* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
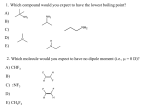
Download 1 Q. What are Saturated and unsaturated hydrocarbons?imp
Survey
Document related concepts
Homoaromaticity wikipedia , lookup
Marcus theory wikipedia , lookup
George S. Hammond wikipedia , lookup
Fischer–Tropsch process wikipedia , lookup
Woodward–Hoffmann rules wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Petasis reaction wikipedia , lookup
Wolff–Kishner reduction wikipedia , lookup
Ene reaction wikipedia , lookup
Asymmetric induction wikipedia , lookup
1,3-Dipolar cycloaddition wikipedia , lookup
Cracking (chemistry) wikipedia , lookup
Ring-closing metathesis wikipedia , lookup
Hydroformylation wikipedia , lookup
Aromaticity wikipedia , lookup
Strychnine total synthesis wikipedia , lookup
Transcript
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
Q. What are Saturated and unsaturated hydrocarbons?imp
SATURATED HYDROCARBONS
Hydrocarbons containing carbon–carbon single bond are called saturated hydrocarbons.
e.g. alkane and cycloalkanes.
ALKANE
: General formula is CnH2n+2 (n is a positive whole number for all H/Cs)
CYCLOALKANES: General formula is CnH2n (n is a positive whole number for all H/Cs)
UNSATURATED HYDROCARBONS
Hydrocarbons containing carbon–carbon double or triple bond are called unsaturated
hydrocarbons. e.g. alkene , alkyne and aromatics.
ALKENES: general formula is CnH2n
ALKYNES: general formula is CnH2n–2
AROMATICS: usually contain one or more rings of six C atoms called benzene rings
(n is a positive whole number for all H/Cs) C6H2n-6
NATURE OF CARBON CHAIN
Saturated ,C-C
Unsaturated ,C=C
Unsaturated ,C≡C
PRIMARY SUFFIXES
-ane
-ene
-yne
GENERIC NAME
Alkane
Alkene
Alkyne
ALKANES: General formula: CnH2n+2 simplest organic compounds.
CnH2n+2
n=
formula
of
alkane
name
of
alkane
1
2
3
4
5
6
7
8
9
10
CH4
C2H6
C3H8
C4H10
C5H12
C6H14
C7H16
C8H18
C9H20
C10H22
metha
ne
ethane
propan
e
butane
pentan
e
hexane
heptane
octane
nonane
decane
HYBRIDISATION:
The process of merging (intermixing) of the orbital’s of similar energies so as to form
new orbital’s called hybrid orbital’s.
In order to explain characteristic geometrical shapes of polyatomic molecules
concept of hybridization is used. In alkane hybridisation is sp3 .
All the carbon-carbon and C-H bonds in alkanes are strong sigma (σ) bonds.
Every C-H bond length = 1.112A0 and C-C bond has a and 1.54A0 .
All bond angles are regular at tetrahedral position of four bonds of carbon, the C-C chain
is zig zag and not linear as usually written for sake of simplicity.
Propane
Propane (3d)
A stereochemical formula is the full structural formula in terms of the 3D structural or
spatial arrangement of the atoms.
Composition of methane molecule: CH4.
Geometry: of methane: Tetrahedral in structure in which carbon is central atom and
four H-atoms are surrounding it in three-dimensions.
Bond Angles:
H-C-H bond angles are 109.5o.
Bond Length:
All C-H bonds are 1.09Ao.
1
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
HYBRIDISATION IN METHANE : sp3
1(2s)+ 3(2px, 2py, 2pz) orbital’s (sp3 hybridisation) 4 (sp3 hybrid orbital’s) of 109028’
alignment forming regular tetrahedron. Each orbital has ¼s (25%) character and ¾ p
(75%) character.
12
(ground state electronic configuration) 1s22s22px12py12pz0
6C
12
(excited state electronic configuration) 1s22s1px12py12pz1
6C*
One e- from s orbital is moved from 2s orbital to 2pz orbital which is in higher energy
state. The input energy for exciting an electron is available from lattice energy of
formation, released when bond are formed. Initially energy has to give so as to start the
reaction.
In the excited state one 2s orbital of the C* and three 2p orbitals (Px, Py, Pz) of C*
intermix to form four hybrid orbitals of sp3 hybridisation. These four hybridised orbitals
aligned themselves in such a way that bond angle must be 109028’ so as to have
minimum energy and maximum stability. Orbital structure is shown in diagram. These
04 sp3 orbitals overlaps with 1s1 of four H orbitals forming sigma bonding by end to end
overlap.
GEOMETRY/ SHAPE OF ETHANE:
Sp3
TYPES OF CARBON ATOMS :
(a) PRIMARY CARBON or 10 carbon atom
Carbon atom attached to one or no carbon atoms.
CH3C(CH3)2CH(CH3)CH2CH3
2
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
(b) SECONDARY CARBON or 20 carbon atom
10 40
30
20 10
Carbon atom attached to two carbon atoms. Also known as n.
(c) TERTIARY CARBON or 30 carbon atom
Carbon atom attached to three other carbon atoms. Also known as iso.
(d) QUARTERNARY CARBON or 40 carbon atom.
Carbon atom attached to four other carbon atoms. Also known as neo.
H atoms attached to primary, secondary, tertiary or quaternary carbon atoms are
correspondingly names as primary, secondary, tertiary and quarternary hydrogen atoms.
Q. Mention primary, secondary and tertiary carbons and hydrogens in the following
Compound?
10
Ans 10
20
40 10
10
H atoms attached to primary, secondary, tertiary or quaternary carbon atoms are
correspondingly names as primary, secondary, tertiary and quarternary hydrogen atoms.
Q.In the alkane H3C – CH2 – C(CH3)2 – CH2 – CH(CH3)2, identify 1°,2°,3° carbon atoms
and give the number of H atoms bonded to each one of these.
STRUCTURAL ISOMERISM:
1. CHAIN ISOMERISM: (NUCLEAR OR SKELTON ISOMERISM)
Compounds having same molecular formula but different arrangement of carbon
chains (skeletons), within the molecule are called chain isomers and the phenomenon
is termed as chain isomerism Example: Chain isomers of : C5H12
CHAIN ISOMERS OF THE ALKANE MOLECULAR FORMULA C5H12
(1)
34oC, linear.
,
, pentane, volatile colourless liquid, bpt
(2)
,
, methylbutane (2-methylbutane, but 2- not
needed), volatile colourless liquid/gas, bpt 28oC, minimum branching.
(3),
,
maximum branching.
, 2,2-dimethylpropane, colourless gas, bpt 9.5oC,
3
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
2. RING CHAIN ISOMERISM :
Due to difference in mode of linkage in
C atoms and the isomers may have
either open chain or closed structure. e.g. C3H6
CH3-CH=CH2
3. POSITIONAL ISOMERS OF MOLECULAR FORMULA
(1) 1-bromopropane,
halogenoalkane,
(2) 2-bromopropane,
halogenoalkane.
,
,
C3H7Br
, bpt 71oC, primary
, bpt 59oC, secondary
4. CONFORMATIONS IN ALKANE:
Conformations or conformers or rotamers or conformational isomers:
The different arrangements (spatial) of atoms of a molecule which can be obtained by
rotation around a C-C single bond are called conformations or conformers or rotamers.
CONFORMATIONAL ISOMERES: The isomers which differs in the conformation
are known as conformational isomers.
REPRESENTATION OF CONFORMATIONS:
(a)Sawhorse projection representation (b) Newman projection
(a)Sawhorse projection representation
In this representation molecule is viewed along the axis of the axis at an angle. C-C
atom is a straight line. Front Carbon is shown as the lower left hand carbon while rear
carbon is shown as upper right hand carbon. Three C-H bonds are shown by the three
lines on the each lower and upper carbon.
Sawhorse projection
Newman projection formulae:
Two C-C sigma bonded atoms are viewed from the front along the C-C bond axis so that
only front C is seen. front C is seen as junction of 3C-H bond while rear Carbon is shown
as circle as shown in figure above.
4
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
CONFORMATIONS OF ETHANE: CH3- CH3
Imagine that one of the methyl group is rotated
along the C-C axis keeping the rest molecule
undisturbed. An infinite no. of possible
arrangements of the rotated methyl group w.r.t
undisturbed methyl group are possible,
each of these possible arrangement represents
a conformations. However for the sake of
convenience each rotation is done in the instalment
of 600. Thus we will obtain six no. of different
conformations of ethane. Out of these six, extreme two are worth to study.
Because they are different from each other while other four are similar to each other but
of course different from the extreme two ends.
(a) One extreme conformations will be such in which the rear methyl group is completely
eclipsed by the front methyl group and thus only the front methyl group and thus only
the front methyl group i.e. three H atoms of the methyl group nearer to the eye are
visible. Such conformations is known as eclipsed conformations.
Conformations Eclipsed
Projections
Conformation in which
hydrogen atoms attached
to two carbons are as
closed
together
as
possible
Staggered
Skew
Conformation in which Any
other
hydrogen atoms are as far intermediate
apart as possible
conformation
in between
eclipsed and
sawhorse
Sawhorse
projection
Newman
projection
ECLIPSED CONFORMATIONS:
In this conformation, the H atom of one carbon are directly behind the other.
consequently the repulsion in these atoms is maximum.
In the Newmann projection formula of the eclipsed form, the H atoms are crowded i.e.
the two H atoms come close to each other.
5
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
(b) Another extreme conformations will be such in which the rear methyl group has
been rotated upside down and thus both the methyl group i.e. all the six H atoms are
visible and are as far apart from each other as possible. such conformations is known as
staggered.
STAGGERED CONFORMATIONS: In this conformation, H atoms of the two C atoms are
staggered wrt to one another. As they are at maximum distance apart and have
minimum repulsion between them. Both the conformations are represented as below:
SKEW CONFORMATIONS:
(c) The infinite no. of possible intermediate conformations between the two extreme
conformations between two extreme conformations are referred to as skew
conformations. In these conformations, H atoms are close than in staggered but away
than in eclipsed conformations.
STABILITY OF CONFORMATIONS:
Staggered conformations in which H atoms are as far as apart as possible is
thus most stable while the eclipsed conformations in which H atoms are
perfectly eclipsed is the least stable. Stability of skew conformations lie in
between these are in the following order:
Staggered > Skew > Eclipsed
TORSIONAL STRAIN: weak repulsive interactions between the bonds or electron pair
of bonds on adjacent C atoms. More is the torsional strain lesser is the stability.
TORSIONAL BARRIER: The difference in the energy between staggered and eclipsed
conformations which is equal to 12.5KJ/mol. The energy difference is very small.
Therefore two conformations of ethane goes on changing from one form to another and
consequently it is not possible to isolate the different conformations of ethane.
Q. Eclipsed conformation is less stable than staggered conformation of ethane. Explain?
Ans. It is due to repulsive interactions (non bonded) between bonded pair of electrons,
i.e. electron pair which form six C-H bonds in ethane. In the staggered conformations,
the electron clouds of six C-H bonds are as far as possible with the result there is
minimum repulsive interactions between these electron clouds. Hence this conformations
is quite stable. On the other hand skew and eclipsed conformations, the electron clouds
starts coming closer. This causes repulsive interactions which is maximum in the
eclipsed. Therefore potential energy are of order: Eclipsed
> Skew > Staggered
Stability :
Staggered > Skew > Eclipsed
6
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
TORSIONAL STRAIN: the repulsive interaction between the clouds
affects the solubility of a conformation is called torsional strain.
which
Staggered conformations has the lease torsional strain while eclipsed having maximum.
Due to torsional strain, certain energy called torsional energy, is required to allow the
rotation around the C-C single bond. in other words ethane molecule having staggered
conformation will have to come cross an energy barrier equivalent to the torsional
energy for being converted into eclipsed conformations.
Energy difference in staggered and eclipsed is found to be 2.8Kcal/ mol which constitute
the energy barrier to rotate about C-C bond i.e.for conversion of staggered to eclipsed.
This energy barrier is too small for either form to remain stable. i.e. two forms
are interconvertible.
This implies that rotation about the C-C single bond in ethane is almost free for
all practical purposes, and it is not possible to separate the different
conformations of ethane the mostly in staggered form.
CONFORMATION IN CYCLO ALKANES:
CHAIR CONFROMATION IS MOST STABLE:
Reason: (a) Adjacent H on Carbon is staggered and more distance between them,
resulting lesser repulsions and lesser energy thus more stability.
(b) Energy of boat conformation is 44KJ/mol more than that of chair conformation.
Therefore cyclohexane mainly exists as chair form.
Q. name two extreme type of conformationsof ethane?
Ans. stagerred and eclipsed
Q. Out of chair and boat conformations of cyclohexane? Which is more stable?
Ans. chair form is more stable because of minimum repulsions and lesser energy.
7
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
METHODS OF PREPARATION OF ALKANES:
FROM ALKENE AND ALKYNE ALKANE
A. SABATIER AND SANDEREN’S REACTION: HYDROGENATION REACTION
It involves catalytic (Ni or platinum) hydrogenation of unsaturated hydrocarbons like
alkene and alkyne to alkane. Methane can’t be obtained by this method.
It is a type of addition reaction.
RCH=CHR + H2 Ni 200-3000C
RCH2CH2R
Alkene
Alkane
0
CH2=CH2 + H2 Ni 200-300 C
CH3-CH3
Ethene
Pt
Ethane
CHCH + 2H2
Ethyne
Ni 2500C or 523K
Pt
CH3-CH3
Ethane
FROM ALKYL HALIDE ALKANE
B. WURTZ REACTION. This reaction involves the treatment of an alkyl halide (RX) with
sodium metal in the presence of dry ether to form higher symmetrical alkane. This
reaction can be used for ascending of series. For example,
R-X+2Na
+X-R
Dry ether
R-R + 2NaX
Alkyl halide
Alkane
C2H5-Br
+ 2Na + Br-C2H5
Ethyl bromide
dry ether
C2H5-C2H5 + 2NaBr
Butane
CH3-I
+ 2Na + I-CH3
dry ether
CH3-CH3 + 2NaBr
Ethyl bromide
Ethane
LIMITATIONS OF THE REACTION:
(i) Methane cannot be prepared by this reaction.
(ii) This reaction fails with 30 alkyl halides.
(iii) This is not a suitable reaction to prepare higher unsymmetrical alkanes, as byproducts are also formed during the reaction and thus their separation becomes difficult.
