* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Cancer Chemotherapy
Survey
Document related concepts
Transcript
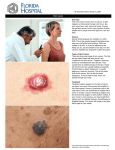
Chapter 54 Cancer Chemotherapy Background It is the second most common cause of death in the developed nations One in three people will be diagnosed with cancer during their lifetime The Both benign and malignant tumours manifest uncontrolled proliferation, but the latter are distinguished by their capacity for dedifferentiation, their invasiveness and their ability to metastasise (spread to other parts of the body cancer, malignant malignant tumour are synonymous terms neoplasm and Background Cancer: is a disease in which there is uncontrolled multiplication & spread within the body of abnormal forms of body’s own cells Cancer cells manifest, to varying degrees, four characteristics that distinguish them from normal cells: 1) 2) 3) 4) Uncontrolled proliferation Dedifferentiation & loss of function Invasiveness Metastasis Causes A normal cell turns into a cancer cell b/c of one or more mutation in its DNA, which can be inherited or acquired , which can be inherited or acquired, usually through exposure to viruses or carcinogens (e.g. tobacco products, asbestos) 2 main categories of genetic changes: – Induce of oncogenic transformation – Inactivation of tumour suppressor genes About 30 tumour suppressor genes and dominant oncogenes have been identified 100 Causes Incidence, geographical distribution, & behaviour of specific types of cancer are related to: age, sex, race, genetic predisposition, & exposure to environmental carcinogens Principles of cancer chemotherapy • Drug therapy is used in patients with cancer to: 1. Eradicate the disease 2. Induce a remission 3. Control symptoms Ideal anticancer drugs would eradicate cancer cells without harming normal tissues Unfortunately, most currently available agents do not specifically recognize neoplastic cells but affect all kind of proliferating cells Principles of cancer chemotherapy The treatment of cancer patients requires a skillful interdigitation of pharmacotherapy with other modalities of treatment (e.g., surgery and irradiation) In biochemical terms, cancer cells and normal cells are so similar in most respects that it is more difficult to find general, exploitable, biochemical differences between them Principles of cancer chemotherapy Individual patient characteristics choice of modalities determine the Not all patients can tolerate drugs, and not all drug regimens are appropriate for a given patient Renal and hepatic function, bone marrow reserve, general performance status, and concurrent medical problems all come into consideration in making a therapeutic plan Principles of Drug Treatment The log cell kill hypothesis: cytotoxic effect of anticancer drugs follow log-cell kinetics: a given dose would be predicted to kill a constant proportion/fraction of cells For example: if an individual drug leads to a 3 log kill of cancer cells and reduces the tumor burden from 1010 to 107 cells, the same dose used at a tumor burden of 105 cells reduces the tumor mass to 102 cells Cell kill is, therefore, proportional, regardless of tumor burden Benefits of combination therapy Combination-drug chemotherapy is more successful than single-drug treatment in most of the cancers for which chemotherapy is effective b/c: 1) It provides maximal cell kill within the range of toxicity tolerated by the host for each drug It provides a broader range of interaction between drugs and tumor cells with different genetic abnormalities in a heterogeneous tumor population It may prevent or slow the subsequent development of cellular drug resistance 2) 3) Benefits of combination therapy Certain principles have been used in designing such treatments Efficacy: each drug should have some individual therapeutic activity against the particular tumor being treated Toxicity: Drugs with different dose-limiting toxicities should be used to avoid overlapping toxicities Optimum scheduling: Intensive intermittent schedules should allow time for recovery from the acute toxic effects, primarily bone marrow toxicity Benefits of combination therapy Drugs that act by different mechanisms may have additive or synergistic therapeutic effects. Combination therapy will thus increase log cell kill and diminish the