* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Physics 228 Today: April 4, 2013 Do we fully
Angular momentum wikipedia , lookup
Quantum vacuum thruster wikipedia , lookup
History of subatomic physics wikipedia , lookup
Elementary particle wikipedia , lookup
Introduction to gauge theory wikipedia , lookup
Magnetic monopole wikipedia , lookup
Electromagnet wikipedia , lookup
Electromagnetism wikipedia , lookup
Quantum electrodynamics wikipedia , lookup
Superconductivity wikipedia , lookup
Nuclear physics wikipedia , lookup
Old quantum theory wikipedia , lookup
Aharonov–Bohm effect wikipedia , lookup
Accretion disk wikipedia , lookup
Spin (physics) wikipedia , lookup
Neutron magnetic moment wikipedia , lookup
Condensed matter physics wikipedia , lookup
Relativistic quantum mechanics wikipedia , lookup
Photon polarization wikipedia , lookup
Introduction to quantum mechanics wikipedia , lookup
Theoretical and experimental justification for the Schrödinger equation wikipedia , lookup
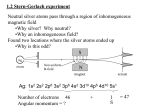
Physics 228 Today: April 4, 2013 Do we fully understand the hydrogen atom? No. Ch 41: 4-7: Zeeman effect, electron (and proton) spin, manyelectron atoms, ... Notes for next week: regular classes, except Thursday April 11: no lecture, exam review session (optional). Exam 10:00 - 11:20 PM in Hill 114 and ARC103. Website: Sakai 01:750:228 or www.physics.rutgers.edu/ugrad/228 Thursday, April 4, 2013 Hydrogen Atom Review: 3 Quantum Numbers Principle QN n = 1, 2, ...: states of same n are degenerate. Angular momentum QN l: L = [l(l+1)]½ħ, l = 0, ... n-1 magnetic" or "z-component" QN ml = 0, 1, ... l Shell K L " M " " N etc... Thursday, April 4, 2013 n 1 2 2 3 3 3 4 l 0 0 1 0 1 2 0 ml 0 0 0, ±1 0 0, ±1 0, ±1, ±2 0 notation 1s 2s 2p 3s 3p 3d 4s semiclassical picture What happens when you put the Hydrogen atom in a magnetic field? Here is the classical E&M physics analysis: If the hydrogen atom has a magnetic moment, then there is an interaction energy U = -μ.B. And a torque τ = μxB and precession. From our study of E&M, if the electron moves around the proton, there is a current, and thus a magnetic moment: μ = IA. Classically, if we have an orbit of radius r and speed v, then the period of the orbit is T = 2πr/v, and the current, the charge per unit time passing some point, is I = e/T = ev/2πr. Thus, the magnitude of the magnetic moment is μ = (ev/2πr)(πr2) = evr/2. Remembering that the angular momentum is L = |rxp| = mvr, we obtain μ = (e/2m)L. The ratio μ/L is called the gyromagnetic ratio (g). Thursday, April 4, 2013 The Bohr atom in a magnetic field The Bohr atom has circular orbits with the constraint that 2πrn = nλn. This leads to pn = h/λn = nh/2πrn = nħ/rn and Ln = mvnrn = pnrn = nħ. Thus μn = (-e/2m) Ln = (-e/2m) nħ. So Un = -μn.B = (eB/2m) nħ. The energy shift of the nth level is proportional to n. But this is not what is found. The spectrum is more complicated, because in hydrogen one has multiple L values for fixed n (except n=1). Thursday, April 4, 2013 The Schrödinger Eq. atom in a magnetic field How do we make the connection between the S.E. atom and the magnetic moment? It is not so obvious in QM how the electron actually moves around in the atom and what to do about a current and area. We use the classical equations: μn = (-e/2m) Ln, and U = (eB/2m) nħ. The combination eħ/2m is common, and we define the "Bohr magneton" as μB = eħ/2m = 5.79x10-5 eV/T. Now the s states have L=0, so μ = 0, and there is no effect from the magnetic field. For the d state shown to the right, μ is aligned to L, is quantized, and has several possible projections, so U = -μ.B is quantized, and the level is split into (2l+1) sub-levels. Thursday, April 4, 2013 The Schrödinger Eq. atom in a magnetic field II We have U = -μ.B = -μzB, with μz = (-e/2m)Lz = -ml(eħ/2m) = -mlμB. Thus U = mlμBB. The (2l+1) sub-levels have even separations / splittings of μBB. The magnetic field removes the degeneracy. Pieter Zeeman discovered this in 1896. But so far, the m=0 states of the same n have equal energy. Thursday, April 4, 2013 iClicker You put an n=3 Hydrogen atom in a magnetic field. You see photons corresponding to n=3 to n=1 transitions. How many lines do you see? a) 1 b) 3 c) 5 d) 8 e) 9 Thursday, April 4, 2013 At this point since the splittings are all the same it might seem there are 5 distinct energies, but we will see in the next few slides that 2 of these are not allowed. Selection Rules Consider the splitting of a d state into 5 levels as the B field is increased. Now consider a transition from, e.g., a 3d to a 2p state, which has split into 3 levels. How many different photon energies are possible? It seems we have (Ed ± (0,1,2)μBB) - (Ep ± (0,1)μBB) = (Ed - Ep) ± (0,1,2,3)μBB. But there are not 7 possible energies. The photon can only carry of 1 unit of angular momentum, so we are limited to Eg = (Ed Ep) ± (0,1)μBB. Only certain transitions are