* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download macromolecules notes
Basal metabolic rate wikipedia , lookup
Two-hybrid screening wikipedia , lookup
Peptide synthesis wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Fatty acid synthesis wikipedia , lookup
Protein–protein interaction wikipedia , lookup
Genetic code wikipedia , lookup
Phosphorylation wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Blood sugar level wikipedia , lookup
Nucleic acid analogue wikipedia , lookup
Metalloprotein wikipedia , lookup
Glyceroneogenesis wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Biosynthesis wikipedia , lookup
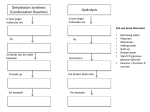
MACROMOLECULES NOTES I. Chemistry of Carbon A. Carbon has four electrons available for bonding. B. Carbon forms strong covalent bonds with hydrogen, oxygen, sulfur, nitrogen, and phosphorus. C. Carbon atoms can form bonds with each other. Ethane Ethene Ethyne Benzene D. Bonding between carbon atoms gives carbon the ability to form chains that can be unlimited in length and shape. II. Macromolecules -Macromolecules are giant molecules that can be made up of thousands of smaller molecules. -Macromolecules are polymers. Polymers are made up of smaller units called monomers. A. Carbohydrates (saccharides) 1. Monosaccharides (simple sugars): General formula = (CH2O)n, where n ≥ 3. a. Glucose 6 -known as blood sugar -formula = C6H12O6 5 #1 Carbon 4 b. Galactose -known as brain sugar -formula = C6H12O6 -found in gums, mucilage, pectin, and milk 3 c. Fructose -known as fruit or honey sugar -formula = C6H12O6 2. Dissacharides (double sugars) a. Sucrose -known as cane sugar -is made of glucose and fructose -formula = C12H22O11 H2O 1 2 Simple version MACROMOLECULES NOTES b. Lactose -known as milk sugar -is made of glucose and galactose -formula = C12H22O11 1→4 glycosidic bond H2O c. Maltose -known as malt sugar -is made of glucose + glucose -formula = C12H22O11 H2O 3. Polysaccharides (Complex carbohydrates) a. Glycogen -used by animals to store extra sugar -sometimes called ‘animal starch’ -made entirely of α-glucose with α(1→4) glycosidic bonds -is highly branched molecule in the form of granules Glycogen Highly -branching occurs every 8 to 12 glucose granule units via (1→6) bonds branched -insoluble in water -primary storage is in adipose tissue (body fat) -primarily made in liver and skeletal muscle -the liver will remove or add glucose to the blood in response to insulin levels: -after a meal, pancreas releases insulin into blood stream -when insulin levels are high, liver removes glucose and makes glycogen. -when insulin levels are low, liver breaks down glycogen and releases glucose. b. Starches -used by plants to store extra sugar -the enzyme amylase breaks down starches into maltose -stored in amyloplasts in the form of starch granules. Amyloplasts in a potato cell Starch granules -found in high concentrations in wheat, corn, rice, roots and tubers (potatoes) 2 MACROMOLECULES NOTES b. Starches continued i. Amylose -can be linear or helical in shape -insoluble in water -made entirely of α-glucose with α(1→4) glycosidic bonds ii. Amylopectin -moderately branched -branching occurs every 24 to 30 glucose units via (1→6) bonds -soluble in water -made entirely of α-glucose with α(1→4) glycosidic bonds c. Cellulose (C6H10O5)n -structural component of plant cell walls -90% of cotton and 40% of wood is cellulose -used to make cardboard and paper -insoluble in water -most abundant substance in nature -ruminants and termites can digest cellulose with help of symbiotic microbes -made entirely of β-glucose with β(1→4) bonds (every other glucose is inverted) d. Chitin (C8H13O5N)n -main component of the cells walls of fungi -main component of arthropod (crabs, shrimps, insects) exoskeletons -main component in the beaks of squid and octopuses -made entirely of N-acetylglucosamine with β(1→4) bonds. 