probability of emergence of resistant clones of tumor cells Several cycles of treatment should be given, since one or two cycles of therapy are rarely sufficient to eradicate a tumor. Most curable tumors require at least six to eight cycles of therapy Benefits of combination therapy Example of combination therapy of advanced Hodgkin’s disease: – MOPP (mechlorethamine, Oncovin [vincristine sulfate], procarbazine, prednisone) – ABVD (Adriamycin [doxorubicin hydrochloride], bleomycin,vinblastine, dacarbazine), has resulted in cure rates of 50 to 60% Cell-cycle specificity of drugs Both normal cells & tumor cells go through growth cycle The number of cells that are in various stages of the cycle may differ in the normal & neoplastic tissues The normal cell cycle consists of a definable sequence of events that characterize the growth and division of cells Cell-cycle specificity of drugs Cell cycle specific (CCS) drugs: effective only against replicating cells (most effective against hematological malignancies & in solid tumors in which large proportion of the cells are proliferating) Cell-cycle nonspecific (CCNS) drugs: many bind to cellular DNA. Useful against low growth and high growth tumors (e.g., carcinomas of the colon or non–small cell lung cancer) Resistance Primary resistance (inherent drug resistance): melanoma, renal cell cancer, & brain cancer Acquired resistance: genetic mutation particularly after prolonged administration of suboptimal doses (minimized by short term, intensive, intermittent therapy administration or by drug combinations) Possible mechanisms of Anticancer Drug Resistance Mechanism Chemotherapy Agents Improved proficiency in repair of DNA Cisplatin, cyclophosphamide, melphalan, mitomycin, mechlorethamine In drug activation Cytarabine, doxorubicin, fluorouracil, mercaptopurine, methotrexate, thioguanine in drug inactivation Cytarabine, mercaptopurine In cellular uptake of drug Methotrexate, melphalan In efflux of drug (multidrug resistance) Doxorubicin, daunorubicin, etoposide, vincristine, vinblastine, teniposide, docetaxel, paclitaxel, vinorelbine Alternative biochemical pathways Cytarabine, fluorouracil, methotrexate Alterations in target enzymes (DHFR, topoisomerase II) Fluorouracil, hyroxyurea, mercaptopurine, methotrexate, thioguanine, teniopside, doxorubicin, daunoruricin, idarubicin Multidrug resistance Tumor cells may become generally resistant to a variety of cytotoxic drugs on the basis of decreased uptake or retention of the drugs It is the major form of resistance to a broad range of structurally unrelated anticancer agents, including the anthracyclines, vinca alkaloids, taxanes, camptothecins, epipodophyllotoxins It is associated with increased expression of the MDR1 gene, which encodes a cell surface transporter glycoprotein (P-glycoprotein) Multidrug resistance Associated with increased expression of a normal gene (the MDR1 gene) for a cell surface glycoprotein (P-glycoprotein) involved in drug efflux Multidrug resistance can be reversed experimentally by calcium channel blockers, such as verapamil, and a variety of other drugs, which inhibit the transporter The multidrug resistance gene MDR1, which encodes the cell-surface molecule P-glycoprotein (PGP), can confer resistance to a wide variety of drugs. PGP transports drugs out of the cell, which is a process that requires the presence of two ATP-binding domains. These domains are a defining characteristic of this family of ATP-binding cassette (ABC) transporters. The exact mechanism of drug efflux is not well understood, but might involve either direct transport out of the cytoplasm or redistribution of the drug as it transverses the plasma membrane. Some cytotoxic drugs that are known substrates for PGP include etoposide, daunomycin, taxol, vinblastine and doxorubicin. PGP is modified by sugar moieties (black) on the external surface of the protein Toxicity of cancer chemotherapy Most anticancer s have a narrow therapeutic index Therapy aimed at killing rapidly dividing cancer cells also affects normal cells undergoing rapid proliferation (for example, cells of the buccal mucosa, bone marrow, gastrointestinal (GI) mucosa, and hair), contributing to the toxic manifestations of chemotherapy Common ADEs 1) 2) Bone marrow suppression that predisposes to infections GIT: – N & V: phenothiazines and