEAT + 32 ATP ALL molecules are in moles: 6 O2 means 6 moles of O2; one mole of O2 has a volume of 22.4 liters 32 ATP means 32 moles of ATP; one mole of ATP = 507 grams In a person 134.4 liters of O2 are used to produce 16224 grams of ATP from the potential energy locked in the covalent bonds of 180 grams of glucose Each liter of O2 consumed is equivalent to production of 120 grams of ATP The typical human ATP and ADP content is < 100 grams Each gram of ADP has to be recycled times 400‐500 times per day 1 gram of ATP has 1,187,968,441,814,600,000,000 molecules of ATP (1/507 gram/mole ATP x 6.023 x 1023 molecules/mole) C6H12O6 6 O2 Uncontrolled oxidation of food outside the body (burning) Explosive release of energy as heat and carbon dioxide C6H12O6 Controlled oxidation food inside the body accomplished by the small steps of CHO Metabolic pathways Energy harnessed as ATP, the common energy currency for the body Partly used to maintain body temperature MODIFIED Fig. 2-14, p. 32 6 O2 Energy released as heat + CO2 Heat + CO2 to the environment Cytosol Glycolysis Glucose (GLYCOGEN) 2 ATP Pyruvate Mitochondrial inner membrane Mitochondrial matrix Pyruvate to acetate Acetyl-CoA Citric acid cycle 2 ATP Electrons carried by NADH and FADH2 Oxidative phosphorylation (electron transport system and chemiosmosis) 28 ATP Fig. 2-9, p. 29 Carbohydrate Catabolism Glycolysis in Non‐aerobic as opposed to anerobic • Non‐aerobic: no oxygen required • Anaerobic: no oxygen available Anaerobic conditions cannot be demonstrated in living mammals Fig. 2-15, p. 33 Glycolysis produces 4 ATP and 2 Lactate from 1 glucose molecule • OLD VIEWPOINT – occurs in cytoplasm – requires 2 NAD, produces 2 NADH2 and 2 Pyruvate under “aerobic” conditions – requires 2 NAD and produces 2 lactate under anaerobic conditions • Cellular handling of NADH2 formed in glycolysis – Oxidative phosphorylation in mitochondria requires O2 to proceed, NADH2 NAD (aerobic = oxygen required) – In cytoplasm 2 Pyruvate + 2 NADH2 2Lactate + 2 NAD (non‐aerobic = no oxygen required) – What happens to lactate ? Old Viewpoint from Pre‐1990’s: accumulates and causes fatigue New Viewpoint from 2000’s • Lactate is shuttled to mitochondria of cell producing and oxidized either as lactate or pyruvate (intracellular shuttle is controversial) • Lactate is shuttled to mitochondria of some other cell and oxidized as either lactate or pyruvate (intercellular shuttle is widely accepted) Intracellular shuttle? Intercellular shuttle Glycolysis occurs in cells regardless of O2 content; O2 is never required for glycolysis; fate is lactate is dependent on what cell produces it and how fast it is produced Blood transport to Cytoplasm O2 NEVER REQUIRED Glycolysis 2 Pyruvate 2 Lactate Glucose 2 Intracellular Lactate shuttle Mitochondria Mitochondrion Glycolysis Glucose 2 Pyruvate 2 O22 required Citric acid cycle/Oxidative phosphorylation Lactate using cells such as heart and slow twitch skeletal Muscle (intercellular) Mitochondrial membranes 30 ATP + CO2+H2O Cytosol Lactate is the natural end product of glycolysis, is not a waste product, and is the link between glycolytic and oxidative metabolism Fig. 2-15, p. 33 Lactic Acid Dehydrogenase Pyruvic Acid Dehydrogenase Complex