3 MACROMOLECULES NOTES B. Lipids 1. Examples: Oils, fats, waxes, steroids, cholesterol 2. Non-polar and insoluble in water 3. Fats provide about 9 kilocalories/gram (compared to carbohydrates at 4 kilocalories/gram) 4. Triglycerides (a common lipid) a. Main constituent of vegetable oil and animal fats b. Formed by combining glycerol with three molecules of fatty acid. c. An ester bond forms when glycerol links to a fatty-acid chain releasing a molecule of water. Before bonding During bonding After bonding d. These are two simple ways of drawing a triglyceride 4 MACROMOLECULES NOTES e. Triglycerides are stored in adipose tissue (body fat) consisting of adipocytes (fat cells). f. Mammals use triglycerides for - Bouyancy (lipids are less dense than water so it helps animals float) g. Insulation (a layer of fat beneath the skin cuts down on heat loss) h. Production of water (fatty acids produce a lot of metabolic water when they are oxidized during cellular respiration). Camels’ humps are made of fat and supply water when needed. 5 MACROMOLECULES NOTES C. Nucleic Acids 1. Contain hydrogen, oxygen, nitrogen, carbon, and phosphorus 2. Monomers are called nucleotides 3. Nucleotide structure: Phosphate group Purine structure 5-carbon sugar (pentose) Nitrogenous base Two types Pyrimidine structure 4. The nucleotide adenosine triphosphate (ATP), helps in storing and transferring energy. Three Phosphate groups Energy is stored in the phosphate bonds Adenine (a purine) 5. Two kinds of nucleic acids (both store and transmit genetic information) a. Ribonucleic acid (RNA) -made of nucleotides that contain the sugar ribose (C5H10O5) -generally exists as a single-strand structure RNA structure b. Deoxyribonucleic acid (DNA) -made of nucleotides that contain the sugar deoxyribose (C5H10O4) -generally exists as a double-helix structure 6 MACROMOLECULES NOTES D. Proteins 1. Contain carbon, hydrogen, oxygen, and nitrogen. 2. Monomers of proteins are called amino acids. 3. There are more than 20 different amino acids found in nature. 4. Amino acid structure: 5. Each amino acid has a different side chain called an R-group. a. The side chains have different properties b. Some side chains are polar (examples: glycine, alanine, serine, tyrosine) c. Some side chains are nonpolar (examples: proline, tryptophan, valine) d. Some side chains are acidic (examples: aspartic acid, glutamine acid) e. Some side chains are basic (examples: lysine, histidine) Glycine Alanine Serine 6. Covalent bonds called peptide bonds link amino acids to form a polypeptide. a. A water is released whenever a peptide bond is formed. b. Proteases are protein enzymes that break peptide bonds between amino acids. 7 MACROMOLECULES NOTES 7. A protein is a molecule made from one or more polypeptides. a. Levels of structure -Primary structure: The order of amino acids in the polypeptide chain. -Secondary structure: The folding of the polypeptide chain. -Tertiary structure: The three-dimensional arrangement of the polypeptide chain. -Quaternary structure: Two or more polypeptide chains intertwined with one another. 8. Example Proteins a. Enzymes: Involved in building and breaking down macromolecules -DNA polymerase assembles nucleotides into a DNA polymer -Amylase breaks down starch into maltose b. Transport proteins: Involved in binding and carrying molecules -Hemoglobin carries oxygen to cells -Glut1 transports glucose across cell membranes c. Storage proteins -Casein, found in milk, stores nutrients -Ferritin stores and release iron -Albumin is found in plasma and binds water and cations (such as Ca2+, K+, and Na+). It regulates osmotic pressure in blood. d. Contractile proteins -Actin & Myocin are found in muscle e. Structural proteins -Collagen fibers are the main component of connective tissue, like tendons and cartilage -Elastin is a component of connective tissue that is elastic and allows many tissues in the body to resume their shape after stretching or contracting. -Keratin is the key structural component of skin, hair, and nails. -Fibroin is the main protein that makes up silk and spider webs. f. Defensive proteins -Immunoglobulin G (IgG) are antibodies are proteins that help defend the body against bacteria, viruses, and fungi. -Fibrin is involved in blood clotting 8 MACROMOLECULES NOTES 9. Protein Models 9