Q. Name the products formed when methyl bromide and ethyl bromide is treated with
sodium metal in ether? Ans. ethane+ propane +butane
8
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
Q. Give mechanism of Wurtz reaction.
Ans. Mechanism :
(A) INTERMEDIATE FORMATION OF AN ORGANOMETALLIC COMPOUND:
CH3CH2Br + 2Na
CH3CH2-Na+ + NaBr
CH3CH2-Na+ + CH3CH2Br CH3CH2CH2CH3 + NaBr
(B) INTERMEDIATE FORMATION AS FREE RADICALS.
CH3CH2Br + 2Na CH3CH2. + NaBr
CH3CH2. + CH3CH2.
CH3CH2CH2CH3
Butane
mechanism may be ionic or free radical mechanism but normally free radical is
preferred. If Mechanism is free radical reaction it involves following steps:
(a)Chain initiation (b)Chain propagation (c)Chain termination step.
C. FRANKLAND REACTION:
2R-X
+ Zn R-R + ZnX2
Alkyl halide
Alkane
CH3Br+ CH3Br + Zn ZnBr2 + CH3-CH3
Bromo methane
Ethane
C2H5I+ CH3I + Zn ZnI2 + CH3-CH3
Iodo ethane
Ethane
D. REDUCTION REACTIONS:
Common reducing agent: LiAlH4 (anhyd ether), Na/ alcohol, NaBH4 (Protic solvent, ROH,
H2O), H2 PtO2+ heat. Or Ni + H2 (g), Zn/ HCl , HI, Red Phosphorus, P+HI
Other Reducing agent:
Zn+ CH3COOH , Zn+ NaOH, Zn Cu couple + alcohol, aluminium amalgam and alcohol,
:LiAlH4. ,
(I)REDUCTION OF ALKYL HALIDES ALKANE
RX
+
[H]
Red P/ 423K
RH
+
Alkyl halide
Pressure
Alkane
C2H5Br +
C2H5I +
2[H]
2[H]
HX
Halogen acid
Red P 423K C2H6 + HBr
Red P/HI 423K C2H6 + I2
Function of red P is to remove iodine formed in the reaction by forming PI3 which is
volatile , otherwise alkane will react back to give alkyl halide.
C2H5Br +
2[H]
LiAlH4 C2H6 + HBr
C2H5Br +
2[H]
Zn/ HCl C2H6 + HBr
(II) REDUCTION OF ALCOHOLS:
ROH
+ 2HI Red P RH + H2O + I2
C2H5OH + 2HI Red P C2H6 + H2O + I2
(III) REDUCTION OF ALDEHYDE, KETONE, ACIDS
RCHO + 4HI Red P RCH3 + H2O + 2I2
CH3CHO + 4HI Red P CH3CH3 + H2O + 2I2
Ethanal
ethane
RCOR
+ 4HI Red P RCH2CH3 + H2O + 2I2
Ketone
CH3COCH3 + 4HI Red P CH3CH2CH3+ H2O + 2I2
Propanone
Propane
9
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
RCOOH
+ 6HI Red P RCH3 + 2H2O + 3I2
Carboxylic acid
Alkane
CH3COOH + 4HI Red P CH3CH3+ 2H2O + 2I2
Propanoic
Ethane
(IV) CLEMMENSEN REDUCTION:
Carbonyl group of aldehydes and ketones is reduced to CH2 group on treatment with zinc
amalgam and concentrated hydrochloric acid
RCHO
+
4[H] Zn-Hg, HCl RCH3 + H2O
Aldehyde
Alkane
CH3CHO
+
4[H] Zn-Hg, HCl CH3CH3 + H2O
Ethanal
Ethane
CH3COCH3 +
4[H] Zn-Hg, HCl CH3-CH2-CH3 + H2O
Propanone
Propane
C6H5COCH3 + 4[H] Zn-Hg, HCl
C6H5-C2H5 + H2O
Acetophenone
Ethyl benzene
Clemmensen Reduction is widely used when carbonyl compounds are sensitive
to alkalies.
(V) WOLFF-KISHNER REDUCTION:
Carbonyl group of aldehydes and ketones is reduced to CH2 group on treatment with
hydrazine followed by heating with sodium or potassium hydroxide in high boiling
solvent such as ethylene glycol
RCHO + H2NNH2 R-CH=N-NH2 + KOH, glycol R-CH3 + N2
- H2O
Hydrazone
453-473K
CH3CHO + H2NNH2 + KOH, glycol CH3-CH3
+ N2
-H2O
453-473K
Aldehyde
alkane
CH3COCH3 + H2NNH2 + KOH, glycol CH3-CH2-CH3 + N2
-H2O
453-473K
Ketone
alkane
C6H5COCH3 + H2NNH2 + KOH, glycol C6H5-CH3
+ N2
Benzophenone
-H2O, 453-473K toluene
Wolf kishner reduction is used when carbonyl compounds are sensitive to acids.
Q. How will you convert acetaldehyde to ethane and acetone to propane?
Ans Wolf kishner reduction
CH3CHO + H2NNH2 + KOH, glycol CH3-CH3
+ N2
-H2O
453-473K
CH3COCH3 + H2NNH2 + KOH, glycol CH3-CH2-CH3 + N2
-H2O
453-473K
Or Clemmensen Reduction
CH3CHO
+
4[H] Zn-Hg, HCl CH3CH3 + H2O
Ethanal
Ethane
CH3COCH3 +
4[H] Zn-Hg, HCl CH3-CH2-CH3 + H2O
Propanone
Propane
10
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
E. DECARBOXYLATION REACTION OR SODA LIME REACTION :
RCOONa
+
NaOH
Na2CO3
+
RH
Sodium salt of carboxylic acid
sodium carbonate
Alkane
CH3COONa +
NaOH
Sod. Salt of ethanoic acid
CH4
+ Na2CO3
Methane
Net result of this reaction is the removal of CO2 in the form of sodium carbonate from a
carboxylic acid, therefore reaction is known as de-carboxylation reaction.
This reaction is used in the descending of series.
Q. sodium salt of which acid will be needed for the preparation of propane? Write
chemical equation for the reaction?
Ans. Butanoic acid reaction do yourself
F. KOLBE’S ELECTROLYTIC METHOD:
Alkali metal salts of carboxylic acids undergo decarboxylation on electrolysis of their
aqueous solutions and form hydrocarbons having twice the number of carbon atoms
present in the alkyl group of the acid.
Electrolysis of sodium salt of carboxylic acids
2CH3COO-Na+ + 2H2O CH3-CH3 +2CO2 + 2NaOH + H2
Sod salt of ethanoic acid
Ethane (Anode )
at cathode
+
+
CH3COO Na CH3COO- + Na
H2O H+ + OH-
Remember: (i) methane can’t be prepared by this method.
(ii) electrolysis of HCOONa produces only H2 and CO2.
(iii) unsymeetrical alkane, i.e. alkane having odd no. of C atoms cannot be prepared by
Kolbe method.
CH3-CH3 symmetrical Ethane
CH3CH2CH3 unsymmetrical propane
G. FROM GRIGNARD REAGENT (RMgX) RH ALKANE.
RMgX
+ H2O dry ether RH + MgX(OH)
Alkyl halide
alkane
CH3MgBr
+ H2O dry ether CH4 + MgBr(OH)
Methyl magnesiumbromide
Methane
R’OH + RMgX R’H + ROMgX (alcohol to alkane)
Alcohol
11
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
CH3OH + CH3MgBr
Methanol
dry ether CH4 + CH3MgBr(OH)
methane
Q. How are alkanes prepared by Grignard's reagent?
CH3OH + CH3MgBr dry ether CH4 + CH3MgBr(OH)
Methanol
methane
H SPECIAL METHOD FOR PREPARATION OF ALKANE:
Al4C3+ 12H2O 4Al(OH)3 + 3CH4 ↑
Aluminium carbide
methane
C+ 2H2 electric arc 1473K CH4
I. COREY HOUSE REACTION:
RBr + Li ether RLi+ LiBr
2RLi + CuI Li(R)2Cu +LiI
Li(R)2-Cu+ R’Br R-R’ + RCu+ LiBr
(main reaction)
USES OF ALKANES:
(a) Lower alkanes upto C4 like LPG is used as domestic fuel.
(b) Alkanes are used for the manufacturing of chemicals like CH3OH, CH3Cl, CCl4 etc.
(c) In the manufacturing of fuel like gasoline which is a mixture of alkane alkene and
aromatic hydrocarbons.
Higher is the quantity of branched chain hydrocarbon better is the octane No, better is
the fuel having anti-knocking property of fuel.
(d) Methane is used in the manufacturing of carbon black used in black paints, printer’s
ink and as a filler in the rubber industry.
PHYSICAL PROPERTIES OF ALKANES:
A. PHYSICAL STATE:
C1 to C4 are colourless and odourless gases,
C5 to C17 are liquids and
C> C18 are colourless solids at 298 K.
Reason: Hydrocarbons are non- polar because of the covalent nature of C-C and C-H
bonds. Thus, they have weak van der Waals forces.
B. BOILING POINTS: BRANCHED CHAIN HAS LOWER BP THAN STRAIGHT
CHAIN.
Alkanes have low boiling points because they have weak Vander Waal’s forces. Higher
alkanes with higher molecular masses have high boiling points than lower alkanes with
low molecular masses. Vander Waal’s forces surface area
This is because magnitude of Vander Waal’s forces increases with increase in molecular
mass or increase in surface area.
Q. Boiling points of isomeric alkanes goes on decreasing with increased branching. Why?
Ans Boiling points of branched chain alkanes are lower than the corresponding straight
chain alkanes. This is because with branching the molecules become more compact and
hence the surface area decreases. E.g. n-pentane has higher bp than iso pentane.
Alkane CH3CH2CH2CH2CH3
(CH3)2CHCH2CH3
(CH3)4C
n-Pentane
iso- Pentane
neo- Pentane
0
0
Boiling point:
36 C
28 C
9.50C
12
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
Q. n-pentane has higher boiling point than neo pentane. Explain.
Ans. Boiling points of branched chain alkanes are lower than the corresponding straight
chain alkanes. This is because with branching the molecules become more compact and
hence the surface area decreases. Therefore n-pentane has higher bp than neo-pentane.
Q. Which of the following has higher bp?
(a) 2-Methylpentane
(b) 2,3-Dimethylbutane (c) 2,2-Dimethylbutane
Reason: least branching
C. MELTING POINTS:
Q. Alkanes with even no. of carbon atoms have high melting point as compare to
alkanes with odd no. of carbon atoms why?
Ans. The alkanes with even number of carbon atoms (i.e. butane, ethane ) have higher
melting points as compared to the immediately next lower alkane with odd number of
carbon atoms. This is because alkanes with even number of carbon atoms have
symmetrical structure and hence it results in closer packing in the crystal structure.
C5H12 MP = 143.3K C6H14 MP 178.5K
D. SOLUBILITY:
Alkanes are soluble in non- polar solvents like benzene, toluene, acetone etc and are
insoluble in polar solvents like water since alkanes are non- polar molecules.
Like dissolves like principle: Polar dissolves in polar solvents and non polar in non
polar solvents.
CHEMICAL PROPERTIES OF ALKANES:
Alkanes are not very reactive molecules. Most reactions require some energy
input to initiate a reaction e.g. high temperature and catalyst for cracking, uv light for
chlorination or a spark to ignite them (initiating free radical reactions).
Q. Why are alkanes stable as compared to alkene and alkyne?
Ans. Alkanes are extremely stable and un-reactive substances because:
(a) small difference in Electronegativity between C and H.
Therefore C-H bond is almost non- polar and C-C bond is completely non polar. Attack of
nucleohile and electrophile attack on the alkane finds no site for attack. Therefore no
reactive as compared to alkene and alkyne.
(b) Bond Strength C-C and C-H bonds are strong bonds:
Alkanes having these two types of bonds are unaffected by acids, alkali and oxidising
agents under normal conditions.
The single covalent C-C (bond enthalpy 348 kJ mol-1) and C-H (bond enthalpy 412 kJ
mol-1) bonds are very strong so bond fission does not readily happen. The carbon
atom radius is small, giving a short and strong bond with other small atoms. Therefore
the reactions will tend to have high activation energies resulting in slow/no reaction.
CHEMICAL REACTIONS OF ALKANES:
1.
2.
3.
4.
SUBSTITUTION
DECOMPOSITION
ISOMERISATION
OXIDATION
13
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
CHEMICAL REACTIONS OF ALKANE:
SUBSTITUTION REACTIONS:
1. HALOGENATION: (IMP.)
14
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
(a) CHLORINATION (B) BROMINATION (C) IODINATION
It is a type of substitution reaction where H atom of alkane is substituted with Cl
atom. This reaction takes place in the presence of sunlight (hv) at a temperature
of 250-4000C (673K).
CH4+ Cl2 SUNLIGHT (hν) 673K CH3Cl + HCl
CH3Cl+ HCl (hν) 673K
CH2Cl2
CH2Cl2+ HCl (hν) 673K
CHCl3 + HCl
CHCl3+ HCl (hν) 673K
CCl4 + HCl
In the uv light catalysed reaction of bromine and propane gases, the free radical
substitution reaction can produce two initial mono-substitution products.
CH3CH2CH3 + Br2
{CH3CH2CH2Br or CH3CHBrCH3} + HBr
Relative reactivity: F2 > Cl2 > Br2 > I2
Relative reactivity of 10 20 30 H replacement by halogen:
Tertiary H (30) > secondary (20) > primary (10)>CH4
LIMITATIONS:
It is difficult to stop halogenations at CH3Cl. when equimolar amount of CH4 and
Cl2 is used, it gives mixture of four products viz CH3Cl, CH2Cl2, CHCl3, CCl4.
MECHANISM OF CHLORINATION REACTION (FOR INFORMATION)
(i) Free Radical Substitutions
e.g. alkanes undergo free radical substitution
reactions
CH4+ Cl2 light , hv CCl4+ HCl radicals are chlorine free radicals (Cl.)
Methane
Carbon Tetrachloride
MECHANISM STEPS:
(a) Chain initiation (requires energy- endothermic reaction)
Cl-Cl hv or 2500C 2Cl. radical formation
Reaction with hv= photolysis, heat (Δ) thermolysis
C6H5COOOC6H5
dark 2C6H5COO.
C6H5COO.
C6H5. + CO2
C6H5. + 2Cl. C6H5Cl + Cl.
