* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download (+e) + - Purdue Physics
Survey
Document related concepts
Magnetic monopole wikipedia , lookup
Introduction to gauge theory wikipedia , lookup
Electromagnetism wikipedia , lookup
Aharonov–Bohm effect wikipedia , lookup
Speed of gravity wikipedia , lookup
Nuclear physics wikipedia , lookup
Fundamental interaction wikipedia , lookup
Elementary particle wikipedia , lookup
History of subatomic physics wikipedia , lookup
Maxwell's equations wikipedia , lookup
Circular dichroism wikipedia , lookup
Lorentz force wikipedia , lookup
Field (physics) wikipedia , lookup
Atomic nucleus wikipedia , lookup
Electrical resistivity and conductivity wikipedia , lookup
Electric charge wikipedia , lookup
Transcript
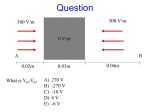
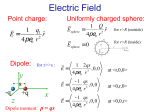
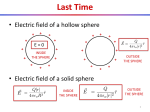
Chapter 15 Matter and Electric Fields Question 1 (Chap. 14) What is the direction of the electric field at location X, due to the dipole? + C B D A E X Question 2 (Chap. 14) Locations 1 and 2 are equidistant from the center of the dipole. At which location is the magnitude of the electric field larger? d + 1 d A. at location 1 B. at location 2 C. magnitudes are the same 2 Net Charge Matter is made out of atoms. Atom contains charged particles: electrons (-e), protons (+e) Neutral atom: number of electrons and protons is equal: Example: Hydrogen atom: 1 proton, 1 electron net charge = (+e) + (-e)=0 Sodium atom: 11 protons, 11 electrons Sodium atom (Na) can lose an electron: Sodium ion (Na+): (+11e) + (-10e) = +e Ordinary matter is electrically neutral. However, can be charged by adding/removing charged particles Can we create an excess charge inside a sample? Conservation of Charge The net charge of a system and its surroundings cannot change If one object gets charged positively, there must be an object which gets charged negatively. The net electric charge is conserved in any physical process. Charge can be transferred from one object to another. Annihilation : e+ + e- = g + g Induced Dipole An applied electric field creates induced dipoles! E • it is not a permanent dipole • an induced dipole is created when a neutral object is polarized by an applied electric field Polarization Amount of polarization p in most materials is proportional to the magnitude of the applied electric field: p = aE - “polarizability” of a material In an induced dipole, is the distance between the charges fixed? The distance is proportional to the strength of the applied field. A Neutral Atom and a Point Charge 1. Charge q1 creates field E1 at the location of the atom q1 E1 = rˆ 2 4pe 0 r 1 A Neutral Atom and a Point Charge 1) E1 = q1 rˆ 2 4pe 0 r 1 2. Field E1 polarizes the atom creating a dipole 1 aq1 p = aE1 = rˆ 2 4pe 0 r A Neutral Atom and a Point Charge 1) E1 = q1 rˆ 2 4pe 0 r 2) p = 1 aq1 rˆ 2 4pe 0 r 1 3. Dipole creates field E2 at the location of q1 æ 1 ö 2aq1 1 2p 1 2a E2 = = E1 = ç r̂ ÷ 3 3 5 4 pe 0 r 4 pe 0 r è 4 pe 0 ø r 2 A Neutral Atom and a Point Charge q1 E = 1) 1 4pe r 2 rˆ 0 2 1 2) p = 3) 1 aq1 rˆ 2 4pe 0 r 4. Induced dipole exerts force F1 on the charge: 2 æ 1 ö 2aq12 ÷÷ F1 = q1E2 = çç rˆ 5 è 4pe 0 ø r æ 1 ö 2aq1 ÷÷ E2 = çç rˆ 5 è 4pe 0 ø r A Neutral Atom and a Point Charge q1 E = 1) 1 4pe r 2 rˆ 0 2 1 1 aq1 p = rˆ 2) 2 4pe 0 r 3) æ 1 ö 2aq1 ÷÷ E2 = çç rˆ 5 è 4pe 0 ø r 4) æ 1 ö 2aq12 ÷÷ F1 = çç rˆ 5 è 4pe 0 ø r 2 5. The charge q1 exerts force F2 on the dipole (reciprocity): 2 æ 1 ö 2aq12 ÷÷ F2 = - F1 = -çç rˆ 5 è 4pe 0 ø r A Neutral Atom and a Point Charge 1) E1 = q1 rˆ 2 4pe 0 r 2) p = 1 aq1 rˆ 2 4pe 0 r 1 2 3) æ 1 ö 2aq1 ÷÷ E2 = çç rˆ 5 è 4pe 0 ø r 4) æ 1 ö 2aq12 ÷÷ F1 = çç rˆ 5 è 4pe 0 ø r 5) æ 1 ö 2aq12 ÷÷ F2 = -çç rˆ 5 è 4pe 0 ø r 2 2 Neutral atoms are attracted by charges! Interaction strength ~ 1/r5 Exercise Atom A is easier to