other centrally acting – Damage to the the GIT muycosa: stomatitis, dysphagia, and diarrhea 3) 4) 5) Alopecia Sterility Hyperuricemia (tumor lysis syndrome) Drugs used in cancer chemotherapy 1) 2) 3) 4) 5) 6) Alkylating agents and related compounds Antimetabolites Cytotoxic antibiotics Plant derivatives (vinca alkaloids, taxanes, campothecins) Hormones Miscellaneous agents ALKYLATING AGENTS The alkylating agents are the largest class of anticancer agents 1) Nitrogen mustards: chlorambucil, cyclophosphamide, mechlorethamine 2) Nitrosureas: carmistine, lomustine 3) Alkylsulfonates: busulfan 4) Platinum analogs: cisplatin, carboplatin, and oxaliplatin 5) Other Alkylating Agents: dacarbazine, procarbazine, & bendamustine Mechanism of actions Form reactive molecular species/ intermediate that transfer of their alkyl groups to various cellular constituents The macromolecular sites of alkylation damage include DNA, RNA, proteins, and various enzymes Alkylations of DNA within the nucleus represent the major interactions that lead to cell death The major site of alkylation within DNA is the N7 position of guanine Most major alkylating agents are bifunctional, with two reactive groups ALKYLATING AGENTS Are cell cycle-nonspecific, but cells are most susceptible to alkylation in late G1 & S phases Used in combination with other agents to treat a wide variety of lymphatic and solid cancer Mutagenic & carcinogenic and can lead to secondary malignancies, such as acute leukemia Drug resistance 1) 2) 3) Increase capability to repair DNA lesions Decrease permeability of the cell to the alkylating agents Increase production/activity of glutathione of glutathione S-transferase, which can conjugate with and detoxify electrophilic intermediates A. Cyclophosphamide Pro-drug that needs hepatic activation by CYP450 (4hydroxy cyclophosphamide):The active compounds, are phosphoramide mustard and acrolein Clinical uses: Cyclophosphamide has a wide spectrum of antitumor activity: Breast cancer, ovarian cancer, non-Hodgkin's lymphoma, CLL, soft tissue sarcoma, neuroblastoma, Wilms' tumor, rhabdomyosarcoma Alternative to azathioprine in suppressing immunological rejection of transplant organs 1) 2) A. Cyclophosphamide Specific ADE: Hemorrhagic cystitis: Dysuria and decreased urinary frequency Due to acrolein in the urine Can be minimized by vigorous hydration & by use of sodium 2-mercaptoethane sulfonate (MESNA) which “traps” acrolein Other common ADEs: Alopecia NVD Bone marrow suppression B. Mechlorethamine Originally developed as a vesicant (nitrogen mustard) during world war I IV administered The major indication for mechlorethamine is Hodgkin’s disease; the drug is given in the MOPP regimen B. Mechlorethamine Specific ADEs: Marked vesicant action (blistering agent): care should be taken to avoid extravasation into Sc tissues or even spillage onto the skin Reproductive toxicity includes amenorrhea and inhibition of oogenesis and spermatogenesis Common ADEs: Bone marrow suppression Immunosuppression (herpes zoster infections, especially in patients with lymphomas) Alopecia NVD C. Nitrosureas: Carmustine (IV) & Lomustine (orally) Are highly lipid-soluble and are able to cross the BBB, making them effective in the treatment of brain tumors Both alkylation and carbamoylation contribute to the therapeutic and toxic effects of the nitrosoureas C. Nitrosureas: Carmustine (IV) & Lomustine (orally) ADEs: – Bone marrow depression: 4 to 5 weeks – Severe nausea and vomiting – Pulmonary toxicity, manifested by cough, dyspnea, and interstitial fibrosis (long term) – Less frequent: alopecia, stomatitis, and mild abnormalities of liver function – Potentially, mutagenic, teratogenic, and carcinogenic Streptozocin (STZ) Sugar-containing nitrosourea It has a high affinity for cells of the islets of Langerhans & is transported into the cell by the glucose transport protein GLUT2 Clinical use: insulin-secreting islet cell carcinoma of the pancreas ADEs: – Abnormal glucose tolerance (hypoglycemic coma) – Renal tubular damage in 5 to 10% of patients D. Platinum analogs (cisplatin, carboplatin, & oxaliplatin) 1) First generation: Cisplatin Clinical uses: solid tumors (non-small cell and small cell lung cancer, esophageal and gastric cancer, head and neck cancer, and genitourinary cancers, particularly testicular, ovarian, and