(b) chain propagation steps: (release energy- exothermic reaction)
Cl. + H- CH3 HCl + .CH3
.CH3 + Cl-Cl CH3-Cl + .Cl
(c)Chain termination step.
.Cl + .Cl Cl-Cl
.CH3+ .Cl CH3Cl
.CH3+ .CH3 CH3-CH3
Ease of substitution at various carbon atoms is of the order:
Tertiary > secondary > primary
2. NITRATION OF ALKANE BY HNO3:
It is a type of substitution reaction where H atom of alkane is substituted with
–NO2. Temperature: 400-5000C (vapour phase nitration)
CH3CH2CH3 +
HNO3 4500C CH3CH2CH2NO2 + H2O
Propane
Nitropropane
3. SULPHONATION OF ALKANE H2SO4:
It is a type of substitution reaction where H atom of alkane is substituted with –SO3H
Temperature: 5000C Sulphonation is possible with C>6.
C6H14 +
HO-SO3H 5000C C6H13SO3H + H2O
Hexane
n-Hexanesulphonic acid
Q. Give mechanism of sulphonation of alkanes?
Ans. Mechanism: free radical substitution
(a) Chain initiation (requires energy- endothermic reaction)
15
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
HO-SO3H 675 HO. +.SO3H
(b) chain propagation steps: (release energy- exothermic reaction)
R-H +HO.
R. + H2O
(c)Chain termination step.
.
R +.SO3H
R-SO3H alkane sulphonic acid
DECOMPOSITION REACTIONS:
4A. THERMAL DECOMPOSITION OR PYROLYSIS
A decomposition reaction in which higher alkanes on heating to higher temperature decompose into lower
alkanes,
alkenes
etc.
called
pyrolysis
4B. CATALYTIC DECOMPOSITION OR CRACKING:
A decomposition reaction in presence of catalyst
Ni, Pt etc higher alkanes are decomposed
into lower alkanes, alkenes etc at high temperature.
It is used in the preparation of petrol (gasoline), petrol gas, oil gas etc.
5. ISOMERISATION:
Process of conversion of one isomer of a compound to another isomer is known as
isomerisation.
Reaction takes place in the presence of lewis acid like AlCl3 and at temperature of 473K
and pressure of 35atm.
CH3
CH3CH2CH2CH3
Anhd AlCl3 / HCl CH3-CH-CH3
n-Butane
2-Methyl Propane
or
Isomerisation is widely used to increase the octane rating of gasoline.
OCTANE RATING:
Gasolines are rated on a scale known as octane rating, which is based on the way they
burn in an engine. The higher the octane rating, the greater the percentage of
complex-structured hydrocarbons that are present in the mixture, the more
uniformly the gasoline burns, and the less knocking there is in the automobile
engine. Thus, a gasoline rated 92 octane will burn more smoothly than one rated 87
octane.
Knocking: objectionable metallic sound produced during the working of internal
combustion engine.
Knocking order :
Straight chain paraffins > branched chain paraffins > Olefins> Aromatic hydrocarbons.
Knocking may be prevented by using tetraethyl lead (TEL) (C2H5)4Pb in small proportion
0.5%. Such petrol is known as leaded petrol or ethyl petrol.
16
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
6A. AROMATIZATION OR CATALYTIC REFORMING:
The conversion of aliphatic compound into aromatic compound in the presence of
catalyst is known as aromatisation. E.g.
6B. REFORMING: The conversion of aliphatic compound into aromatic compound in the
presence of H as hydro forming and in the presence of platinum is known as
Platforming.
Cracking, isomerisation and aromatisation are widely used to increase increase the
octane rating of gasoline.
7. OXIDATION REACTIONS:
7A. COMPLETE COMBUSTION REACTIONS: (Exothermic reactions)
Complete combustion:
CH4+ 2O2 CO2+ H2O +heat (212KCal/mol)+light
C2H6+ 7O2 4CO2+ 6H2O +heat (745KCal/mol)+light
2C4H10+ 13O2 8CO2+ 10H2O +heat +light
General reaction:
CnH2n+2 + (3n+1) O2 nCO2+ (n+1) H2O +heat + light
2
7B. INCOMPLETE COMBUSTION OR PARTIAL COMBUSTION:
2CH4+ 3O2 2CO+ 4H2O +lesser heat +light
2C4H10+ 9O2 8CO+ 10H2O +heat +light
It burns with a black soot.
7C.CONTROLLED OXIDATION:
Methane to methanol
(a) CH4+ O2
Cu tube
CH3OH
9: 1
100atm, 2000C
Methane to methanal
(b) CH4+ O2 Mo2O3 , 2750C HCHO + H2O
METHANE
METHANAL
Methane to methanoic acid
(c) 2CH4+ 3O2 Ag2O HCOOH + 2H2O
Methane
methanoic acid
Action of steam:
(d) CH4 + H2O (stm) CO + 3H2
Q. Complete the reactions:
(i) CH3CH3 + HNO3 vapor phase
(ii) CH4 +O2 Cu tube
(iii) C2H5COONa + NaOH CaO
Ans(1) nitroethane (2) CH3OH (3) C2H6 + Na2CO3
Q. Write structures of different chain isomers of alkanes corresponding to the molecular
formula C6H14. Also write their IUPAC names?
Q. Write chemical equations for combustion reaction of the following
hydrocarbons: (i) Butane (II) HEXANE?
Q. In the alkane H3C – CH2 – C(CH3)2 – CH2 – CH(CH3)2, identify 1°,2°,3° carbon atoms
and give the number of H atoms bonded to each one of these?
Q. What effect does branching of an alkane chain has on its boiling point?
Ans. branching decreases bp.
17
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
Q. Why is Wurtz reaction not preferred for the preparation of alkanes containing odd
number of carbon atoms? Illustrate your answer by taking one example?
Q. what is difference between conformers and isomers?
Ans. isomers cannot be changed into one another while conformations can be changed
into one another.
Q.bp of 2,3-dimethylbutane is greater than 2,2-dimethylnutane? T/F
Q. staggered conformations of ethane are mopre stable than eclipsed? T/F
Q. Cyclohexane mainly occurs in boat conformaitions ?T/F : chair conformations
Q. sodiumpropanate on heating with soda lime gives ethane/ propane? Ans ethane
Q. how do you account for the formation of ethane during chlorination of methane?
Ans. CH4+ Cl2 SUNLIGHT (hν) 673K CH3Cl + HCl
(a) Chain initiation (requires energy- endothermic reaction)
Cl-Cl hv or 2500C homolytic fission 2Cl. radical formation
(b) chain propagation steps: (release energy- exothermic reaction)
Cl. + H- CH3 HCl + .CH3
.CH3 + Cl-Cl CH3-Cl + .Cl
(c)Chain termination step.
.Cl + .Cl Cl-Cl and
.CH3+ .Cl CH3Cl and .CH3+ .CH3 CH3-CH3
Ease of substitution at various carbon atoms is of the order:
Tertiary > secondary > primary.
ALKENE:
Gen formula: CnH2n Hybridisation of C is sp2 Also known as olefins i.e. oil forming.
Bond angle is almost 121.70 , C=C 1sigma and 1pi bond
σ bond is strong than dissociation energy 95Kcal i.e. strong
bond is weak dissociation energy is 68Kcal i.e. weak
Therefore C-C in ethene are stronger than C-C in ethane because bond length in ethene
is less (1.34A0) than the C-C distance in ethane (1.54A0) Smaller is the bond length,
more is the dissociation energy. Ethylene (ethene): It is used as a general anaesthetic
and for making mustard gas. Isobutene is used for production of iso-octane , a high
octane fuel used for cars and aeroplanes.
HYBRIDISATION IN ETHENE
Molecular formula = C2H4
Empirical formula = CH2
Molecular mass C2H4= 28
Empirical mass CH2= 14
Homologous series = alkene CnH2n
HYBRIDISATION OF ETHENE: sp2
1(2s)+ 2(2px, 2py, ) orbital’s (sp2 hybridisation) 3 (sp2 hybrid orbital’s) of 1200
12
(ground state E.configuration):1s22s2px12py12pz0
6C
12
(excited state electronic configuration) 1s22s1 2px12py12pz1
6C*
alignment forming trigonal planar arrangement .
Each orbital has 1/3 (33%) s character and
2/3 (67% ) p character. On un-hybridised orbital
2pz is oriented in a plane at right angle to the
Plane containing three hybridised orbital’s.
one e- from s orbital is moved from 2s orbital to
2p orbital which is in higher energy state. The input
energy for exciting an electron is available from
lattice energy of formation, released when bond are formed. Initially energy has to give
so as to start the reaction. In the excited state one 2s orbital and two (2px, 2py) orbitals
of C* intermix to form 3 hybrid orbitals of sp2 hybridisation. These three sp2 hybridised
18
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
orbitals aligned themselves in such a way that bond angle must be 1200 so as to have
minimum energy and maximum stability. Orbital structure is trigonal planar.
Note: 01 orbital of 2pz remains unhybridized.
When molecule of ethene is formed, three sp2 hybridised orbitals interacts with other sp2
hybrid orbital of ethene forming 1 sigma overlapping between each C of Ethene. 2 each
sp2 hybrid orbitals forms 4 sigma bond by overlapping with 1s1 orbital of 4 H atoms in
end to end manner. One 2pz orbital of one C atom overlap sidewise forming pi bond with
other C atom of Ethene. Therefore Bonds between C-C atom is double in nature forming
1 sigma and 1 pi (π). Others 4 C-H sigma (σ) bonds
C=C bond length = 134pm, C- H bond length = 108pm,
H-C-H bond angle 117.60, H-C-C bond angle = 1210 .
above orbital structure clear all the overlapping:
PHYSICAL PROPERTIES OF ALKENES:
a. Physical state: The first three members are gases, the next fourteen are liquids and
the higher ones are solids. C1 to C4 are colourless and odourless gases except ethylene
which has pleasant smell. C5 to C18 are liquids and C> C18 are solids.
Reason: Hydrocarbons are non- polar because of the covalent nature of C-C and C-H
bonds. Thus, they have weak van der Waals forces of attraction.
Q. Why branched chain alkene is having lower bp?
b. Boiling points: The boiling points of alkenes increase with the increase in molecular
mass or no. of Carbon atoms.
Boiling points of branched chain alkenes are lower than the corresponding straight chain
alkenes. This is because with branching the molecules become more compact and hence
the surface area decreases. Vanderwaal forces surface area
Vander Waals forces increases with increase in molecular mass or increase in surface
area.
Note: Cis alkene bp will be high due to more polarity and trans alkene is less
polar due to low polarity.
Trans alkene Mp will be high due to more symmetrical structure than cis alkene
which is less symmetrical structure. Cis trans isomer is discussed in detail.
c. Solubility: Alkenes are soluble in non- polar solvents like benzene CCl4 and toluene
and are insoluble in polar solvents like water.
Alkene are generally non polar but certain alkene are weakly polar due to unsymmetry
geometry. e.g. propene and 1-butene have dipole moment of 0.35D.
ISOMERSISM: same molecular formula but different arrangement of atoms.
A. STRUCTURAL ISOMERISM:
(I) CHAIN ISOMERISM:
CH3-CH2-CH=CH2
CH3-C=CH2
But-1-ene
2-Methylpropene
CH3
(2) POSITION ISOMERISM:
The isomerism differs in the position of the double bonds. E.g.
Two linear positional isomers of molecular formula C5H10
(1)
(2)
, pent-1-ene,
bpt 30oC,
, pent-2-ene, cis bpt 37oC, trans bpt 36oC,
19
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
Q.Draw different structural isomers of alkenes corresponding to molecular formula C5H10.
B. GEOMETRICAL ISOMERS OF ALKENES:
Isomerism due to the difference in spatial arrangements of groups about the doubly
bonded carbon atoms is known as geometrical isomerism.
Geometrical isomerism in alkenes is due to restricted rotation around carbon-carbon
double bond. Example: Geometrical isomers C4H8
Trans isomer is non- polar and has zero dipole moment where as cis isomer is polar. cis
isomer has higher boiling points than trans isomer because it is polar.
In case of solids, cis isomer has a lower melting point than trans isomer. This is because
trans isomer is symmetrical and fits well into the crystal lattice.
Cis isomer is having less symmetry, symmetry leads to stability and thus high
Mpt.
Due to poor symmetry cis molecules do not fit into crystal lattice so well.
Therefore trans isomer has higher mp as they are more symmetrical. e.g.
H3C-C-H
H3C-C-H
║
║
H3C-C-H
HC-C-CH3
Cis- But-2-ene
Trans - But-2-ene
Dipole moment = μ > 0
Dipole moment = μ = 0
Bpt 40C (high)
Bpt 10 C (low)
0
Mpt -139 C (low)
Mpt -1060C (high)
H-C-COOH
H-C-COOH
H3C-C-COOH
║
║
║
H-C-COOH
HOOC-C-H
H-C-COOH
Maleic acid
Fumaric acid
Citraconic acid
(cis- isomer)
(trans- isomer)
(cis-isomer)
H-C-Cl
H-C-Cl
║
║
H-C-Cl
lC-C-H
Cis-1,2-Dichloroethylene
Trans -1,2-Dichloroethylene
H3C-C-COOH
║
HOOC-C-H
Mesaconic acid
(trans- isomer)
H3C-C-H
H3C-C-H
║
║
H3C-C-H
HC-C-CH3
Cis- But-2-ene
Trans - But-2-ene
Q. What is geometrical isomerism and what is its cause?
Q. What are the necessary conditions for the geometrical isomerisation?
Q. Cis alkenes show higher boiling point as compared to trans-isomer. Why? Hint polar
20
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
HOW TO DISTINGUISH BETWEEN CIS AND TRANS:
ON THE BASIS OF PHYSICAL PROPERTY:
1. DIPOLE MOMENT: 2 MELTING POINT
3. BOILING POINT
(a) Lesser dipole moment of trans isomers (symmetrical alkene) while cis has non zero
dipole moment or more than the trans isomer.
or otherwise Cis isomer is having higher D moment than Trans.
H-C-Cl
H-C-Cl
║
║
H-C-Cl
Cl-C-H
Cis-1,2-Dichloroethylene
Trans -1,2-Dichloroethylene
Dipole moment = μ = 1.91D
Dipole moment = μ = 0
H3C-C-H
H3C-C-H
║
║
H3C-C-H
HC-C-CH3
Cis- But-2-ene
Trans - But-2-ene
Dipole moment = μ > 0 Dipole moment = μ = 0
Dipole moment is high means Polarity of cis isomer > trans isomer.
