25., Fatty ocid oxidation
... In Chapter 24 we saw that the carbons of the aceryl CoA produced by the catabolism of glucose can be completely oxidized to carbon dioxide in the citric acid cycle. Each molecule of acetyl CoA oxidized in this fashion yields enough energy to make one molecule of AIB one molecule of FADH2,and three m ...
... In Chapter 24 we saw that the carbons of the aceryl CoA produced by the catabolism of glucose can be completely oxidized to carbon dioxide in the citric acid cycle. Each molecule of acetyl CoA oxidized in this fashion yields enough energy to make one molecule of AIB one molecule of FADH2,and three m ...
glucose
... phosphoenolpyruvate formation • Carboxylation of pyruvate is located in mitochondrial matrix – at the same time it can serve as anaplerotic reaction of citric acid cycle (se lecture citric acid cycle) • Oxaloacetate cannot be transported across mitochondrial membrane – it must be transported in form ...
... phosphoenolpyruvate formation • Carboxylation of pyruvate is located in mitochondrial matrix – at the same time it can serve as anaplerotic reaction of citric acid cycle (se lecture citric acid cycle) • Oxaloacetate cannot be transported across mitochondrial membrane – it must be transported in form ...
Phylogenomic Investigation of Phospholipid Synthesis in Archaea
... have allowed some tolerance to recruitment of evolutionary unrelated enzymes not only in archaea but also in some eukaryotes that have replaced their ancestral PMK by a non-homologous one [35, 36]. In agreement with this idea, the archaeal IPK itself appears to be able to use different substrates [37 ...
... have allowed some tolerance to recruitment of evolutionary unrelated enzymes not only in archaea but also in some eukaryotes that have replaced their ancestral PMK by a non-homologous one [35, 36]. In agreement with this idea, the archaeal IPK itself appears to be able to use different substrates [37 ...
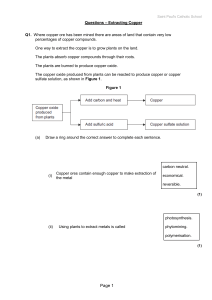
Lesson 22 - extracting copper questions
... Balance the symbol equation for the reaction. Cu2S + ........O2 → .........CuO + SO2 ...
... Balance the symbol equation for the reaction. Cu2S + ........O2 → .........CuO + SO2 ...
Oxidative metabolism in thermogenic tissues of the swordfish and
... served in another type of muscle, insect flight muscle (Sacktor, '76). Such activation of mitochondrial respiration may play a role in coupling SR calcium release to mitochondrial respiration. In swordfish brain heater organ, calcium leaking from the SR may stimulate mitochondrial respiration before ...
... served in another type of muscle, insect flight muscle (Sacktor, '76). Such activation of mitochondrial respiration may play a role in coupling SR calcium release to mitochondrial respiration. In swordfish brain heater organ, calcium leaking from the SR may stimulate mitochondrial respiration before ...
Camp 1 - University of California, Santa Cruz
... • Pyruvate metabolized three ways: • depends on organism & presence/absence of O2 ...
... • Pyruvate metabolized three ways: • depends on organism & presence/absence of O2 ...
Metabolism of BCAAs
... represent the amino acid cysteine, while the X’s can be any amino acid. Both isozymes of BCAT are reversible, and it is this redox center that permits this reversibility. In most cells, BCAT enzymes operate near equilibrium, with both substrate and product concentrations being at or below their Km v ...
... represent the amino acid cysteine, while the X’s can be any amino acid. Both isozymes of BCAT are reversible, and it is this redox center that permits this reversibility. In most cells, BCAT enzymes operate near equilibrium, with both substrate and product concentrations being at or below their Km v ...
Identification of possible tworeactant sources of
... adenylate-dependent steps in the system prescribes that the sum o f concentrations o f ATP a n d ADP will remain constant. D u e t o this adenylate conservation constraint, the concentrations of A T P and A D P become interdependent and it suffices t o consider one of them as an independent variable ...
... adenylate-dependent steps in the system prescribes that the sum o f concentrations o f ATP a n d ADP will remain constant. D u e t o this adenylate conservation constraint, the concentrations of A T P and A D P become interdependent and it suffices t o consider one of them as an independent variable ...
ch_25_lecture_presentation
... cytosol releases small amounts of ATP that are significant only under unusual Aerobic Metabolism conditions. (in mitochondria) ...