possible. Thursday, April 4, 2013 Allowed transitions md mp Δm = md-mp = mγ 2 1 1 2 0 2 2 -1 3 1 1 0 1 0 1 1 -1 2 0 1 -1 0 0 0 -1 0 1 The 6 md < 0 transitions are left as an exercise for those interested. Thursday, April 4, 2013 The table shows the change in the z-component of the orbital angular momentum. Since angular momentum is conserved, the photon must carry at least an angular momentum l = Δm, since its z-component of angular momentum must equal the change in the atomic z component. (Angular momentum, like momentum, is conserved in each component.) But the photon is spin 1, so the red-colored lines are forbidden. Only 3 of the 7 possible energies are allowed. The Stern-Gerlach Experiment In the magnet the energy of an atom with a magnetic moment is U = -μzB = mlμBB. But if the magnetic field is not constant, classically their is a force F = ∇μ.B = mlμB(dB/dz). So a bunch of atoms coming from the oven will separate into 2l+1 separate bunches if there is a B field with enough spatial variation. Thursday, April 4, 2013 When you run the experiment, you can get an even number of blobs. Thursday, April 4, 2013 The Stern-Gerlach Experiment and More on Line Spectra - iclicker! For odd Z nuclei, not only do we get an even number of blobs, but we also can get extra splittings of spectral lines in magnetic fields not explained by the normal Zeeman effect - the book has some confusing pictures on this. With the angular momentum, we have an integral magnitude l, and the degenerate states are split into the 2l+1 levels by a magnetic field. But 2l+1 is always an odd number, so how can we get an even number of blobs in the Stern Gerlach experiment? a) The data are wrong - there is an invisible line off to the side. b) You don't see the m=0 line, since there is no force on those atoms. c) 2l+1 can be even for integral l - what's the problem? d) l can be half-integral, not just integral. e) the electron has an intrinsic spin of 1/2. Thursday, April 4, 2013 Electron spin Subatomic particles have intrinsic spins, that can be integral (0 for the pi meson, 1 for the photon, rho meson, ...) or halfintegral (1/2 for the electron or neutrino or proton or neutron, 3/2 for the Delta resonance, ...) The classical analogy almost always made is to a top or the earth, which have rotational angular momentum, in addition to, in the case of the each, orbital angular momentum of revolution around the sun. This is a bad analogy as the particles are not actually rotating. The algebra of spin is: sz = ±½ħ = msħ, S = √((1/2)(3/2)) ħ = √(¾) ħ, and μz = -2.00232 (e/2m) sz. Thursday, April 4, 2013 The algebra of spin is: Huh? 2.00232? sz = ±½ħ = msħ, S = √((1/2)(3/2)) ħ = √(¾) ħ, and μz = 2.00232 (-e/2m) sz = g (-e/2m) sz. = gμB/2. In non-relativistic quantum mechanics, there is not natural explanation for spin; it is added in an ad hoc manner. Two different ways of combining relativity with quantum mechanics were developed in the 1920s (now combined into QFT). One (Klein and Gordon) describes particles with integral spin. The other (Dirac) describes particles with half-integral spin. In a first approximation, the electron has a g factor of 2. You might say spin contributes twice as much to the magnetic moment as orbital angular momentum. In modern QED theory higher order corrections can be calculated, these make the g factor of point particles (electrons) slightly different from 2, and predict it to about 1/1012. Thursday, April 4, 2013 Anomalous Zeeman Effect We now have a spin-½ electron. As a result, each of our atomic energy levels can have two electrons, one "spin up" and the other "spin down". Thus, even for s orbits, the spin of the electron can give rise to a splitting between the spin-up and spin-down states in the atom, and we can get doublets of lines. Note that with spin 1/2, but g = 2, the coefficient of B above is the same as for orbital angular momentum. Thursday, April 4, 2013 Spin-Orbit Coupling & fine structure In the rest frame of the electron - thinking semi-classically the proton is revolving about the electron. Since the proton is a charged particle, it generates a magnetic field at the position of the electron. (In the view with the proton at rest the electron generates a B field at the position of the proton.) As a result, there is a magnetic correction to the energy U = μ.B where B is proportional to L. Since for the electron μ is proportional to S, we can rewrite the energy as proportional to L.S; this is preferred since we know L and S of the orbit, but not the B field. This is then referred to as the spin-orbit force. It leads to a splitting of spin-up and spin-down electron states even in the absence of a magnetic field! These splittings related to the magnetic interactions are referred to as "fine structure". Thursday, April 4, 2013 Proton Magnetic Moment & hyperfine structure Note also the proton has an intrinsic magnetic moment. Like the electron, the proton is a spin-1/2 