CHE 1401 - Fall 2013 - Chapter 7 Homework 7 (Chapter 7: Periodic
... A) alkali metals have lower densities B) alkali metals have greater electron affinities C) alkali metals have lower ionization energies D) alkali metals have lower melting points E) alkali metals are not more reactive than alkaline earth metals ...
... A) alkali metals have lower densities B) alkali metals have greater electron affinities C) alkali metals have lower ionization energies D) alkali metals have lower melting points E) alkali metals are not more reactive than alkaline earth metals ...
1st Term Review
... 14. Based on the gold foil experiment, what did Rutherford conclude about the atom? 15. An atom of chromium-60 contains how many protons, neutron and electrons? 16. What is the difference between a compound and an element? 17. What is the electron configuration of a neutral calcium atom? 18. Atomic ...
... 14. Based on the gold foil experiment, what did Rutherford conclude about the atom? 15. An atom of chromium-60 contains how many protons, neutron and electrons? 16. What is the difference between a compound and an element? 17. What is the electron configuration of a neutral calcium atom? 18. Atomic ...
Classification of Matter
... Also the original solid (HgO) and the product (Hg) are not the same colour. HgO is red and Hg is shiny and silvery. We have gas escaping (as suggested by the loss in solid mass: 432 vs. 400g) and a solid that is different from the original (difference in colour); the combination of these two observ ...
... Also the original solid (HgO) and the product (Hg) are not the same colour. HgO is red and Hg is shiny and silvery. We have gas escaping (as suggested by the loss in solid mass: 432 vs. 400g) and a solid that is different from the original (difference in colour); the combination of these two observ ...
Name Objective 1: Matter and Energy C3H8 + 5O2 → 3CO2 + 4H2O
... a. It has mass but no charge. b. It has no mass and a positive charge. c. It has a large mass and a negative charge. d. It has no mass and an equal number of positive and negative charges. ...
... a. It has mass but no charge. b. It has no mass and a positive charge. c. It has a large mass and a negative charge. d. It has no mass and an equal number of positive and negative charges. ...
February 25 – Periodicity
... • under proper conditions, these gases can combine with very reactive elements such as fluorine ...
... • under proper conditions, these gases can combine with very reactive elements such as fluorine ...
Topic 4: Classifying Elements What did the early chemists use to
... • NH3(g) à nitrogen trihydride or ammonia • CH4(g) à carbon tetrahydride or methane • H2O2(l) à dihydrogen monoxide or water A MOLECULAR COMPOUND can contain what two combinations of elements? Non-‐metal + ...
... • NH3(g) à nitrogen trihydride or ammonia • CH4(g) à carbon tetrahydride or methane • H2O2(l) à dihydrogen monoxide or water A MOLECULAR COMPOUND can contain what two combinations of elements? Non-‐metal + ...
22 diatomic molecules
... Diatomic molecules Molecules made up of only two atoms are called diatomic molecules (two atom), e.g. hydrogen chloride, HCl (one carbon atom and one chlorine atom), and carbon monoxide, CO, (one carbon atom and one oxygen atom). Certain elements normally exist as diatomic molecules. Since diatomic ...
... Diatomic molecules Molecules made up of only two atoms are called diatomic molecules (two atom), e.g. hydrogen chloride, HCl (one carbon atom and one chlorine atom), and carbon monoxide, CO, (one carbon atom and one oxygen atom). Certain elements normally exist as diatomic molecules. Since diatomic ...
SCH3U - Norbraten
... Write the word and skeletal equation for the following reactions. Be sure to include the proper states of matter by using the appropriate subscripts. You do not need to balance the equation. If the reaction does not occur write NO REACTION (NR) 10. Magnesium metal burns in air to produce solid magne ...
... Write the word and skeletal equation for the following reactions. Be sure to include the proper states of matter by using the appropriate subscripts. You do not need to balance the equation. If the reaction does not occur write NO REACTION (NR) 10. Magnesium metal burns in air to produce solid magne ...
View PDF
... 1. Using Tables and Graphs Which of the elements shown in Figure 5-2 are in the same period? 2. Classifying Which element in Figure 5-2 is a transition metal? Which is a noble gas? 3. Using Tables and Graphs Which elements in Figure 5-2 have the same number of valence electrons? How do you know? 4. ...
... 1. Using Tables and Graphs Which of the elements shown in Figure 5-2 are in the same period? 2. Classifying Which element in Figure 5-2 is a transition metal? Which is a noble gas? 3. Using Tables and Graphs Which elements in Figure 5-2 have the same number of valence electrons? How do you know? 4. ...
Unit 1: Building Blocks Homework
... Chlorine has a greater attraction than hydrogen for the bonded electrons in a hydrogen chloride molecule. What term is used to describe this type of covalent bond? ...
... Chlorine has a greater attraction than hydrogen for the bonded electrons in a hydrogen chloride molecule. What term is used to describe this type of covalent bond? ...
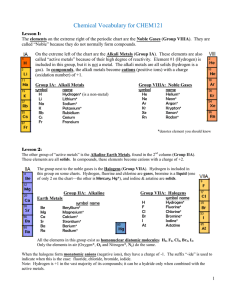
Vocabulary CHEM121
... The elements on the extreme right of the periodic chart are the Noble Gases (Group VIIIA). They are called “Noble” because they do not normally form compounds. On the extreme left of the chart are the Alkali Metals (Group IA). These elements are also called “active metals” because of their high degr ...