Boiling point: cis isomer has higher due to high polarity, high density and higher
Refractive index than the corresponding trans isomer (Auwers-skita rue).
H3C-C-H
H3C-C-H
║
║
H3C-C-H
HC-C-CH3
Cis- But-2-ene
Trans - But-2-ene
Dipole moment = μ > 0
Bp 40C (high)
Mp -1390C (low)
Dipole moment = μ = 0
H-C-Cl
║
H-C-Cl
Cis-1,2-Dichloroethylene
Dipole moment = μ = 1.91D
Bp 600C (high)
MP -800C (low)
Bp 10 C (low)
Mp -1060C (high)
H-C-Cl
║
lC-C-H
Trans -1,2-Dichloroethylene
Dipole moment = μ = 0
Bp 480 C (low)
MP -500C (high)
The trans isomer has higher mp than cis- isomer due to symmetrical nature and
more close packing of the trans-isomer.
Note: Mpt relates to symmetry which is more in case of trans
While Bpt relates to polarity which relates to dipole moment.
Q. Draw cis and trans isomer and write IUPAC naming of :
(a) CHCl=CHCl
(b) C2H5C(CH3)=C(CH3)C2H5
Naming Hint: 1,2-Dichloroethene
3,4-Dimethylhex-3ene
Q. Which of the following compounds will show cis-trans isomerism?
(i) (CH3)2C=CH–C2H5 (ii) CH2=CBr2 (iii)C6H5CH=CH–CH3 (iv)CH3CH=CClCH3
Solution (iii) and (iv). In structures (i) and (ii), two identical groups are attached to one
of the doubly bonded carbon atom.
Q.Draw the geometrical isomer of hex-2-ene? Which isomer has higher bp and why?
Hint: Cis -Hex-2-ene because of more polar.
21
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
CH3
Q. which of the following will show geometrical isomerism?
(a) H2C=CBr2
(b) (CH3)2C=CHCH3 (c) C6H5CH=CHCH3 (d) CH3CH-C=CHCH2CH3
(e) H3C-CH=CCl(CH3)
Q. Which will exhibit geometrical isomerism?
(a) 2-Methyl but-2-en-1-ol (b) 3-Methyl but-2-en-1ol
Q. Cis-1,2-dichloroethene has less/more dipole moment than trans-1,2-dichloro ethne
Q. For the following compounds, write structural formulas and IUPAC names for all
possible isomers having the number of double or triple bond as indicated: C4H8 (one
double bond).
Q. Draw the cis and trans structures of hex-2-ene. Which isomer will have higher b.p.
and why? Cis -Hex-2-ene because of more polar
METHODS OF PREPARATION OF ALKENES:
1. DEHYDRATION OF ALCOHOL
(Removal of water by H2SO4, dehydrating agent)
Alcohol + conc H2SO4 alkene + H2O
RCH2CH2OH conc H2SO4, 443K
RCH=CH2 +H2O
CH3CH2OH conc H2SO4, 443K
CH2=CH2 + H2O
Dehydration order for alcohols 30 > 20 >10
2. DEHYDROHALOGENATION : (1,2 ELIMINATION OR β ELIMINATION)
It involves the treatment of an alkyl halide, with concentrated alcoholic solution of a
strong base like NaOH/ KOH to form alkene.
22
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
RCH2-CH2-X + KOH (alc) Δ RCH=CH2 +KX + H2O
β
CH3-CH2-CH2Br + KOH (alc) Δ CH3-CH=CH2 +KBr + H2O
(1-Bromopropane)
Ease of dehydrohalogenation in terms of alkyl halides : 30 > 20> 10
Ease of dehydrohalogenation in terms of halides: RI>RBr>RCl
alkyl iodide > alkyl bromide > alkyl chloride
V Imp.
Saytzeff rule: more substituted is the preferred product.
It states that “In dehydrohalogenation reactions, the preferred product is that alkene
which has lesser no. of H atom on doubly bonded Carbon atoms or contains greater
number of alkyl groups attached to the doubly bonded carbon atoms”.
CH3-CH2-CH2-CH2Br + KOH (alc) Δ CH3-CH=CH-CH3 + CH3-CH2-CH=CH2 + KBr + H2O
1-bromo Butane
2-Butene
1-Butene
3. DEHALOGENATION OF VICINAL DIHALIDES WITH ZN DUST IN ALCOHOL.
CH2Br
+
Zn
CH2=CH2 + ZnBr2
CH2Br
1,2-Dibromoethane
Ethene
4. KOLBE’S ELECTROLYTIC METHOD:
Electrolysis of aqueous solution of Na or K salt of saturated carboxylic acids.
CH2COONa
+ 2H2O electrolysis CH2=CH2 + 2CO2 + 2NaOH + H2
At anode
At cathode
CH2COONa (Sodium succinate)
Ethene
Q. How are alkenes prepared by Kolbe's Electrolytic process?
5. PARTIAL HYDROGENATION OF ALKYNE:
RCCH + H2 Ni RCH=CH2
CHCH + H2 Ni 523K CH2=CH2 ethene (hydrogenation reactions)
Using Ni catalyst or Lindlar catalyst (palladised charcoal deactivated with poison like S
compounds or quinoline). This method gives cis-alkene.
Lindlar catalyst
RCH=CH2 Pd/BaSO4/C
RCCH + H2 Ni RCH=CH2
S
However if Na/ liq NH3 is used as reducing agent , trans alkene is formed
known as birch reduction.
Q. What is Lindlar's catalyst? What is its use?
Ans. Lindlar catalyst (Pd palladised
charcoal (C) deactivated with poison like S
compounds or quinoline). It is used for hydrogenation of alkyne to alkene (cis-alkene).
LINDLAR CATALYST:
CH3-CC-CH3 + H2 Pd/BaSO4/C
S
CH3 CH3
C=C
cis- But-2-ene
H H
Q. What is Birch reduction ? What is its use?
23
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
BIRCH REDUCTION
CH3-CC-CH3 + H2 Na/NH3
CH3 H
C =C
trans- But-2-ene
H CH3
Q. What is the order of stability of alkene towards hydrogenation reactions?
Ans. Relatives stability of alkene can be compared by their enthalpy of hydrogenation
when alkene reacts with H2 in presence of Ni catalyst?
Ans. greater is the number of alkyl groups attached to the double bonded carbon atoms,
the more stable is the alkene.
Q. Alkyne on reduction with sodium in liquid ammonia form trans alkene? Write reaction
will it shows geometrical isomerism? Hint Birch reduction, yes.
CHEMICAL PROPERTIES OF ALKENE:
C=C bond in alkene is having 1 σ sigma and 1 bond. Due to presence of bond ,
alkene are very much reactive. Since bond is a weak bond and held less firmly with the
double bonded C as compared to σ bond. therefore in a reaction when electron is needed
electrons are easily available for a reaction.
C=C bond act as source of electrons and thus electron loving reagent which are
deficient in electrons are most likely to attack at the site of electrons of the double
bond which are electron in nature.
Further addition of elctrophilic reagents relieves the strain of the C=C double bond(1200)
by converting it into C-C (109.50). Electrophilic addition reactions are most common are
the most common reactions of C=C bond.
Example: Unsymmetrical alkene addition
(1-Bromopropane)
CH3-CH=CH2
+ H-Br (presence of peroxides) CH3-CH2-CH2Br
n- CH2=CH2
Δ, 1000atm ,organic peroxides -(CH2-CH2-)n
Ethene
Polyethene
Q. Why alkenes undergo electrophilic addition and not electrophilic substitution reaction?
SUBSTITUTION REACTIONS:
Ethylene does not undergoes substitution reactions. However, alkene having alkyl group
undergoes halogenations at higher temperature to form substituted products.
Mechanism :free radical substitution reactions:
CH3CH=CH2 + Cl2 500-6000C gas phase CH2=CH-CH2Cl + HCl
Propene
allyl chloride
CHEMICAL PROPERTIES OF ALKENES:
24
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
(A) ADDITION REACTIONS:
The reactions which involve the combination of two or more molecule (same or different
substances) to form an addition product (adduct) without any loss of any species from
the reactants are known as addition reactions. e.g.
CH3-CH=CH2
+ H-Br (presence of peroxides) CH3-CH2-CH2Br
n- CH2=CH2
Δ, 1000atm ,organic peroxides -(CH2-CH2-)n
Ethene
Polyethene
Reaction mechanism: Free Radical addition reactions:
OTHER EXAMPLES OF ADDITION REACTIONS:
I. ADDITION OF HYDROGEN (H2) (REDUCTION REACTIONS)
SABATIER AND SENDREN’S REACTION: (ALREADY DISCUSSED IN ALKANE)
Catalytic addition reaction: catalytic hydrogenation reaction.
It involves catalytic (Ni or platinum) hydrogenation of unsaturated hydrocarbons like
alkene and alkyne to alkane. Methane can’t be obtained by this method.
It is a type of addition reaction.
CH2=CH2 + H2
Ethene
Ni 2000C-300C or 473K CH3-CH3 +heat
Pt
Ethane
CHCH + 2H2
Ethyne
Ni 2000C-3000C or 473K CH3-CH3 + heat
Pt
Ethane
2. ADDITION OF HALOGEN (Br2, I2) in presence if inert solvent CCl4
25
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
Reddish brown colour of Br2 is discharged due to formation of colourless
dibromide.
NOTE: This reaction is used as a quick diagnostic test for the presence of unsatutration
i.e. >C=C<. Reddish brown colour of Br2 is discharged due to formation of colourless
dibromide.
Test for Unsaturation: There are two tests to know whether a compound is
unsaturated or not:
a. Bromine water test: When an unsaturated compound is reacted with bromine
solution in carbon tetrachloride, the reddish orange colour of bromine solution in carbon
tetrachloride is discharged.
b. KMnO4 TEST (oxidation reactions).
3. ADDITION OF HALOGEN ACID HX (HCl>HBr>HI)
CH2=CH2
+
HX
CH3-CH2-X
Order of reactivity of halogen acid: HI > HBr> HCl > HF
V. IMP.
PEROXIDE EFFECT OR KHARASCH EFFECT.
The addition of HBr (but no any other halogen acid) to unsymmetrical alkenes in the
presence of organic peroxide such as benzoyl peroxicle, (C6H5CO)2O2 or (B2H6) takes
place contrary to the Markovnikov's rule, -ve part of addendum goes to that C atom of
the double bond which is having larger no. of H atoms. This is known as Peroxide effect
or Kharasch effect.
CH3-CH=CH2
+ H-Br (presence of peroxides)
Unsymmetrical alkene
CH3-CH2-CH2Br (1-Bromopropane)
Type of reaction: Free radical mechanism:
Stability order: 30>20>10 (same as free radical).
Note: HF, HCl, HI do not exhibit peroxide effect and addition takes place according to
Markovnikov’s rule even in the absence of peroxides.
Q. Give mechanism of Kharash effect.
Ans. Mechanism of kharash effect : free radical addition reaction
(a) FORMATION OF FREE RADICALS:
(C6H5CO)2O2 HOMOLYSIS 2C6H5. +2CO2
Benzoyl peroxide
free radical
(b) C6H5. + H-Br
homolysis C6H6 + Br.
26
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
(c) Br. +CH3-CH=CH2 CH3.CH-CH2Br 20 free radical
CH3CH-CH2.
10 free radical
Br
Stability order of free radical: 30>20>10
Therefore CH3.CH-CH2Br 20 free radical is the more stable product.
(d) CH3.CH-CH2Br + HBr CH3CH2CH2Br
20 free radical
1-Bromopropane
V. IMP.
MARKOVNIKOV’S RULE: It states that when addition across an unsymmetrical alkene
takes place, +ve part of addendum (molecule to be added goes to that carbon atom of
the double bond which holds the greater no. of H atoms). It takes place in the presence
of mercuric acetate Hg(CH3COO)2 or B2H6
Br
CH3-CH=CH2 + HBr
MARKONIKON RULE
CH3-CH-CH3
Propene
2 Bromo propane
Type of reaction: Electrophyllic addition reactions: Stability order: 30>20>10
This is due to formation of carbocation, carbocation stability order is 30>20>10
Order of reactivity of halogen acid: HI > HBr> HCl > HF
Q. Explain and Justify Markownikoff's rule.
Q. Write IUPAC names of the products obtained by addition reactions of HBr to hex-1ene (i) in the absence of peroxide and (ii) in the presence of peroxide.
4. POLYMERISATION REACTIONS:
Large no. of simple molecules combine to form a bigger molecules of higher molecular
mass called polymer. Small molecule is called monomer.
e.g. Polymerisation of alkene in presence of organic peroxides like benzoyl peroxides,
Zieglar Natta catalyst. monomer is smallest repeating unit.
(a) High density polythene (HDPE) or polyethylene
Monomer: Ethene (Ethylene).
Catalyst: Zeigler Natta catalyst : (C2H5)3Al+ TiCl4
n- CH2=CH2
Δ, 1000atm ,
-(CH2-CH2-)n Polyethene
Ethene
organic peroxides
Uses: It is used for manufacturing buckets, dustbins, bottles, toys, pipes, etc
CH3
(b) Polypropylene
n CH2=CH-CH3
polymerisation -(CH-CH2-)n
Propylene
polypropylene (PP)
USES: milk crates, plastic buckets, seat covers, carpet fibres, ropes, heat shrinkable
wraps.
Cl
n CH2=CH-Cl
Polymerisation -(CH2-CH-)n
Chloroethene (vinyl chloride)
Polyvinylchloride (PVC)
PVC pipes, electric fitting, rain coats etc.
27
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
(c) POLY TETRA FLUOROETHENE (TEFLON):
monomer: CF2=CF2 1,2 difluoroethylene
Uses: It is used in making oil seals and gaskets and also used for non – stick surface
coated utensils.