bladder cancer) ADEs: – Renal toxicity (major) – N and V – Anemia: require transfusions of RBCs – Hearing loss (10 to 30% of patients) D. Platinum analogs (cisplatin, carboplatin, & oxaliplatin) 2) 3) Second generation: Carboplatin MOA, mechanisms of resistance, and clinical uses are identical to cisplatin it exhibits significantly less renal toxicity and IT toxicity, peripheral nerves, and hearing loss It is more myelosuppressive than cisplatin Third generation: oxaliplatin Similar to cisplatin and carboplatin, but with significant activity against colorectal cancer ADEs: Neurotoxicity manifested by a peripheral sensory neuropathy Other Alkylating Agents Procarbazine: Activated by hepatic CYPs to highly reactive alkylating species that methylate DNA Oxidative metabolism of this drug by microsomal enzymes generates azoprocarbazine and H2O2, which may be responsible for DNA strand scission It is commonly used in combination regimens for Hodgkin's and non-Hodgkin's lymphoma and brain tumors Other Alkylating Agents 1. Procarbazine: Procarbazine is a weak MAOI: hypertensive reactions may result from its use concurrently with sympathomimetic agents, TCA, or ingestion of foods with high tyramine content CNS toxicity with neuropathy, ataxia, lethargy, and confusion The carcinogenic potential of procarbazine is thought to be higher than that of most other alkylating agents Other Alkylating Agents 2. Dacarbazine It is used in the treatment of malignant melanoma It is a potent vesicant, and care must be taken to avoid extravasation Other alkylating agents 1. 2. 3. 4. 5. 6. 7. 8. Dacarbazine Temzolomide (brain tumor) Melphalan Chlorambucil Busulfan Bendamustine Thiotepa Altretamine ANTIMETABOLITES (Structural Analogs) Are structurally similar to endogenous compounds, such as vitamins, amino acids,and nucleotides These drugs can compete for binding sites on enzymes or can themselves become incorporated into DNA or RNA and thus interfere with cell growth and proliferation ANTIMETABOLITES (Structural Analogs) The antimetabolites in clinical use include: - Folic acid analogues: methotrexate - Purine analogues: mercaptopurine, thioguanine - Pyrimidine analogues: fluorouracil, cytarabine CCS drugs acting primarily in S phase Are also used as immunosuppressants Folate antagonist Methotrexate (MTX) Folic acid is an essential dietary factor that is converted by enzymatic reduction to a series of tetrahydrofolate (FH4) cofactors that provide methyl groups for the synthesis of precursors of DNA (thymidylate and purines) and RNA (purines) Interference with FH4 metabolism reduces the cellular capacity for one-carbon transfer and the necessary methylation reactions in the synthesis of purine ribonucleotides and thymidine monophosphate (TMP), thereby inhibiting DNA replication Folate antagonist Methotrexate (MTX) MoA: Cellular uptake of the drug is by carrier-mediated active transport MXT competitively inhibits the binding of folic acid to the enzyme dihydrofolate reductase (DHFR), interfering with the synthesis of tetrahydrofolate (FH4), which serves as the key one-carbon carrier for enzymatic processes involved in de novo synthesis of thymidylate, purine nucleotides, and the amino acids serine and methionine Folate (Diet) Methotrexate DHFR FH2 dTMP DHFR FH4 N5, N10- Ch2FH4 Purine biosynthesis Folate antagonist Methotrexate (MTX) MTX is metavolized to polyglutamte derivatives which are retained in the cell and are also potent inhibitors DHFR Clearance depends on renal function Drugs such as aspirin, penicillin, cephalosporins, and NSAIDs inhibit the renal excretion of MTX Folate antagonist Methotrexate (MTX) Resistance: (1) Decrease drug transport (2) Decrease formation of cytotoxic MTX polyglutamates (3) Increase levels of DHFR (4) Alter DHFR protein with reduced affinity for MTX Folate antagonist Methotrexate (MTX) Clinical uses – Cancer: Breast cancer, head and neck cancer, osteogenic sarcoma, primary central nervous system lymphoma, nonHodgkin's lymphoma, bladder cancer, choriocarcinoma – Immunosuppressive agent in severe rheumatoid arthritis Folate antagonist Methotrexate (MTX) Specific Toxicity 1. Common toxicities - GIT (NVD & ulcerative mucositis), stomatitis, melosuppression, alopecia, and dermatitis - Prevented or reveresed by administration of folinic acid (leucovorin) “leucovorin rescue” 