particle, and its magnetic moment is given by μ = g (-e/2m) sz. But here the mass is the proton mass, which is about 1800 times the electron mass, and the magnetic moment is much smaller. As a result, we use instead μ = gμN/2, where μN is the "nuclear magneton". Finally the proton is a particle with structure, so g = 5.586, it is not close to 2. This is because the proton has a complex internal structure. Nuclei, made up of protons and neutrons, tends to have magnetic moments of similar size to the protons. The splittings resulting from interactions of the nuclear magnetic moments with the B field from the electron lead to small splitting, referred to as "hyperfine structure" since they are much smaller than the fine structure splittings resulting form the electron magnetic moment interactions. Thursday, April 4, 2013 Adding Angular Momenta Classically, if we add angular momentum vectors, depending on their relative orientation, we can get any result from the sum of the two vectors to the difference. This idea holds in quantum mechanics. For a single electron for example, we would write J = L + S, the total angular momentum is the orbital angular momentum plus the spin angular momentum. But the angular momenta are still quantized. Thus j = |l ± ½|. (We use the magnitude, since otherwise for l = 0, we could get j = -½.) Or J = √(j(j+1)) ħ. Thursday, April 4, 2013 Summary In the simple picture, we have levels with quantum numbers n, l, ml (and ms) that are degenerate. We find that an external magnetic field removes the degeneracy, splitting a level with angular momentum l into (2l+1) levels. With the spin of the electron, we find an external field leads to doublets for (spin up vs spin down) even for s states. Even without an external field we have magnetic effects: The magnetic interaction between magnetic fields from protons and electron magnetic moment leads to an L.S force and "fine structure". The magnetic interaction between the smaller proton magnetic moment and the magnetic field from the electron leads to a similar "hyperfine structure". Thursday, April 4, 2013 Many Electron Atoms So far we have considered mainly the hydrogen atom. When we go to heavier atoms, we can largely treat the nucleus as being a point object with some mass and some charge Ze. But the electrons in orbit about the nucleus interact with each other. You can see that if one electron is generally outside another, then in effect the binding charge of the nucleus is reduced by 1. Thus we cannot simply say that the energy of an orbit n becomes En = -13.6Z2/n2 eV. Na: Z=11, En = -1646/n2 eV But E(3s) = -5.14 eV E(3p) = -3.035 eV E(3d) = -1.521 eV E(4s) = -1.947 eV Thursday, April 4, 2013 Pauli Exclusion Principle For many electron atoms, we cannot simply use the 1/r Coulomb potential, but the potential is mainly U(r), independent of θ and φ. As a result, the characterization of the angular momentum states still holds. We still have n>0, 0≤l<n, |ml|≤l, and ms=±½. The Pauli exclusion principle is important for understanding atomic structure: for electrons (and other spin-½ particles, or more generally Fermions) each state can hold only 1 particle. No two electrons can have the same 4 quantum numbers. For example: the more bound orbits fill first, and when a major shell is filled there is a large energy gap up to the next orbit. This leads to the non-reactive "noble" gases. There are techniques to do actual calculations; they are too complicated for this class. Also: atoms with similar configuration of outer electrons tend to be similar in their chemical properties - this leads to the Periodic Table. Thursday, April 4, 2013 Closing iClicker You have one electron in the l = 1 2p state, and a second electron in the l=2 3d state. What are the possible total angular momenta of the two electrons? (The answers are j in units of ħ, not J = √(j(j+1)) in units of ħ.) a) 1, 2, 3 b) 0, 1, 2, 3, 4 c) 3 d) when you add the angular momenta of two electrons, it can be anything between the limits - it is continuous, not quantized e) none of the others are right, since the electron is spin ½. The answer has ½-integral numbers. Thursday, April 4, 2013 Closing iClicker You have one electron in the l = 1 2p state, and a second electron in the l=2 3d state. What are the possible total angular momenta of the two electrons? (The answers are j in units of ħ, not J = √(j(j+1)) in units of ħ.) a) 1, 2, 3 b) 0, 1, 2, 3, 4 We add l=1 to l=2 to s=½ to s=½; they can add to numbers from 0 to 4. c) 3 d) when you add the angular momenta of two electrons, it can be anything between the limits - it is continuous, not quantized e) none of the others are right, since the electron is spin ½. The answer has ½-integral numbers. Thursday, April 4, 2013























![NAME: Quiz #5: Phys142 1. [4pts] Find the resulting current through](http://s1.studyres.com/store/data/006404813_1-90fcf53f79a7b619eafe061618bfacc1-150x150.png)