5. ADDITION OF HYPOHALOUS ACID (HOCl, HOBr, HOI):
CH2=CH2
+
HOCl
H2O/ Cl2 CH2-CH2
Ethylene
hypochlorous
OH Cl
2-Chloro Ethanol (Ethylene chlorohydrin)
6. ADDITION OF COLD CONC. SULPHURIC ACID:
CH2=CH2
+
HSO3H
CH2-CH2
Ethylene
alkyl hydrogen sulhphate
H
SO3H
Ethylene hydrogensulphate
7. ADDITION OF HYDRACID HCN (Nucleophilic addition reactions)
CH2=CH2 + HCN CH3-CH2-CN
Propanenitrile
8. ADDITION OF WATER (H2O):
CH2=CH2
+
HOH
CH2-CH
Ethylene
water
OH H
Ethanol (Ethyl alcohol)
9. CATALYTIC ADDITION OF OXYGEN:
CH2=CH2
+ ½ O2
Ag
CH2-CH2
Ethylene
525-675k
O Ethylene oxide
10. ADDITION OF OZONE:
V.IMP
OZONOLYSIS: O3 is added to an alkene molecule to form ozonide which in turn can
hydrolysed to yield smaller carbonyl (ALDEHYDE / KETONES) fragments. The process of
addition of ozone to form an ozonide and then to hydrolyse the product is known as
Ozonolyis. Ozonolysis involves the replacement of olefinic bond, >C=C<, by two
carbonyl groups >C=O.
O
CH2=CH2
+
O3
CH2Cl2 CH2
CH2 + H2O/Zn ZnO + HCHO+ HCHO
195-200K
O
O
The addition of Zn, checks the formation of H2O2 which can otherwise oxidise the
products (aldehyde and ketone) to acids.
28
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
Q. Give ozonolysis reaction of ethene.
Q. How is structure of alkene elucidated by ozonolysis ?
ANS. Identification of the aldehyde and / or ketone formed during the ozonolysis will
give useful information about the structure of the original olefin or the position of double
bond.
DETECTION OF POSITION OF DOUBLE BOND:
H
H
CH3-C=O O=C-CH3
join the C atom of carbonyl group CH3-CH=CH-CH3
Ethanal
Ethanal
But-2-ene
CH3
CH3
CH3 CH3
CH3-C=O + O=C-CH3
join the C atom of carbonyl group CH3-CH=CH-CH3
Propanone (acetone)
2,3-Dimethyl but-2-ene
Q. An olefinic was treated with ozone and the resulting product on hydrolysis gave 2pentanone and acetaldehyde. What is the structure of the original olefin? Write
equations for the reactions involved?
Ans. by knowing the structure of 2-pentanone (CH3CH2CH2COCH3 ) and acetaldehyde
the structure of olefin may be written as below.
CH3
O
CH3CH2CH2C=CHCH3 +O3 CH3CH2CH2-C
CHCH3 + H2O/Zn A + B
- ZnO
CH3
O O
3-Methylhex-2-ene
A: CH3CH2CH2COCH3
B: CH3CHO
2-Pentanone
Acetaldehyde
Q. Write IUPAC names of the products obtained by the ozonolysis of the following
compounds :
29
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
(i) Pent-2-ene (ii) 3,4-Dimethylhept-3-ene (iii) 2-Ethylbut-1-ene (iv) 1-Phenylbut-1-ene
Ans(i) propanal, ethanal (ii) penan-2-one and butan-2-one (iii) pentan-3-one and
methanal (iv) propanal and benzaldehyde.
Q. An alkene ‘A’ on ozonolysis gives a mixture of ethanal and pentan-3- one. Write
structure and IUPAC name of ‘A’. A= 3-Ethylpentan-3-one.
Q. An alkene ‘A’ contains three C – C, eight C – H σ bonds and one C – C bond. ‘A’ on
ozonolysis gives two moles of an aldehyde of molar mass 44 u. Write IUPAC name of ‘A’.
Q. Propanal and pentan-3-one are the ozonolysis products of an alkene?
What is the structural formula of the alkene? Hint 3-Ethylhex-3-ene.
Q. Propanal and Butan-2-one are the ozonolysis products of an alkene?
What is the structural formula of the alkene? Hint 3-Methylhex-3-ene
Q. Ozonolyis of an alkene ‘X’ followed by decomposition with water and reducing agent
gives mixture of two products which are isomers of formula C3H6O? What is the
structural formula and IUPAC name of the alkene?
Ans. propanal CH3CHO propanone CH3COCH3
Alkene 2-Methylpent-2-ene
B. OXIDATION REACTIONS:
(i) OXIDATION BY ALKALINE KMnO4 SOLUTION (BAEYER’S REAGENT) TO FORM
ethylene glycol
CH2=CH2 + H2O + O
CH2-CH2
Cold KMnO4
V Important:
OH OH Ethylene glycol
BAEYER’S REAGENT TEST:
When an unsaturated compound is
reacted with cold, dilute aqueous
solution of potassium permanganate
(Baeyer’s reagent, the pink colour
of KMnO4 solution is discharged.
CH2=CH2
+ H2O + [O]
2HCOOH
Hot KMnO4
formic acid
2HCOOH
+ [O]
2CO2 + 2H2O
Formic acid
NOTE: Br2 water test and Baeyer’s test used for detecting the presence of double bond
while ozonolysis helps in locating the position of double bond.
(ii) HYDROBORATION OXIDATION:
CH3-CH2=CH2 +H2O DIBRORANE (B2H6) CH3-CH2-OH
Propene
Propanol
H2O is added opposite to Markonikov’s addition.
C. COMBUSTION REACTIONS:
(i) COMPLETE COMBUSTION REACTIONS: (Exothermic reactions)
Complete combustion:
C2H4+ 3O2 2CO2+ 2H2O +heat +light ΔH=-ve
C4H8+ 6O2 4CO2+ 4H2O +heat +light ΔH=-ve
General reaction:
CnH2n+ 3n O2 nCO2+ nH2O +heat +light ΔH=-ve
2
(ii) INCOMPLETE COMBUSTION OR PARTIAL COMBUSTION:
Incomplete combustion: C2H4+ 2O2 2CO+ 2H2O +Less heat +light ΔH=-ve
C4H8+ 4O2 4CO+ 4H2O +Less heat +light ΔH=-ve
It burns with black soot.
30
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
Q. Write chemical equations for combustion reaction of the following hydrocarbons: (i)
Pentene?
SUMMARY OF REACTIONS OF ETHENE ARE AS BELOW:
TYPES OF DIENES: COMPUNDS
COMULATIVE DIENES:
CH2=C=CH2
1,2-Propadiene
ALLENE
CONTAINING TWO DOUBLE BONDS ARE KNOWN AS DIENES
CH3-CH=C=CH-CH3
2,3-Pentadiene
SYM-DIMETHYL ALLENE
CH3
CH2=C-CH=CH2
2-METHYL-1,3-BUTADIENE(ISOPRENE)
CONJUGATED DIENES:
CH2=CH-CH=CH2
1, 3- BUTADIENE
ISOLATED DIENES:
CH2=CH-CH2-CH=CH2
CH2=CH-CH2-CH2-CH=CH2
1,4-PENTADIENE
1,5-HEXADIENE
Among the three classes of dienes, conjugated dienes are the most important
since the chemical properties of conjugated dienes are very important from
those of ordinary alkene.
Q. Which is more stable 1,4 or 1,3 Butadiene?
Ans 1,3-Butadiene is more stable as: highly symmetrical and highly substituted.
Q. give example of conjugated diene? 1, 3- BUTADIENE
Practice questions:
Q. Formula of Teflon? ans.–(CF2-CF2-)n
Q. C7H14 on ozonolysis gives ethanal and pentan-3-one. What is the structure and IUPAC
name of the alkene?hint: CH3-CH=C(C2H5)CH2CH3 3-Ethyl pent-2-ene
Q. Name two test to check the presence of double bond or triple bond in a molecule?
Ans. Bayers test (decolour pink color) , Br2 water test (decolourise brown color)
Q. CH2=CH2 + Alk KMnO4 ? ans ethylene glycol. CH2(OH)-CH2(OH)
Q. What is the cause of geometrical isomers of alkene?
31
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
Ans.Alkene containes 1 sigma bond and 1 pi bond, therefore rotation about C=C bond is
restricted which rise to geometrical isomers.
Q. Draw the geometrical isomers of but-2-ene and which of the two is nonpolar? ans.
trans is non polar.
Q. Write the structural formulae and possible isomers of C2H2Cl2? Cis and trans1,2Dichloroethylene.
Q. What is the order of stability of alkene? 30>20>10 stability.
Q. Bromine water is used to distinguish ethene and Ethyne T/F? false as both give
reactions.
Q. 1-butanol reacts with conc H2SO4 to give But-2-ene?
Q. Reaction of ethene with bromine electrophilic addition reaction.
Q. Addition of HBr to propene in presence of peroxides follows ___ rule. Anti mk rule.
Q. The alkaline potassium permanganate solution is ___ reagent. Baeyers reagent.
Q. Name the metal present in Grignard reagent? (Mg) RMgX
Q. Why are alkene called olefins?
Q. What happen (a) when ethyl alcohol is treated with conc H2SO4 443K?
(b) Ethyl bromide is treated with alcoholic KOH?
Q. How does ethylene reacts with (1) br2 (2) alkaline potassium permanganate (3)
ozone (4) water
Q. How will you distinguish between ethylene and ethane?
Q. Complete the reaction:
(i) CH3-CCH +H2 Pd, BaSO4, S?
(ii) CH3CH=CH-CH3 + O3
Q. Convert ethylene to acetylene?
Q. Convert acetic acid to methane?
Q.
Q.Write structures of different isomers corresponding to the 5th member of alkyne
series. Also write IUPAC names of all the isomers. What type of isomerism is exhibited
by different pairs of isomers?
Solution 5th member of alkyne has the molecular
formula C6H10. The possible isomers are:
(a) HC C– CH2 – CH2 – CH2 – CH3 Hex-1-yne
(b) CH3 –CC–CH2–CH2–CH3
Hex-2-yne
(c) CH3–CH2 -CC–CH2–CH3
Hex-3-yne
(d) 3-Methylpent-1-yne
(e) 4-Methylpent-1-yne
(f) 4-Methylpent-2-yne
(g) 3,3-Dimethylbut-1-yne
Position and chain isomerism shown by different pairs
32
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
ALKYNES:
Triple bond between two C C atoms.
CC consists of 1 sigma and 2 bonds.
General Formula: CnH2n-2
Hybridisation: sp
Strength: Stronger than C=C bond.
HYBRIDISATION IN AKYENE:
Molecular formula = C2H2
Empirical formula = CH
Molecular mass = 26
Empirical mass = 13
Common name = Acetylene
Homologous series = Alkynes
STRUCTURE OF ETHYNE: linear
BOND LENGTH: The C-H bond is 1.2Ao.
BOND ANGLE: HCC bond angle is 180o.
HYBRIDISATION OF ETHYNE: sp (linear)
1(2s) + 1(2px) orbitals (sp hybridisation) 2 (sp hybrid orbitals) of 1800 alignment
forming linear arrangement. Each orbital has 1/2 s (50%) character and ½ p(50%)
character. Two unhybridised orbital 2py, 2pz is oriented in a plane at right angle to the
plane containing three hybridised orbitals.
12
(ground state electronic configuration) 1s2 2s2px12py12pz0
6C
12
(excited state electronic configuration) 1s22s1 2px12py12pz1 one e- from s
6C*
orbital is moved from 2s orbital to 2p orbital which is in higher energy state.
The input energy for exciting an electron is available from lattice energy of
formation, released when bond are formed. Initially energy has to give so as to start the
reaction.
In the excited state one 2s orbital and one (2px ) orbitals of C* intermix to form 2 hybrid
orbitals of sp hybridisation. These two sp orbitals (sp) aligned themselves in such a way
that bond angle must be 1800 so as to have minimum energy and maximum stability.
Orbital structure is linear.
Note: 02 orbital of 2py and 2pz remains un-hybridized state.
When molecule of Ethyne is formed, sp hybridised orbitals interacts with other sp hybrid
orbital of ethyne forming 1sigma which overlap between C-C bond of ethyne. 1 sp2
hybrid orbital of each C of Ethyne forms sigma bond by overlap with 1s1 orbital of 2 H
atoms in end to end manner (σ).
Two 2py and 2pz orbital of C atom of ethyne overlap sidewise forming 2pi (π)bond with
other C atom of ethyne. Therefore bond between C-C is triple in nature 1 sigma (σ) and
2 pi (π). 2 C-H bonds forming sigma bond (σ).
CC bond length = 120pm, C- H bond length = 106pm, H-C-C-H bond angle 1800
Structures of Ethyne:
PHYSICAL PROPERTIES OF ALKYNES:
C1-C4 gases
C5-C12 liquids
C>C13 solids
33
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
Colourless, odourless gases, however common acetylene has garlic odour due to
presence of impurities like H2S, PH3 Phosphine gas etc.
Liquid acetylene is dangerously explosive, use of storage acetylene is prohibited by law.
Acetone is therefore stored and transported in acetone solution.
a. Solubility: Alkynes are weakly polar in nature. They are lighter than water and
immiscible with water but soluble in organic solvents like ethers R-O-R, carbon
tetrachloride CCl4 and benzene C6H6. Hence alkynes are sparingly soluble in water
but dissolves in organic solvents like acetone, alcohols etc.
b. Melting and boiling point: The melting and boiling points of the members of the
alkyne family are slightly higher as compared to those of corresponding alkanes
and alkenes. This is because alkynes have a linear structure and therefore their
molecules are more closely packed as compared to alkanes and alkenes. The
magnitude of attractive forces among them is higher and therefore, the melting and
boiling points are also higher.
The melting point, boiling point and density of alkynes increase with increase in molar
mass. This is because magnitude of van der Waals forces increases with increase in
molecular mass or increase in surface area.
ETHANE
ETHENE
ETHYNE
Mpt (K)
101
104
191
Bpt (K)
184.5
171
198
ISOMERISM IN ALKYNE:
A. STRUCTURAL ISOMERISM:
(I) CHAIN ISOMERISM:
Same molecular formula but different arrangement of carbon chains.
CH3-CH2-CHCH
CH3-CCH
But-1-yne
2-Methylprop-1-yne
CH3
(2) POSITION ISOMERISM:
Same molecular formula but differ in the position of the functional group, carboncarbon multiple bond.
Two linear positional isomers of molecular formula C5H8
(1) CH3-CH2-CH2-CCH
Pent-1-yne
(2) CH3-CH2-CC-CH3
Pent-2-yne
(3) FUNCTIONAL ISOMERISM:
Same molecular formula but different functional groups in the molecule
diene and alkyne
CH3-CC-CH3
CH3=CH-CH=CH2
Buta-1,3-diene
Butyne-2
(4) RING CHAIN ISOMERISM:
Due to difference in mode of linkage in C atoms and the isomers may have either open
chain or closed structure. e.g. C3H4 and cyclopropene.