... cytosol releases small amounts of ATP that are significant only under unusual Aerobic Metabolism conditions. (in mitochondria) ...
2566 Part 1
... ATP exists in every living tissue its breakdown gives energy for all life functions ...
... ATP exists in every living tissue its breakdown gives energy for all life functions ...
The Mechanism of Propionic Acid Formation by
... round hypothetical pathways for anaerobic utilization of acetate (Wood & Werkman, 1938). Krebs & Eggleston (1941) rejected Wood & Werkman’s hypothesis and suggested that acetate was oxidized to CO,, but produced no convincing evidence to support this. Carson (1948) obtained indications in isotope ex ...
... round hypothetical pathways for anaerobic utilization of acetate (Wood & Werkman, 1938). Krebs & Eggleston (1941) rejected Wood & Werkman’s hypothesis and suggested that acetate was oxidized to CO,, but produced no convincing evidence to support this. Carson (1948) obtained indications in isotope ex ...
Gluconeogenesis
... than in the cytoplasm. Thus, for the final step of gluconeogenesis, G6P must be transported into the ER, the phosphate is cleaved off, and then glucose and phosphate are transported back out. Deficiencies in either glucose-6-phosphatase or any of the three transporters result in von Gierke’s disease ...
... than in the cytoplasm. Thus, for the final step of gluconeogenesis, G6P must be transported into the ER, the phosphate is cleaved off, and then glucose and phosphate are transported back out. Deficiencies in either glucose-6-phosphatase or any of the three transporters result in von Gierke’s disease ...
AMP-activated protein kinase regulation of fatty acid oxidation in the
... ability of AMPK to control both the production of malonylCoA (via ACC) and the degradation of malonyl-CoA (via MCD) is an attractive hypothesis. Our early studies have shown that purified AMPK does not modify MCD activity in vitro [30]. However, work by Saha et al. [50] suggests that 5-amino-4-imida ...
... ability of AMPK to control both the production of malonylCoA (via ACC) and the degradation of malonyl-CoA (via MCD) is an attractive hypothesis. Our early studies have shown that purified AMPK does not modify MCD activity in vitro [30]. However, work by Saha et al. [50] suggests that 5-amino-4-imida ...
A newly discovered Anaerococcus strain responsible for axillary
... structure similar to that of 3M2H. This fatty acid derivative is released from precursor, which is conjugates of HMHA with L-glutamine, by the action of bacterial Naacylglutamine aminoacylase (Natsch et al., 2003, 2006). In addition, Taylor et al. (2003) found that there was a clear correlation betw ...
... structure similar to that of 3M2H. This fatty acid derivative is released from precursor, which is conjugates of HMHA with L-glutamine, by the action of bacterial Naacylglutamine aminoacylase (Natsch et al., 2003, 2006). In addition, Taylor et al. (2003) found that there was a clear correlation betw ...
Seminario Glúcidos 3 y lípidos 1. Comente los mecanismos de
... up in distilled water, sufficient KC1 was added to the test flasks to provide a fmal concentration of about 0.05 M. Throughout these fractionations, it was found essential that low temperatures be maintained in order to preserve enzyme activity. We have found that the Sorvall angle centrifuges are e ...
... up in distilled water, sufficient KC1 was added to the test flasks to provide a fmal concentration of about 0.05 M. Throughout these fractionations, it was found essential that low temperatures be maintained in order to preserve enzyme activity. We have found that the Sorvall angle centrifuges are e ...
Photo Album
... (A) Net synthesis of TCA cycle-derived amino acids requires the ATP-dependent CO2 fixation reaction catalyzed by pyruvate carboxylase to generate oxaloacetate (OAA) from pyruvate. Condensation of the OAA with acetyl CoA from a second molecule of pyruvate forms a “new” molecule of citrate, a 6-carbon ...
... (A) Net synthesis of TCA cycle-derived amino acids requires the ATP-dependent CO2 fixation reaction catalyzed by pyruvate carboxylase to generate oxaloacetate (OAA) from pyruvate. Condensation of the OAA with acetyl CoA from a second molecule of pyruvate forms a “new” molecule of citrate, a 6-carbon ...
respiration
... 2b Differences between burning and respiration: vi In burning, energy is released in the forms of heat and light while in respiration, energy released is used to form ATP while some is lost as heat . ...