2. Renal damage: – High doses of MTX and its 7-OH metabolite, which can ppt in the renal tubules – Alkalinization of the urine and adequate hydration can help Folate (Diet) Methotrexate DHFR FH2 dTMP DHFR FH4 N5, N10- Ch2FH4 Purine biosynthesis Leucovorin Folate antagonist Methotrexate (MTX) Specific Toxicity 3. Hepatotoxicity: long term use may lead to cirrhosis 4. Pulmonary toxicity (cough, dyspnea, fever, & cyanosis 5. Neurologic toxicities (intrathecal adminiosrtation): stiff neck, headach, meningeal irritation Folate antagonist Pemetrexed A new antifolate therapeutic agent that inhibits enzymes within the pyrimidine and purine cycle It works by inhibiting DHFR & thymidylate synthase It also inhibits glycinamide ribo-nucleotide formyl transferase (GARFT), an enzyme that is involved in purine synthesis and therefore reduces purine production Sites of Action of Pemetrexed. Actions of pemetrexed on purine and pyrimidine pathways in DNA synthesis. dUMP, Deoxyuridine Monophosphate; TMP, Thymidine Monophosphate; CH2FH2, Methylenetetrahydrofolate; CHOFH4, 10-Formyltetrahydrofolate; FH2, Dihydrofolate; FH4, Tetrahydrofolate; DHFR, Dihydrofolate Reductase; GARFT, Glycinamide Ribonucleotide Formyl Transferase. Folate antagonist Pemetrexed Clinical uses: in combination with cisplatin in the treatment of mesothelioma & as first-line treatment of non-small cell lung cancer, as a single agent in the second-line therapy of nonsmall cell lung cancer ADEs: myelosuppression, skin rash, mucositis, diarrhea, fatigue, & hand-foot syndrome Toxicity is reduced by the coadministration of folic acid, vitamin B12 and dexamethasone Purine analoges 6-thiopurine: 6- Mercaptopurine (6-MP) & 6thioguanine (6-TG) a. 6-MP Analogue of hypoxanthine Inactive and is activated intracellularly by hypoxanthineguanine phosphoribosyltransferases (HGPRT) to to form the monophosphate nucleotide 6-thioinosinic acid (TIMP) TIMP: – Inhibits several enzymes of de novo purine nucleotide synthesis – Incorporation into nucleic acids: the monophosphate form is eventually metabolized to the triphosphate form, which can then get incorporated into both RNA and DNA Purine analoges 6-thiopurine: 6- Mercaptopurine (6-MP) & 6thioguanine (6-TG) a. 6-MP Clinical use: acute myelogenous leukemia Potential D-D interaction with xanthine oxidase inhibitor (allopurinol): the dose of mercaptopurine must be reduced by 50–75% Specific toxicity: hepatic dysfunction (cholestasis jaundice, necrosis) Bone marrow is the principle toxicity Purine analoges 6-thiopurine: 6- Mercaptopurine (6-MP) & 6thioguanine (6-TG) b. 6-TG Analogue of the natural purine guanine 6-TG is converted intracellularly by the enzyme hypoxanthine guanine–phosphoribosyltransferase (HGPRTase) to 6-TGMP TGMP is further converted to the di- and triphosphates, thioguanosine diphosphate and thioguanosine triphosphate, which then inhibit the biosynthesis of purines Purine analoges 6-thiopurine: 6- Mercaptopurine (6-MP) & 6thioguanine (6-TG) b. 6-TG Clinical use: with cytarabine in the treatment of adult acute leukemia Interaction does not occur with allopurinol Specific toxicity: hepatic dysfunction (cholestasis jaundice, necrosis) less common than with mercaptopurine Bone marrow is the principle toxicity Pyrimidine analoges 1-Fluorouracil (5-FU) 1) 2) Halogenated pyrimidine analogue that must be activated metabolically: 5-fluoro-2’-deoxyuridine-5’-monophosphate (5FdUMP): forms a covalently ternary complex with the enzyme thymidylate synthase and the 5,10-methylenetetrahydrofolate 5-fluorouridine-5'-triphosphate (FUTP):incorporate into RNA, where it interferes with RNA processing and mRNA translation Pyrimidine analoges 1-Fluorouracil (5-FU) Clinical uses: Colorectal cancer, anal cancer, breast cancer, gastroesophageal cancer, head and neck cancer, hepatocellular cancer IV administered b/c of its severe toxicity to the GIT Resistance: Depletion of enzymes (especially uridine kinase) that activate 5-fluorouracil to nucleotides Increased thymidylate synthase level Altered affinity of thymidylate synthetase for FdUMP Increase in the pool of the normal metabolite deoxyuridylic acid (dUMP) Increase in the rate of catabolism of 5-fluorouracil Pyrimidine analoges 1-Fluorouracil (5-FU) 1. 2. 3. 