Q. Why Alkyne do not shows geometrical isomerism?
Ans. Alkyne involves sp hybridisation , thus angle is 1200.
Shape is linear, thus do not show geometrical isomerism
34
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
METHODS OF PREPARATION of ALKYNES:
1. HYDROLYSIS OF CALCIUM CARBIDE (LAB METHOD)
By the action of water on calcium carbide (CaC2) placed in a flask
CaC2
+ 2H2O
C2H2
+
Ca(OH)2
Ethyne
:CC:2- + 2HOH
H-CC-H
+
2OHBase + acid
Conjugated acid +
Base
2. BY DEHYDROHALOGENATION OF VIC-DIHALIDES:
CH2=CH2 + Br2 CH2-CH2 + KOH(alc) CH2 +KOH (alc) CH
Br Br
CHBr
CH
Ethene
Ethylene bromide
Vinyl bromide
Acetylene
CH3
+ KOH (alc)
CHBr2
Ethylidene bromide
CH2
+KOH (alc)
CHBr
Vinyl bromide
CH
CH
Acetylene
3. BY DEHYDROHALOGENATION OF VIC-tetra HALIDES:
With active metals like Zn, Mg etc and CH3OH as solvent.
Br Br
Br Br
H-C-C-H +
Zn
H-C=C-H +
Zn
HC=C-H
-ZnBr2
-ZnBr2
Ethyne
Br Br
1,1,2,2-Tetrabromoethane
4. BY KOLBE ELECTROLYTIC METHOD:
CHCOONa
+ 2H2O electrolysis CH=CH + 2CO2
║
At anode
CHCOONa
Sodium fumarate
+
2NaOH + H2
At cathode
5. BY HEATING IODOFORM OR CHLOROFORM WITH SILVER POWDER OR ZINC :
CHCl3 + 6Ag + Cl3HC Δ heat HC≡CH + 6AgCl white ppt
6. BERTHOLOT SYNTHESIS:
2C+ H2 electric arc CHCH
35
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
V. Imp.
7. PREPARATION OF HIGHER ALKYNES FROM SODIUM ACETYLIDE:
CHCH
+ Na Liquid NH3 475K
HCC-Na
+
Acetylene
Sodium acetylide
HCC-Na +
BrCH3
Sodium Acetylide
1/2H2
H-CC-CH3 + NaBr
Propyne
HCC-H
+ 2Na liquid NH3 NaCC-Na + 2CH3Br CH3-CC-CH3
2-Butyne
NOTE: This reaction is used to convert lower alkyne to higher alkyne.
R-CC-H + 2Na
Lower alkyne
liquid NH3 R-CC-Na + 2CH3Br R-CC-CH3 + NaBr
Higher alkyne
CHEMICAL PROPERTIES OF ALKYNES:
Alkyne undergoes addition reaction
(a) Electrophilic addition reactions with electron deficient electrophile. E.g. addition of
Br2 ,Cl2, I2 etc.
(b) Nucleophilic addition reactions with electron rich nucleophiles.e.g. hydration of
alkyne, addition of HCN, NaHSO3, RMgX, Carboxylic acids and alcohols are examples of
nucleophilic additions.
Q. Why CC triple bond is less reactive than C=C toward electrophillic addition reactions?
Alkynes are more reactive than alkane and alkene due to the following reasons:
(i) Presence of electrons:
Alkynes are characterised by CC bonds (1σ, 2). Due to presence of electrons which
are loosely held in alkynes, electrophile (+ve ) charge species can attack on the electron
rich electrons hence undergo electrophilic addition reactions.
(ii) Hybridisation:
This is because in alkynes C atom is sp hybridised and thus has more s character of
carbon than that of sp2 hybridised carbon of alkene. Now since greater is the s character
of C atom, more strongly will be the attraction for electrons and hence lesser will be
the availability for reaction.
ACIDIC NATURE OF ALKYNES:
Q. Why alkynes are acidic?
Hydrogen atoms of ethyne attached to triply bonded carbon atom are acidic in
nature. Example: HC=CH
+ Na HC=C-Na+ + ½ H2
HC=C-Na+ + Na Na+ C-C-Na+ + ½ H2
s character in alkynes is maximum i.e.50%.
Therefore electrons constituting this bond are more strongly held by the Carbon nucleus
(sp hybridised C is having more electronegative).With the result the H present on such a
carbon atom (C-H) can be easily removed as a proton.
The acidic nature of differently hybridised C of C-H bonds are as follows:
RC-H
> R=CH2
> R-CH3
sp
sp2
sp3
Being most electronegative C in alkyne, the carbon of acetylene is best able to
accommodate the electron pair in the anion left after the proton is lost.
36
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
In other words, anion of acetylene is most stable and hence more acidic. While
anion of ethane is the least stable (sp3 carbon is the least electronegative) and hence
least acidic.
CHCH + :B-
HB + HCC:-
CH3-CH3 + :B-
HB + H3C-CH2:
(most stable)
-
(least stable)
Presence of acidic H atoms:
H atom or atoms attached to the triply bonded C atom can be easily removed by means
of a strong base and hence acetylene or 1-alkyne sre considered as weak acids.
R-CC-H
+
base
R-CC+
H-Base
Acidic alkyne
RELATIVE ACIDIC CHARACTER :
H-O-H R-OH > CHCH > CHCR > :NH3
> CH2=CH2
Water
alcohol
alkyne
alkyne ammonia alkene
> CH3-CH3
alkanes
RELATIVE BASIC CHARACTER (CONJUGATE BASE):
OH -OR < -CC-R < -NH2 <-CH=CH2 <-CH2-CH3
REACTION SUPPORTING ACIDIC CHARACTER:
CHCH
+ Na Liquid NH3 475K
HCC-Na
+
Acetylene
Sodium acetylide
HCC-Na +
BrCH3
Sodium Acetylide
1/2H2
H-CC-CH3 + NaBr
Propyne
CHCH +Cu2Cl2 + 2NH4OH
Ammonical cuprous chloride
Cu-CC-Cu + 2NH4Cl + 2H2O
copper acetylide (red ppt)
CHCH + 2AgNO3 + 2NH4OH
Ammonical silver nitrate
Ag-CC-Ag + 2NH4NO3 + 2H2O
silver acetylide (white ppt.)
The above reaction is used for detecting the presence of Acetylenic H atom. For
example butyne-1 have acetylenic H while 2-butyne do not. Acidic H atoms
gives red and white ppt.
Q. Which of the following is acidic? (a) 2-Butene (b) 2-Butyne (c) 1-Butyne (d) 1-Butene
Q. Propyne is less acidic than acetylene? True.
Q. When ______is passed through red hot iron tube at 873K, benzene is formed.
Ethyne.
Q. Alkynes are _____ acidic than alkene. (less/ more)
REACTIONS OF ALKYNE:
37
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
ADDITION REACTIONS:
1. ADDITION OF HYDROGEN:
SABATIER & SANDEREN’S REACTION: (Already discussed in Alkane / Alkene)
It involves catalytic (Ni or platinum) hydrogenation of unsaturated hydrocarbons like
alkene and alkyne to alkane. Methane can’t be obtained by this method.
CHCH + 2H2
Ethyne
Ni 2500C OR 5230K
CH3-CH3 + heat
Ethane
COMMON CATALSYT USED ARE:
(a) Lindlar catalyst: Palladium supported over barium sulphate partially poisoned by
quinoline (b) Brown catalyst: nickel boride
LINDLAR CATALYST:
CH3-CC-CH3 + H2 Pd/BaSO4/C
S
BIRCH REDUCTION:
CH3-CC-CH3
+ H2 Na/NH3
Used for producing trans-But-2-ene.
CH3 CH3
C=C
cis- But-2-ene
H H
CH3 H
C =C
trans- But-2-ene
H CH3
2a. ELECTROPHYLIC ADDITION OF HALOGENS: Cl2, Br2, I2
CHCH + Cl2
CHCl
║
CHCl
+ Cl2
CHCl2
CHCl2
38
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
Acetylene
Acetylenedichloride Acetylene tetrachloride (Westron)
1,2-Dichloroethene
1,1,2,2-tetrachloroethane
2b. ADDITION OF HALOGEN ACIDS:
Order of reactivity HI > HBr > HCl
CHCH + HCl
CH2
+ HCl
CH3
║
CHCl
CHCl2
Acetylene
vinyl chloride
Ethylidene chloride
2c. ADDITION OF HYPOHALOUS ACIDS:
CHCH + HOCl
Acetylene
CHOH + HOCl
║
CHCl
Vinyl chloride
CH(OH)2
CHCl2
-H2O CHO
CHCl2
Dichloroacetaldehyde
3. NUCLEOPHILIC ADDITION REACTIONS:
3a. ADDITION OF WATER HYDRATION:
HC≡CH (Acetylene) + H-OH
(1% HgSO4, 42% H2SO4 333K)
HgSO4 CH2=CH-OH CH3-CHO ethanal
H2SO4
3b. ADDITION OF HCN TO ALKYNE
HC≡CH (Acetylene) + HCN
Ba(CN)2 CH2=CH-CN (Vinyl cyanide or Acrylonitrile)
3c. ADDITION OF ACETIC ACID:
HC≡CH (Acetylene) + CH3COOH
HgSO4 CH2=CH-OCOCH3 + H2O CH3-CHO ethanal
H2SO4
+ C2H5-OH ethanol
3d. ADDITION OF ARSENIC TRICHLORIDE:
HC≡CH (Acetylene) + ClAsCl2
CHCl=CHAsCl2
Lewsite (poisonous gas)
Antidote: BAL (British Anti Lewsite)
4. OZONOLYIS OF ALKYNE :
O
CH=CH +
O3
CH--------CH
O--------O
+
H2O H2O2 + CHO
CHO
5. OXIDATION REACTION:
(a) Oxidation with alkaline potassium permanganate:
COOH
CHCH +
4[O]
Alkaline KMnO4
298K
COOH (Oxalic acid)
Ethane1,2- dioic acid
39
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
Cold KMnO4 at 298K CH part is oxidised to –COOH
CR part is oxidised to R–CO- group
(b) Oxidation with acidic KMnO4 or K2Cr2O7
CH=CH
+
4[O]
K2Cr2O7+ H2SO4
COOH
2CH3COOH
COOH
Ethane1,2- dioic acid
With acidic KMnO4 at high temperature:
CH part is oxidised to –COOH
CR part is oxidised to RCOOH.
6. COMBUSTION REACTIONS:
CnH2n-2 + (3n-1) O2 nCO2+ (2n-2) H2O + heat
2
2
2CHCH + 5O2 4CO2+ 2H2O
Used in welding purpose in oxyacetylene flame. It has a flame temperature of 30000C
7. ISOMERISATION REACTIONS:
CH3-CH2-CCH + alc KOH , heat
1-Butyne
CH3-CH=C-CH3
2-Butyne
8. POLYMERISATION REACTIONS:
(a) CYCLIC POLYMERISATION:
Alkynes when passed through a red hot iron tube, undergo cyclic polymerisation to from
aromatic hydrocarbons.
3CHCH red Hot Cu tube benzene or C6H6
V. Imp
9. FORMATION OF METALLIC DERIVATIVES:
CHCH +Na liquid NH3 HCC-Na+ 1/2H2
HCC-Na + BrCH3 H-CC-CH3 + NaBr
Sod. Acetylide
Propyne
HCC-H + 2Na liquid NH3 NaCC-Na + 2CH3Br CH3-CC-CH3
2-Butyne
CHCH +Cu2Cl2 + 2NH4OH
Cu-CC-Cu + 2NH4Cl + 2H2O
Ammonical cuprous chloride
copper acetylide (red ppt)
CHCH + 2AgNO3 + 2NH4OH
Ag-CC-Ag + 2NH4NO3 + 2H2O
Ammonical silver nitrate
Silver acetylide (white ppt.)
The above reaction is used for detetcting the presence of Acetylenic H atom. for
example butyne-1 have acetylenic H atom gives red and white ppt.
Q. Distinguish between butyne-2 and butyne-1?
Ans. Butyne-1 have acidic H atom which can give red and white ppt. with ammonical
cuprous chloride and ammonical silver nitrate silver nitrate solution, respectively while
butyne-2 does not give such ppt.
CH3-CH2-CHCH +Cu2Cl2 + 2NH4OH
butyne-1
Ammonical cuprous chloride
CH3-CH2-CH2-CCu + 2NH4Cl + 2H2O
copper butynlide (red ppt)
CH3-CHCH-CH3 +Cu2Cl2
no reaction
+
2NH4OH
40
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
Or
CH3-CH2-CHCH + AgNO3 + NH4OH
Butyne-1
Ammonical silver nitrate
CH3-CH2-CH2-CAg + NH4NO3+ 2H2O
Silver butylnide (White ppt)
CH3-CHCH-CH3 +AgNO3
no reaction
+
2NH4OH
10. OTHER REACTIONS OF ETHYNE:
(a) HCCH + N2 ELECTRIC ARC 2HCN
(b) 2HCCH + S CH-CH
║ ║
CH CH
S
(c) 2HCCH + NH3 CH-CH
║ ║
CH CH
+H2
N
H
INDUSTRIAL APPLICATIONS OF ETHYNE (ACETYLENES)
1. It is used in form of oxyacetylene flame for welding, cutting and cleaning iron & steel.
2. Acetylene is used for illumination purposes in hawker’s lamps and light house.
3. It is used for artificial ripening of fruits.
4. It is used for the production of several chemicals like acetaldehyde (CH3CHO), acetic
acid (CH3COOH), ethyl alcohol (C2H5OH), acetone (CH3COCH3), benzene etc.
5. It is used for the production of Westron (acetylene tetrachloride) and Westrosol
(trichloroethylene) , industrial solvents for rubber, fats and varnishes.
6. It is used in the preparation of vinyl chloride CH2=CH-Cl, vinyl acetate CH2=CHCOOCH3 and vinyl ethers CH2=CHOR used in the production of plastics aand artificial
rubber. E.g. Polyvinylchloride(PVC).
7. It is used in the preparation of acrylonitrile (CH2=CH-CN) used for the production of
synthetic rubber (Buna-N) and synthetic fibre (orlon).
8. It is used in the preparation of chloroprene and butadiene used in synthetic rubber
industry.
