* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Beyond Element 83 are very unstable (radioactive)
Nuclear fission product wikipedia , lookup
Fallout shelter wikipedia , lookup
Radioactive waste wikipedia , lookup
Nuclear binding energy wikipedia , lookup
Radioactive decay wikipedia , lookup
Isotope analysis wikipedia , lookup
Isotopic labeling wikipedia , lookup
Valley of stability wikipedia , lookup
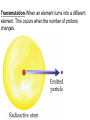
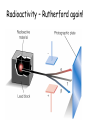
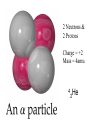
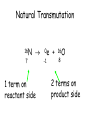
TOPIC: Radioactivity & Natural Transmuations Do Now: Where on your reference table can you find radioactive particles? Not all isotopes are stable Stable isotopes have… 1:1 ratio of n0 to p+ (for elements <20) 1.5:1 ratio of n0 to p+ (for elements >20) Beyond Element 83 are very unstable (radioactive) • No amount neutrons can hold nucleus together once it has 83+ protons • All Elements 83 and above on PT are radioactive • Other elements may have radioactive isotopes applet Radioisotopes- unstable nucleus – they are radioactive the nucleus of element emits subatomic particles and/or electromagnetic waves… by emitting these particles, nucleus changes into different element (it’s trying to become more stable) Radioactive Decay Series • Sometimes 1 transmutation isn’t enough to achieve stability • Some radioisotopes go through several changes before they achieve stability (and are no longer radioactive) U-238 PB-206 Transmutation-When an element turns into a different element. This occurs when the number of protons changes. These subatomic particles that are emitted were discovered around 1900 • Alpha rays • Beta rays • Gamma rays Radioactivity – Rutherford again! Penetration & Shielding 2 Neutrons & 2 Protons Charge = +2 Mass = 4amu 4 He 2 Beta Particle – fast moving electron Transmutation • 2 types –Natural –Artificial Natural Transmutation Happens all by itself (spontaneous) Not affected by anything in environment • 1 term on reactant side • Original isotope • 2 terms on product side • Emitted Particle • New Isotope Natural Transmutation 16N 7 1 term on reactant side 0e -1 + 16O 8 2 terms on product side Balancing Nuclear Equations 16N 7 0e -1 + 16O 8 Conservation of mass number: 16 = 0 + 16 Conservation of atomic number: 7 = -1 + 8 so Y = 228 232 = 4 + Y 232Th 90 4He 2 + Y X Z Conservation of Mass Number: sum of mass numbers on left side must = sum of mass numbers on right side 232Th 90 42He + 228 X Z 90 = 2 + Z so Z = 88 Conservation of Atomic Number: sum of atomic numbers on left side must = sum of atomic numbers on right side