4. ADEs Myelosuppression GIT (mucositis and diarrhea) Skin toxicity manifested by the hand-foot syndrome Neurotoxicity Hand-foot syndrome is a side effect of some types of chemotherapy that causes redness, swelling, and pain on the palms of the hands and/or the soles of the feet. Hand-foot syndrome occurs when small amounts of chemotherapy leak out of the capillaries (small blood vessels) in the hands and feet. Once out of the blood vessels, the chemotherapy damages the surrounding tissues http://www.cancer.net Pyrimidine analoges 2-Capecitabine Prodrug that is enzymatically converted to 5-FU in the tumor Adv: orally administered Clinical use: metastatic breast cancer that is resistant to first line drugs & colorectal cancer ADEs: – Main: diarrhea and the hand-foot syndrome – Myelosuppression, NV, and mucositis: the incidence is significantly less than that seen with IV 5-FU Pyrimidine analoges 3-Cytarabine (cytosine arabinoside, Ara-C) It is a CCS (S-phase) MOA: Is an analogue of the pyrimidine nucleosides cytidine and deoxycytidine Is activated by kinases to AraCTP: an inhibitor of DNA polymerases which will incorporate into DNA and can retard chain elongation This agent has absolutely no activity in solid tumors Its activity is limited exclusively to hematologic malignancies (e.g. acute myelogenous leukemia and non-Hodgkin's lymphoma) Pyrimidine analoges 3-Cytarabine (cytosine arabinoside, Ara-C) Resistance Defect in the transport process Changes in the kinase enzymes activity Increased deamination of the drug Specific Toxicity Leukoenphalopathy: high doses with intrathecal administration Pyrimidine analoges 4-Gemcitabine MOA: Activated to 2',2'-difluorodeoxycytidine triphosphate which will inhibit DNA synthesis by being incorporated into sites in the growing strand that ordinarily would contain cytosine Pyrimidine analoges 4-Gemcitabine Resistance: Alteration in the deoxycytidine kinase Increase tumor production of endogenous deoxycytidine Clinical use: First-line treatment of locally advanced or metastatic adenocarcinoma of the pancreas Non-small cell lung cancer, bladder cancer, ovarian cancer, soft tissue sarcoma, and non-Hodgkin's lymphoma PLANT ALKALOIDS These classes differ in their structures and MOA but share the multidrug resistance mechanism, since they are all substrates for the multidrug transporter Pglycoprotein Cell cycle specific agents Vinca alkaloids (vinblastine, vincristine) Podophyllotoxins (etoposide, teniposide) Camptothecins (Topotecan & Irinotecan) Taxanes (paclitaxel, docetaxel) A. Vinca alkaloids: Vinblastine, Vincristrine, & vinorelbine Structurally related compounds derived from Vinca rosea (Vinblastine & vincristrine) Vinorelbine is a semi-synthetic derivative Despite their structural similarity, there are significant differences between them in regard to clinical usefulness and toxicity MOA: The vinca alkaloids bind avidly to tubulin & inhibition tubulin polymerization, which disrupts assembly of microtubules. This inhibitory effect results in mitotic arrest in metaphase (M) prevent, and cell division cannot be completed A. Vinca alkaloids: Vinblastine, Vincristrine, & vinorelbine Resistance: – Decreased rate of drug uptake – Increased drug efflux : multidrug resistance & cross-resistance usually occurs with anthracyclines, dactinomycin, and podophyllotoxins A. Vinca alkaloids: Vinblastine, Vincristrine, & vinorelbine ADRs Vinblastine: NV, bone marrow suppression, alopecia, & vesicant Vincristine: Neurotoxicity: peripheral sensory neuropathy Syndrome of inappropriate secretion of antidiuretic hormone (SIADH) Vinorelbine: Bone marrow suppression with neutropenia B. Taxanes: Paclitaxel, Docetaxel, & Ixabepilone Cell cycle specific (G2/M phase of the cell cycle) MoA: They bind reversibly to the β-tubulin subunit promoting polymerization and stabilization of the polymer rather than disassembly. Thus, they shift the depolymerization-polymerization process to accumulation of microtubules. The overly stable microtubules formed are nonfunctional, and chromosome desegregation does not occur. This results in death of the cell Figure 1 Mechanism of action of docetaxel Mackler NJ and Pienta KJ (2005) Drug Insight: use of docetaxel in prostate and urothelial cancers. Nat Clin Pract Urol 2: 92–100 doi:10.1038/ncpuro0099 B. Taxanes: Paclitaxel, Docetaxel, & Ixabepilone Resistance: Multidrug resistant P-glycoprotein Mutation in the tubulin structure