9. It is used for the production of Lewsite (Cl-CH=CHAsCl2) , a powerful poisonous gas.
SUMMARY OF REACTIONS OF ETHYNE:
41
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
DIFFERENCE IN ALKANE ALKENE AND ALKYNE HYDROCARBONS :
Hydrocarbons
Alkyne
Alkene
Alkane
Hybridisation
of
Sp
sp2
sp3
carbon to which H
atom is attached
Percentage s character
50%
66.6 %
25% (minimum)
of carbon
(maximum)
Electronegativity
of
Highest
Less than alkyne and
Lowest
carbon atom
more than alkane
Extent of attraction of
Highest
Less than alkyne and
Lowest
hydrogen atoms of C-H
more than alkane
bonds towards C
Ease of liberation of H
Highest
Less than alkyne and
Lowest
atoms as protons
more than alkane
Acidic character
Highest
Less than alkyne and
Lowest
more than alkane
Bond length C-C all
154pm
148pm
138pm
containing 2C atoms.
348
383
433
Bond strength KJ
Bond length C-H
111pm
110pm
108pm
Bond strength KJ
436
443
507
Multiplicity of bonds
C-C
C=C
CC
Q. Write structures of different isomers corresponding to the 5th member of alkyne
series. Also write IUPAC names of all the isomers. What type of isomerism is exhibited
by different pairs of isomers?
42
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
Solution 5th member of alkyne has the molecular formula C6H10. The possible isomers
are: (a) HC ≡ C – CH2 – CH2 – CH2 – CH3 Hex-1-yne
(b) CH3 – C ≡ C – CH2 – CH2 – CH3
Hex-2-yne
(c) CH3 – CH2 – C ≡ C – CH2– CH3
Hex-3-yne
(d) 3-Methylpent-1-yne
(e) 4-Methylpent-1-yne
(f) 4-Methylpent-2-yne
(g) 3,3-Dimethylbut-1-yne
Q. How will you convert ethanoic acid into benzene?
Hint: ethanoic acid sod salt of ethanoic acid CH4 CH3Cl by chlorination wurtz
reaction, ethane chlorination C2H5Cl + KOH elimination C2H4 Br2 CH2Br-CH2Br +
2KOH CHCH
Aromatisation C6H6
Q. Following compounds, write structural formulas and IUPAC names for all possible
isomers having the number of double or triple bond as indicated:(a)C5H8(one triple
bond) Ans. 1-Pentyne, Pent-2-yne, 3-Methylbut-1-yne.
Q. How would you distinguish propene from propyne?
Ans. alkyne are acidic while alkene do not.
Propyne gives reaction with ammonical silver nitrate solution (tollens reagent) and give
white ppt while propene do not.
CH3-CHCH + AgNO3 + NH4OH
CH3-CH2-CAg + NH4NO3+ 2H2O
Propyne
Ammonical silver nitrate Silver Propylnide (White ppt)
CH3-CH=CH2 + AgNO3 + NH4OH
no white ppt.
Q. How will you convert acetaldehyde from acetylene?
Hint: hydration of water in presence of HgSO4+ H2SO4.
Q. Which of the following hydrocarbon reacts with Na in liquid ammonia?
(a) Propyne (b) But-1ene (c) But-1-yne(d) But-2-ene
Q. What happens when water is dropped on calcium carbide? Ethyne is produced.
Q. Name the reagent : 1,2-Dibromoethane + X Acetylene ans . alc KOH.
Q. Write the chemical reaction for the combustion reaction of hexyne?
2C6H10 + 17O2 12CO2 + 10H2O
Q. 1,1,2,2-tetrachloroethane + Zn, alcohol + heat A iron tube 675K B
What is A and B? Hint A= Ethyne B= benzene
Q.Propyne + H2, Pd, BaSO4 quinoline A + O3, Zn/ H2O B + C
What is A , B ? ans A= CH3-CH=CH2 B= CH3CHO C= HCHO
Q. convert: Ethyne to methane: hint ethyne ethanal ethanoic acid soda lime CH4
Q. convert ethene to Ethyne? Hint : + Br2 + KOH alc
Q. Ethane to Ethyne: hint: ethane chloroethane KOH ethene + br2 + 2KOH alc.
Q. Ethyne to 2-butyne: + Na liq NH3 + 2CH3Br 2-butyne + 2NaBr.
Q. suggest a method to separate a mixture of ethane , ethene and Ethyne?
Ans. bubble through ammonical silver nitrate solution, white ppt will separate alkyne.
Treat white ppt with HCl alkyne can be separated. Balance mixture containing alkene
and alkane is passed through Br2, CCl4 then vic dihalides ppt will be formed which can be
separated by treating with Zn as ZnBr2 and alkene can be separate. Balance remains
only alkane.Thus alkane,alkene & alkyne are separated by this combination of methods.
Q.2- butyne on catalytic reduction with lindar catalyst gives: ---alkene ans. cis alkene
Q. electrolysis of potassium fumarate gives _________(alkane/ alkene/ alkyne)
Q. hydration of acetylene gives ---(ethanol/ ethanal).
Q. But-2yne cannot react with tollens reagent? T/F
Terminal alkynes are acidic in nature and undergo ionic reactions.
43
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
Q. Chloroform on heating with silver powder gives Ethyne/ ethene?
Q. Ethyne on ozonolysis gives ________ glycol/ glyoxal ()
Q. convert Ethyne to benzene? Hint Cyclic polymerisation reactions.
Q. complete the reactions:
(a) HC CH + alk KMnO4 hint Oxalic acid
(b) CH3C CH + hot KMnO4 hint ketone will be formed.
Q. Distinguish between Buty-2-yne and But-1-yne? But-1-yne gives white ppt with TR.
Q. Why cylco propane more reactive than propane? Bond angle in cyclo propane is 600
while 1090 in alkane therefore more repulsions, more strain , more energy least stable
and hence reactive.
Important topics in alkane , alkene and alkyne:
conformations: butane anti>skew>eclipsed >fully eclipsed
Ethane and propane: staggered are more stable than eclipsed.
Chair conformations are more stable than boat for cyclohexane.
Cis more polar than trans (more symmetrical) geometrical isomer
Lindlar catalyst: Pd palladised + (C) deactivated with S /quinoline) cis alkene
Birch reduction : trans alkene. Na/NH3
Bayers reagent : alkaline KMnO4
Markonikov rule, saytzeff rule, peroxide effect,
Ozonolysis of alkene, testing between alkane and alkene, alkene and alkyne.
Acidic character of alkyne.
Hybridisation of Ethane , Ethene, Ethyne.
AROMATIC COMPOUNDS:
These molecules contain a benzene ring based a 'special' cyclic C6 system, which is
an unsaturated ring (BUT not an alkene system) e.g.
44
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
or
or
Benzene C6H6,
Methylbenzene(Toluene) C6H5CH3 or Phenylethene('Styrene'),
2,3-Dichloromethylbenzene,
C6H5Cl,
chlorobenzene,
C6H4Cl2,
,
1,2-dichlorobenzene,
or
Phenol.
or 1,4-dichlorobenzene,
C7H6O3,
2-Chlorophenol
(o-chlorophenol)
3-Hydroxybenzoic acid
C7H8O,
C6H7NO,
3-Methylphenol
2-Aminophenol,
(m-methylphenol, meta-cresol)
C7H6O,
benzaldehyde,
,
or 1,3-dichlorobenzene,
or
2-Hydroxybenzoic acid
4-Aminophenol
2-Nitrophenol
, C7H6O2,
,
,
,
benenecarboxylic acid, benzoic acid
C6H5NO2,
Aniline
benzene diazonium chloride. nitrobenzene, benzenesulphonic acid,
C6H6SO3, C6H5SO3H, C6H5SO2OH, (or benzenesulphonic acid)
(1)
,
(2)
,
(3)
,
45
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
Methyl-2-nitrobenzene
3-aminobenzoic acid
4-hydroxybenzamide
ISOMERSISM:
POSITION ISOMERISM IN ETHYL BENZENE
C8H10 (1)
(2)
(3)
(4)
(1) Ethylbenzene, (2) 1,2-Dimethylbenzene, (3) 1,3-Dimethylbenzene, (4) 1,4-Dimethylbenzene
m pt -94oC,
m pt -25oC,
m pt -47 oC,
m pt 14oC,
o
o
o
b pt 136 C,
b pt 144 C ,
b pt 139 C ,
b pt 137oC,
all colourless liquids.
Ortho (Substituent at 1,2) Meta(Substituent at 1,3) Para(substituent at 1,4)
POSITION
ISOMERISM
CH3C6H4SO2OH (C7H8SO3)
IN
METHYL
BENZENE
SULPHONIC
ACID
IN
1)
,
(2)
and
(3)
2-Methylbenzenesulphonic acid 3 Methylbenzenesulphonic acid
4 Methylbenzenesulphonic acid
Ortho- methylbenzenesulphonic acid meta- methylbenzenesulphonic acid para methylbenzenesulphonic acid
POSITIONAL ISOMERS OF XYLENE
C8H10 or
,
1,2-Dimethylbenzene,
1,3-Dimethylbenzene
Ortho xylene
Meta xylene
POSITIONAL ISOMERS OF C9H12
1,4-Dimethylbenzene
Para-xylene
C9H12,
C9H12,
1-Ethyl-2-methylbenzene, 1-Ethyl-3-methylbenzene, 1-Ethyl-4-methylbenzene,
Q. Draw the structure of p-nitroaniline, o-Ethylanisole, 2,3-Dibromo-1-phenylpentane,
4-Ethyl-1-fluoro-2-nitrobenzene?Benzene, Methylbenzene (Toluene), Naphthalene, Biphenyl, 1,2Dimethylbenzene (o-Xylene)
BENZENE:
Molecular formula = C6H6
Molecular mass = 78
Percentage of carbon = 93.6%
Empirical formula = CH
Empirical formula mass = 13
Percentage of hydrogen = 6.4%
ORBITAL STRUCTURE OF BENZENE:
46
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
Kekule Structural formula of benzene : It is an aromatic hydrocarbon.
ALL bond angles 120o, symmetrical hexagonal ring,
planar molecule
HYBRIDISATION IN BENZENE:
This is the most accurate concept of the structure of benzene. The structure of
benzene molecule is
best described in terms of molecular orbital theory.
According to this theory, all the C-atoms in benzene are sp2-hybridized. Two sp2hybrid orbitals of each C-atom overlap with two sp2-hybrid orbital of two other C-atoms
to form sigma bonds. In this way there are six sigma bonds are formed between six Catoms which are 120o apart. Remaining six sp2-orbital of six C-atoms overlap with 1s
orbital of six H-atoms individually to form six sigma bonds. Since sigma bond results
from the overlap of above said planar orbital, all H and C atoms are in the same plane
and their generate a hexagonal ring of C-atoms.
The overlap of these 2pz-orbital result in the formation of a fully delocalized pi-bond,
which extends all over the six C-atoms of benzene nucleus. The molecular orbital
approach clearly indicates that these six electrons could be found anywhere in highly
delocalized manner. As a result of delocalization, a stronger -bond and a more stable
benzene molecule is obtained which undergo substitution reactions more frequently than
addition reactions.
BOND LENGTH ANALYSIS IN BENZENE
C-C length in alkane = 1.54oA, C=C length in alkene = 1.34oA
But in benzene, C-C length = 1.397oA.
This shows that in benzene single and double bonds have quite extraordinary character
as they do not resemble to alkane and alkene in bond lengths. That is why benzene
shows a behaviour of saturated as well as an unsaturated hydrocarbon simultaneously.
MODERN REPRESENTATION OF BENZENE
With the help of molecular orbital behaviour we conclude that benzene has a regular
hexagonal structure with an inscribed circle. A hexagon with alternate double and single
bonds therefore benzene undergoes addition reactions but it does not discharge bayers
reagent which is pink in colour which can be discharged by unsaturated hydrocarbons
while not by saturated hydrocarbons.
C6H6
+
H2
Ni catalyst 2000C Cyclohexane or C6H12
Benzene undergoes electrophilic substitution reactions like nitration, sulphonation,
acylation which are not shown by unsaturated hydrocarbons but shown by saturated
hydrocarbons etc .
47
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
Conclusion: benzene give reactions of both saturated hydrocarbons and unsaturated
hydrocarbons because of presence of alternate single and double bond.
Q. How does the result support Kekulé structure for benzene?
RESONANCE IN BENZENE:
RESONANCE:(Heisenberg)
It is the phenomenon in which two or more structures can be written for the true
structure of a molecule, but none of them is said to represent it uniquely.
The true structure of the molecule is said to be a resonance hybrid of the various
possible alternative structures which themselves are known as resonating structures
or canonical structures. Kekule structures 1, 2 are contributing structures along with
deawar structures, structure (r) is the resonance hybrid of the structures (1) and (2).
(1)
(2)
(r)
E.g. C-C bond length in benzene is 1.39A0 as against the value of 1.54A0 for C-C single
bond and 1.34A0 for C-C double bond. (1A0 =10-10m)
48
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
Q. Why is benzene extra ordinarily stable though it contains three double bonds?
Ans. due to resonance . in benzene all the 6 electrons of three double bonds gets
delocalised resulting the stability of the molecule.
NECESSARY CONDITIONS FOR AROMATICITY:
a. Planarity: The ring should be planar due to delocalisation of electron cloud. In simple
words, all the carbons of the ring should be sp2 hybridised.
b. Complete delocalisation of the electrons in the ring: The electrons should be
completely delocalized above and below the plane of the molecule. In simple words, in
the ring alternate single and double bonds should exist.
c. Hückel Rule: Presence of (4n + 2) electrons in the ring where n is an integer (n =
0, 1, 2, . . .)
Benzene C6H6: n=1 Presence of electrons = (4n + 2) = 6 electrons.
Naphthalene: n=2 Presence of electrons = (4n + 2) = 10 electrons.
Anthracene:
n=3 Presence of electrons = (4n + 2) = 14 electrons.
49
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
Q. What are the necessary conditions for any system to be aromatic?
POLYNUCLEAR AROMATIC RING COMPOUNDS:
Examples of fused ring of arenes or polynuclear aromatic ring compounds:
Naphthalene
Anthracene
, C10H8 consists of two fused aromatic rings
or C14H10 consists of three fused rings.
Naphthalene and anthracene are two of the simplest examples of what are
called polynuclear aromatic compounds or in this specific case polycyclic aromatic
hydrocarbons(PAHs), also known as polycyclic arenes or polyaromatic hydrocarbons.