polarize than atom B. Which atom would experience a greater attraction to a point charge a distance r away? A B - FA + - + FB 2 æ 1 ö 2aq12 ÷÷ F2 = çç ~a 5 è 4pe 0 ø r Conductors and Insulators Different materials respond differently to electric field Conductor: contains mobile charges that can move through material Insulator: contains no mobile charges Polarization of Insulators Insulator: Electrons are bound to the atoms or molecules. Electrons can shift slightly (<1 Å), but remain bound to the molecule. Individual atoms or molecules can be polarized by external electric field. There are a lot of molecules – the net effect produced by the induced dipoles can be very large. Polarization of Insulators Diagram showing polarization of an insulator: Dipoles: exaggerated in size; stretch: degree of polarization No mobile charges: excess charges stay where they are Low Density Approximation p = aE ¹ aEapplied Field E at a location of a molecule is a superposition of the external applied field and the field created by other induced dipoles: ( p = a Eapplied + Edipoles Simplifying assumption: ) Eapplied >> Edipoles p » a Eapplied Conductors There are charges which can move freely throughout the material E In contrast to an insulator, where electrons and nuclei can move only very small distances, the charged particles in a conductor are free to move large distances. Polarization of conductors differs from that of insulators. Ionic Solutions are Conductors (home) Salt water: Na+ and ClH+ and OH- Apply external electric field Enet = Eapplied + Ech arg es When an electric filed is applied to a conductor, the mobile charged particles begin to move in the direction of the force exerted on them by the field. As the charges move, they begin to pile up in one location, creating a concentration of charge creates electric field. The net electric field is the superposition of the applied field and field created by the relocated charges. Ionic Solutions are Conductors (home) Assumption: charges will move until Enet=0 (static equilibrium) Proof: by contradiction: Assume Enet≠0 Mobile ions will move This is not equilibrium Enet = Eapplied + Echarges Assumption Enet≠0 is wrong Enet=0 It is not a shielding effect, but a consequence of superposition! A Model of a Metal Metal lattice Positive atomic cores and mobile-electron sea Electrons are not completely free – they are bound to the metal as a whole. There is no net interaction between mobile electrons Drude Model of Electron Motion in a Metal E p Fnet (e) Enet t - No net interaction between mobile electrons Forget previous velocity after collision p p 0 eEnet t v v p eEnet t me me et Enet me et me (mobility) Later we will show that conductivity () μ Some important sources of collision: - impurities - thermal motion of atoms (more motion at higher temperature T shorter t lower [common feature of metals]) Metal in Electric Field Enet inside the conductor will be: A. Uniform positive B. Uniform negative C. Zero D. will have complex pattern Metal in Electric Field Simplified diagram of polarized metal Note: It is not charged! Net charge is still zero Electric Field inside Metal In static equilibrium: Enet = Eapp + E pol = 0 Enet= 0 everywhere inside the metal! Mobile charges on surface rearrange to achieve Enet= Actual arrangement might be very complex! It is a consequence of 1/r2 distance dependence Enet= 0 only in static equilibrium! 0 Excess Charge on Conductors Excess charges in any conductor are always found on an inner or outer surface! Conductors versus Insulators Conductor Insulator yes no Polarization entire sea of mobile charges moves individual atoms/molecules polarize Static equilibrium Enet= 0 inside Enet nonzero inside Excess charges only on surface anywhere on or inside material Distribution of excess charges Spread over entire surface located in patches Mobile charges