Clinical uses: advanced ovarian cancer and metastatic breast cancer. Non-small cell in combination with cisplatin B. Taxanes: Paclitaxel, Docetaxel, & Ixabepilone ADEs: Neutropenia: treatment with colony stimulating factor (Filgrastim) can help Peripheral neuropathy Transient, asymptomatic bradycardia: Paclitaxel Fluid retention: Docetaxel Serious hypersensitivty: patients are pretreated with dexamethazone, diphenylhydramine, and an H2 blocker C. podophyllotoxins: Etoposide & Teniposide Cell cycle specific (most active in the late S to G2 phase of the cell cycle) MoA: Both drugs bind to the topoisomerase II -DNA complex and prevent resealing of the break that normally follows topoisomerase binding to DNA The enzyme remains bound to the free end of the broken DNA strand, leading to an accumulation of DNA breaks and cell death Mechanism of action of etoposide D. Camptothecins: Topotecan & Irinotecan Cell cycle specific (most active in the S phase) MoA: Interfere with activity of topoisomerase I, the enzyme responsible for cutting & religating single DNA strands. Inhibition of the enzyme results in DNA damage DNA CPT Topoisomerase I Binding of CPT to topo I and DNA http://en.wikipedia.org/wiki/Camptothecin Action of Type I DNA topoisomerases ANTITUMOR ANTIBIOTICS Antitumor antibiotics produce their effect mainly by direct action on DNA, leading to disruption of the DNA function All the anticancer antibiotics now being used in clinical practice are products of various strains of the soil microbe Stremptomyces Cell cycle non-specific A. Anthracyclin antibiotics Agents: Doxorubicin, daunorubicin, idarubcin, epirubicin , & mitoxantrone MoA: a. b. c. d. Inhibitoin of topoisomerase II Intercalation in the DNA: block the synthesis of DNA and RNA, and DNA strand scission Binding to cellular membranes to alter fluidity and ion transport Generation of semiquinone free radicals and oxygen free radicals through an iron-dependent, enzyme-mediated reductive process Doxorubicin Doxorubicin–DNA complex A. Anthracyclin antibiotics Specific Toxicity: Cardiotoxicity: arrhythmias and conduction o o abnormalities, pericarditis, and myocarditis Results from the generation of free radical and lipid peroxidation Reduced with: Lower weekly doses or continuous infusions of doxorubicin Treatment with the iron-chelating agent dexrazoxane Liposomal-encapuslated formulations of doxorubicin A. Anthracyclin antibiotics Radiation recall reaction with erythema and desquamation of the skin observed at sites of prior radiation therapy Doxorubicin will impart a reddish color to the urine for 1 or 2 days after administration Bone marrow suppression Hyperpigmentation of nail beds and skin creases, and conjunctivitis B. Mitomycin (mitomycin C) It is sometimes classified as an alkylating agent b/c it undergoes metabolic activation through an to generate an alkylating agent that cross-links DNA ADRs: – Hemolytic-uremic syndrome: microangiopathic hemolytic anemia, thrombocytopenia, and renal failure C. Bleomycin It is a small peptide that contains a DNA-binding region and an iron-binding domain at opposite ends of the molecule CCS drug active in G2 phase MOA: It acts by binding to DNA, which results in singlestrand and double-strand breaks following free radical formation, and inhibition of DNA biosynthesis. The fragmentation of DNA is due to oxidation of a DNA-bleomycin-Fe(II) complex and leads to chromosomal aberrations C. Bleomycin Specific Toxicity: Cause: Bleomycin hydrolase, which inactivates bleomycin, virtually absent in lungs and skin Pulmonary toxicity: pneumonitis with cough, dyspnea, dry inspiratory crackles on examination, and infiltrates on chest x-ray physical Skin toxicity: hyperpigmentation, erythematosus rashes, and thickening of the skin over the dorsum of the hands and at dermal pressure points, such as the elbows MISCELLANEOUS ANTICANCER DRUGS L-Asparaginase Enzyme that depletes serum L-asparagine to aspartic acid and ammonia It is used in treatment of childhood acute lymphocytic leukemia ADEs: hypersensitivity reactions, decrease in clotting factors, liver abnormalities, pancreatitis, seizures, and coma due to ammonia toxicity Because tumor cells lack asparagine synthetase, they require an exogenous source of L-asparagine. Thus, depletion of Lasparagine results in effective inhibition of protein synthesis