SOURCES OF AROMATIC COMPOUNDS:
1. Aromatic compounds from coal (Anthracite %age free C=90% , Bituminous C=70%,
lignite C=40%, peat C10-15%) by destructive distillation of coal. Destrucitve distillation
of coal gives coal gas, coal tar.
2. Aromatic compounds from petroleum by catalytic reforming or hydro forming.
PREPARATION OF BENZENE:
1. FROM ACETYLENE:
Alkynes when passed through a red hot iron tube, undergo cyclic polymerisation to from
aromatic hydrocarbons.
3CHCH red Hot Cu tube 873K benzene or C6H6
Similarly propyne can be converted to mesitylene.
2. FROM SODIUM BENZOATE (LABORATORY METHOD) SODA LIME REACTION:
DECARBOXYLATION REACTION AS CO2 IS REMOVED:
C6H5COONa
+ NaOH CaO
C6H6
+
Na2CO3
Sod. Benzoate
LIME
Benzene
3. FROM PHENOL:
C6H5OH
Zn dust
C6H6
+ ZnO
50
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
4. FROM CHLOROBENZENE: (REDUCTION)
C6H5Cl + 2[H] Ni-Al alloy C6H6 + HCl
NaOH
5. FROM BENZENE SULPHONIC ACID:
C6H5SO3H + H2O 150-2000C C6H6 + H2SO4
Benzenesulphonic acid
Benzene
6. FROM BENZENE DIAZONIUMCHLORIDE:
(a) C6H5N2Cl + 2[H]
SnCl2/ NaOH C6H6 + HCl + N2
Benzenediazonium chloride
Benzene
(b) C6H5N2Cl + H3PO2 + H2O Cu+1 ions C6H6 + H3PO3 + HCl + N2
Benzenediazonium chloride
Benzene
7. AROMATIZATION OR CATALYTIC REFORMING:
The conversion of aliphatic compound into aromatic compound in the presence of
catalyst is known as aromatisation. E.g.
REFORMING: The conversion of aliphatic compound into aromatic compound in the
presence of H as hydro forming and in the presence of platinum is known as platforming.
Cracking, isomerisation and aromatisation are widely used to increase increase the
octane rating of gasoline.
8. FROM PHENYL MAGNESIUM HALIDE:
RMgX
+
H2O
RH + MgX(OH)
C6H5MgBr +
H2O
C6H6 + MgBr(OH)
9. WURTZ FITTIG REACTION: Formation of alkyl benzene
C6H5Br + 2Na + CH3Br Wurtz fittig reaction C6H6CH3+ 2NaBr
dry ether
USES OF BENZENE:
1.
2.
3.
4.
Benzene is used in dry cleaning and as a solvent for oils, fats, rubber etc.
It is use in manufacturing of nitrobenzene which is used in medicine and dyes.
It is also used to make gammexene (an insecticide).
When mixed with petrol, it may be used as motor fuel.
CHEMICAL PROPERTIES OF BENZENE:
(A) ELECTROPHILIC SUBSTITUTION REACTIONS:
51
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
Q. Why does benzene undergo electrophilic substitution reactions easily and nucleophilic
substitutions with difficulty?
ANS. Orbital structure shows benzene has delocalised pi electrons lying above and below
the benzene ring is loosely held and thus these sites are available for the attack of
electrophile (e deficient species). Therefore benzene undergoes electrophilc addition
reactions with ease as compared to nucleophilic addition reaction.
(B) ADDITION REACTIONS
(C) OXIDATION REACTIONS:
52
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
CHEMICAL PROPERTIES OF AROMATIC HYDROCARBONS:
A. ELECTROPHILIC SUBSTITUTION REACTIONS:
(I) HALOGNENATION (CHLORINATION, BROMINATION, IODINATION)
Chlorination: C6H6 + Cl2 An AlCl3 310-320K C6H5Cl + HCl
(Electrophile: Cl+)
53
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
(II) SULPHONATION
Sulphonation: C6H6 + HSO3
(Electrophile : +SO3H)
(H2SO4) C6H5SO3 + H2O
(III) NITRATION
Examples: Nitration of benzene: C6H6 + HNO3 C6H5NO2 + H2O
(Electrophile: +NO2)
(IV) ALKYLATION OR FRIEDEL CRAFT ALKYLATION REACTION:
Friedal craft reaction: C6H6 + CH3Cl An AlCl3 C6H5CH3 + HCl
(Electrophile: CH3+)
(V) ACYLATION OR FRIEDEL CRAFT ACYLATION REACTION:
Friedal craft acylation: C6H6 + CHCOCl An AlCl3 C6H5COCH3 +HCl
Electrophile: (CH3CO+)
MECHANISM:(a)Formation of Electrophiles(b)formation of complex, (c) release of proton
MECHANISM OF NITRATION REACTION:
Examples: Nitration of benzene: C6H6 + HNO3 C6H5NO2 + H2O
(Electrophile:
+
NO2)
Q. Give mechanism of sulphonation of benzene? OR
Q. What is the mechanism for sulphonating / sulfonating benzene? or methyl benzene?
ANS. Benzene: C6H6 + SO3 ==> C6H5SO2OH
or C6H6 + H2SO4 ==> C6H5SO2OH + H2O
Benzene is heated with concentrated sulphuric acid or even better, 'fuming' sulphuric
acid, which has a higher sulphur trioxide content and more efficient at introducing the
sulphonic acid group into the benzene ring.
54
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
Step (1) Sulphur trioxide is formed (or already present). It is a
powerful electrophile, i.e. electron pair acceptor because of the effect of the three very
electronegative oxygen atoms bonded to the central sulphur atom.
Step (2) An electron pair from the delocalised pi electrons of the benzene ring forms a
C-S bond with the electron pair accepting sulphur trioxide forming a second highly
unstable carbocation. It is very unstable because the stable electron arrangement of the
benzene ring is partially broken to give a 'saturated' C (top right of ring).
Step (3) A hydrogensulphate ion removes a proton and the complete benzene ring is
reformed giving the anion of the aromatic sulphonic acid.
Step (4) is a proton transfer to give the sulphonic acid.
(VI) GATTERMAN KOCH REACTION:
Benzene or its derivatives on treatment with carbon monoxide and HCl in presence of
anhydrous aluminium chloride or cuprous chloride (CuCl) gives benzaldehyde or
substituted benzaldehydes + HCl
B. ADDITION REACTIONS:
(a) Addition of hydrogen: (catalytic reaction)
C6H6
Benzene
+ 3H2
Ni (2000C) C6H12
Cyclohexane
(b) Addition of halogen: chlorination
C6H6
Benzene
+ 3Cl2
sunlight (hv)
C6Cl6 + 6HCl
BHC (Benzene hexachloride)
BHC is a well known insecticide under name of gammexane or lindane.
NOTE: If halogenations is done in presence of AlCl3 it hexachlorobenzene.
55
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
(c) Addition of Ozone (O3):
C6H6 + 3O3 Zn/H2O
3 CHO
CH2Cl2
CHO Ethanedial (Glyoxal)
COUPLING OF BENZENE:
2 C6H6 +
RED HOT IRON TUBE C6H5-C6H5
Benzene
700-8000C
Biphenyl
C. CONTROLLED OXIDATION by KMnO4:
Benzene is un-reactive towards potassium permanganate (KMnO4) under normal
conditions. However oxidation of benzene in presence of V2O5 as catalyst at 5000C gives
maleic anhydride.
Benzene + 9/2 O2 V2O5 5000C
CHCOOH
║
heat Maleic anhydride +H2O
CHCOOH
OXIDATION OF ALKYL BENZENE BY:
From alkyl benzenes: Aromatic carboxylic acids can be prepared by vigorous oxidation
of alkyl benzenes with chromic acid or acidic or alkaline potassium permanganate
ETARD REACTION:
Chromyl chloride ( CrO2Cl2) oxidizes methyl group to a chromium complex, which on
hydrolysis gives corresponding benzaldehyde
COMBUSTION REACTIONS:
Arenes are inflammable liquids and burn with a sooty flame to produce CO2 and H2O.
(a) 2C6H6 +15O2 12CO2+ 6H2O
Q. Write chemical equations for combustion reaction of the following hydrocarbons:
(i) Toluene?
56
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
THE DIRECTIVE INFLUENCE OF VARIOUS GROUPS:
Directive influence of a functional group in mono substituted benzene:
A. ORTHO PARA DIRECTING GROUPS:
Groups which direct the incoming group to o & p positions.
Activating groups: Groups which increase the electron density on ortho and para
positions of the benzene ring. E.g. : –NH2, –NHR, –NHCOCH3,-OH, –OCH3, –CH3, –C2H5,
-SH, CH2Cl, Ph-, X- F-,Cl-, Br-, IDeactivating groups: Groups which decrease the electron density on ortho and para
positions of the benzene ring. E.g. :-X Halogens are moderately deactivating groups
because they have strong – I effect (electron withdrawing effect) which overpowers +R
effect .Due to this the overall electron density on benzene ring decreases. It makes
further substitution difficult. However, due to resonance the electron density on o– and
p– positions is greater than that at the m-position. Hence, they are also o– and p–
directing groups.
As a general rule substituent having atleast one lone pair of e- linked to a benzene ring
is o & p-director, however an alkyl group having no lone pair of electrons is also o and pdirector.
57
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
B. META DIRECTING GROUPS
The groups which direct the incoming group to meta position.
Example: –NO2, –CN, -NC, -COCl, -CONH2, –CHO, –COR, –COOH, –COOR, –SO3H,
COOCOR(as a general rule, substituent containing multiple (=or≡) bond is meta director.
58
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
CARCINOGENS:
Most Poly nuclear aromatic compounds are carcinogenic in nature. E.g. working in coal
prolonged exposure suffers from skin cancer.
Major component of this fraction is : 1,2-Benzanthracene.
Others are 3-methylcholanthracene, 1,2-Benzpyrene etc.
Number and position of the functional group such as –CH3,-OH, -CN, -OCH3 etc. Such
polynuclear hydrocarbons are formed by incomplete combustion of organic matter such
as coal, petroleum and tobacco etc. When these carcinogens enter human body, they
undergoes various biochemical reactions and finally damage DNA and ultimately leads to
cancer.
Q. Convert ethanoic acid to benzene? Hint: ethanoic acid CH4 by Soda Lime CH3Cl
Ethane by Wurtz, C2H5Cl, alc KOH C2H4 + Br2 C2H4Br2+ 2KOH Ethyne
polymerC6H6.
Q. How will you convert benzene into
(i) p-nitrobromobenzene (ii) m- nitrochlorobenzene (iii)p-nitrotoluene
(iv) acetophenone?
Hint (i) benzene bromination bromobenzene + Nitration o+ p nitro bromo
benzene separated by fractional distillation
(ii) benzene + HNO3 nitro benzene + Cl2 anhd AlCl3 m nitro chlorobenzene
(iii)benzene + CH3Cl Friedel Craft Reaction toluene + HNO3 o , p nitro toluene
fractional distillation p nitro toluene.
(iv)FCR C6H6+. CH3COCl acetophenone.
Q. Complete the reaction:
(a) C6H6+ O3 A
Zn, H2O B A= benzene triozonide B=glyoxal.
(b) complete the reaction B MnO2/ H+ C6H5CH3 + KMnO4/ H+ A hint A= benzoic
acid, B= benzaldehyde
(c) difference between alkylation and acylation? Write FCR
(d) what is halogen carrier? FeCl3 , AlCl3 lewis acidetc
(e) what is huckel rule? See notes
(f) which is meta director?-NO2, -SO3H, -Cl, -OH, -NH2, -CHO
Q. how many isomers are possible for mono substituted and disubstituted benzene? Hin
t mono -1, di- 3 (o/p/m)
Q. why benzene is extraordinarily stable although it contains 3 double bond?
Ans. resonance. delocalisation of pi electrons.
Q. arrange in decreasing order of acidic acharacter (a) ethne (b) benzene (c) n hexane
a>b>c ?
59
Class XI
HYDROCARBONS - ALKANES / ALKENE / ALKYNES/ AROMATICS
Q. nitration of nitrobenzene mainly gives meta product?T/F
Q. chlorobenzene on reduction with Ni-Al alloy gives toluene? False/ benzene is formed.
Q. all the C in benzene is __ hybridised? And. sp
Q. when ___________ is passed through red hot cu tube _____ is produced.
Ethyne / benzene.
Ans.
Q. electrophile in sulphonation of benzene is ____ (SO3+)
Q. nitration of bromo benzene or bromination of nitrobenzene gives same/different
bromo nitrobenzene. Different.
Q. nitration of benzene is electrophilic substitution reaction.
Q. IUPAC name C6H5-CH2-CH2-CH=CH2 4-Phenyl-But-1-ene
Q. o- C6H5(OH)(CH3) iupac name =2-methyl phenol
Q. How would you convert the following compounds into benzene?
(i) Ethyne (ii) Ethene (iii) Hexane
Hint (i) red hot cu (ii) ethene to Ethyne red hot cu benzene (iii) aromatisatio reaction n
hexane Cr2O3/ V2O5 cyclohexane aromatisation benzene.
Q. Arrange the following set of compounds in order of their decreasing
relative reactivity with an electrophile, E+
(a) (i)Chlorobenzene,(ii) 2,4-dinitrochlorobenzene, (iii)p-nitrochlorobenzene hint i>iii>iii
(b) (i)Toluene,(ii) p-H3C-C6H4–NO2, (iii)p-O2N–C6H4–NO2. ii>i>iii>iv
Q. Out of benzene, m–dinitrobenzene and toluene which will undergo nitration most
easily and why? Toluene > benzene> m-dinitrobenzene (CH3 eltron releasing gp while –
NO2 electron withdrawing group)
Q. Suggest the name of a Lewis acid other than anhydrous aluminium chloride which can
be used during ethylation of benzene. Ans. anhd FeCl3, SnCl4, BF3 etc.
Q. arrange the following set of compounds in order of decreasing reactivity with an
elctrophile? C6H5NO2, C6H5-OCH3 , C6H5Cl
Ans. C6H5-OCH3 > C6H5Cl > C6H5NO2,
Q. Write down the products of ozonolysis of 1,2-dimethylbenzene (o-xylene). arrange
benzene, n-hexane and ethyne in decreasing order of acidic behaviour. Also give reason
for this behaviour.
ANS METHYL GLYOXAL,1,2 DIMETHYLGLYOXAL
YOU CAN WIN WITH
YOUR
EFFORTS
ONLY.
----XXX---
60