... 2b Differences between burning and respiration: vi In burning, energy is released in the forms of heat and light while in respiration, energy released is used to form ATP while some is lost as heat . ...
LipidMetabolism
... NADPH-reduce this to sphinganine Acetylate the amine group to make Nacylsphinganine Beta-unsaturate the palmitoyl group to make ceramide, the basis for all other sphingolipids Lipid Metabolism ...
... NADPH-reduce this to sphinganine Acetylate the amine group to make Nacylsphinganine Beta-unsaturate the palmitoyl group to make ceramide, the basis for all other sphingolipids Lipid Metabolism ...
6-22 Reaction centres - McGraw Hill Higher Education
... Photosynthetic plant cells differ from animal cells because: a) they don’t contain mitochondria. b) ATP for all cellular processes is produced by chemiosomosis in the chloroplast. c) they do not contain enzymes. d) they are capable of producing carbohydrate from light energy, that can be metabolised ...
... Photosynthetic plant cells differ from animal cells because: a) they don’t contain mitochondria. b) ATP for all cellular processes is produced by chemiosomosis in the chloroplast. c) they do not contain enzymes. d) they are capable of producing carbohydrate from light energy, that can be metabolised ...
Microbial metabolism
Microbial metabolism is the means by which a microbe obtains the energy and nutrients (e.g. carbon) it needs to live and reproduce. Microbes use many different types of metabolic strategies and species can often be differentiated from each other based on metabolic characteristics. The specific metabolic properties of a microbe are the major factors in determining that microbe’s ecological niche, and often allow for that microbe to be useful in industrial processes or responsible for biogeochemical cycles.== Types of microbial metabolism ==All microbial metabolisms can be arranged according to three principles:1. How the organism obtains carbon for synthesising cell mass: autotrophic – carbon is obtained from carbon dioxide (CO2) heterotrophic – carbon is obtained from organic compounds mixotrophic – carbon is obtained from both organic compounds and by fixing carbon dioxide2. How the organism obtains reducing equivalents used either in energy conservation or in biosynthetic reactions: lithotrophic – reducing equivalents are obtained from inorganic compounds organotrophic – reducing equivalents are obtained from organic compounds3. How the organism obtains energy for living and growing: chemotrophic – energy is obtained from external chemical compounds phototrophic – energy is obtained from lightIn practice, these terms are almost freely combined. Typical examples are as follows: chemolithoautotrophs obtain energy from the oxidation of inorganic compounds and carbon from the fixation of carbon dioxide. Examples: Nitrifying bacteria, Sulfur-oxidizing bacteria, Iron-oxidizing bacteria, Knallgas-bacteria photolithoautotrophs obtain energy from light and carbon from the fixation of carbon dioxide, using reducing equivalents from inorganic compounds. Examples: Cyanobacteria (water (H2O) as reducing equivalent donor), Chlorobiaceae, Chromatiaceae (hydrogen sulfide (H2S) as reducing equivalent donor), Chloroflexus (hydrogen (H2) as reducing equivalent donor) chemolithoheterotrophs obtain energy from the oxidation of inorganic compounds, but cannot fix carbon dioxide (CO2). Examples: some Thiobacilus, some Beggiatoa, some Nitrobacter spp., Wolinella (with H2 as reducing equivalent donor), some Knallgas-bacteria, some sulfate-reducing bacteria chemoorganoheterotrophs obtain energy, carbon, and reducing equivalents for biosynthetic reactions from organic compounds. Examples: most bacteria, e. g. Escherichia coli, Bacillus spp., Actinobacteria photoorganoheterotrophs obtain energy from light, carbon and reducing equivalents for biosynthetic reactions from organic compounds. Some species are strictly heterotrophic, many others can also fix carbon dioxide and are mixotrophic. Examples: Rhodobacter, Rhodopseudomonas, Rhodospirillum, Rhodomicrobium, Rhodocyclus, Heliobacterium, Chloroflexus (alternatively to photolithoautotrophy with hydrogen)