... The elements on the extreme right of the periodic chart are the Noble Gases (Group VIIIA). They are called “Noble” because they do not normally form compounds. On the extreme left of the chart are the Alkali Metals (Group IA). These elements are also called “active metals” because of their high degr ...
- sartep.com
... gases or the inert elements is — a. A b. B _ c. C d. D 9. The Lewis electron dot system represents electrons in the — a. outer energy level _ b. inner level c. middle level d. core level 10. The elements that are characterized by having only five electrons in the p sublevel belong to which family of ...
... gases or the inert elements is — a. A b. B _ c. C d. D 9. The Lewis electron dot system represents electrons in the — a. outer energy level _ b. inner level c. middle level d. core level 10. The elements that are characterized by having only five electrons in the p sublevel belong to which family of ...
Welcome to AP Chemistry!
... 1. The oxidation number of any uncombined element is O. 2. The oxidation number of a monatomic ion equal the charge on the ion. 3. The more electronegative element in a binary compound is assigned the number equal to the charge it would have if it were an ion. 4. The oxidation number of fluorine in ...
... 1. The oxidation number of any uncombined element is O. 2. The oxidation number of a monatomic ion equal the charge on the ion. 3. The more electronegative element in a binary compound is assigned the number equal to the charge it would have if it were an ion. 4. The oxidation number of fluorine in ...
Elements
... Generic form – referring to the atoms of element in various forms and combinations (e.g. the human body contains a lot of the element Oxygen) ...
... Generic form – referring to the atoms of element in various forms and combinations (e.g. the human body contains a lot of the element Oxygen) ...
Subatomic Particles
... • Atoms of the same _________ that have the same number of ________ (p+) but different numbers of ________ (n°) are known as _________ of that element. • _________ of an element are represented b adding the number that indicates the ___________ (A) of hat isotope to the ...
... • Atoms of the same _________ that have the same number of ________ (p+) but different numbers of ________ (n°) are known as _________ of that element. • _________ of an element are represented b adding the number that indicates the ___________ (A) of hat isotope to the ...
17.2.3 Interhalogen compounds(65-67)
... In general the compounds have properties intermediate between those of the parent halogens, though a combination of aggressive chemical reactivity and/or thermal instability militates against the determination of physical properties such as mp, bp, etc., in some instances. However, even for such hig ...
... In general the compounds have properties intermediate between those of the parent halogens, though a combination of aggressive chemical reactivity and/or thermal instability militates against the determination of physical properties such as mp, bp, etc., in some instances. However, even for such hig ...
Reading the Periodic Table
... •"halogen" means "salt-former" and compounds containing halogens are called "salts" •exist in all three states of matter: •Solid- Iodine, Astatine •Liquid- Bromine •Gas- Fluorine, Chlorine ...
... •"halogen" means "salt-former" and compounds containing halogens are called "salts" •exist in all three states of matter: •Solid- Iodine, Astatine •Liquid- Bromine •Gas- Fluorine, Chlorine ...
What is hydrogen peroxide?
... substance, said to be capable of turning base metals , especially lead , into gold; it was also sometimes believed to be an elixir of life , useful for rejuvenation and possibly ...
... substance, said to be capable of turning base metals , especially lead , into gold; it was also sometimes believed to be an elixir of life , useful for rejuvenation and possibly ...
noble gases
... Ex. silicon, boron, germanium, arsenic, selenium, antimony, tellurium, polonium, and astatine. ...
... Ex. silicon, boron, germanium, arsenic, selenium, antimony, tellurium, polonium, and astatine. ...
Final Exam Review – Free Response Section Name: 1. A sample of
... If it is a single displacement reaction, use the activity series to determine if it will occur. If it will not occur, write “no reaction.” If it is a double displacement, use the solubility rules to determine if it will occur. If it will not occur, write “no reaction.” If it will occur, write the ba ...
... If it is a single displacement reaction, use the activity series to determine if it will occur. If it will not occur, write “no reaction.” If it is a double displacement, use the solubility rules to determine if it will occur. If it will not occur, write “no reaction.” If it will occur, write the ba ...
Science 9
... in a 100-g beaker, a student added 25 g of lead (II) nitrate to 15 g of sodium iodide. In her notebook, the student recorded the final mass of the products, it was 140 g. Did this reaction conserve mass? Explain your answer. ...
... in a 100-g beaker, a student added 25 g of lead (II) nitrate to 15 g of sodium iodide. In her notebook, the student recorded the final mass of the products, it was 140 g. Did this reaction conserve mass? Explain your answer. ...
Word and Skeleton Equations Practice (ws Fall 2010)
... Write the WORD and SKELETON equations for each chemical reaction. Indicate the state of each reactant and product in the skeleton equation. Remember that the following seven elements are diatomic: hydrogen, H2(g); nitrogen, N2(g); oxygen O2(g); fluorine, F2(g); chlorine Cl2(g); bromine, Br2(l); and ...
... Write the WORD and SKELETON equations for each chemical reaction. Indicate the state of each reactant and product in the skeleton equation. Remember that the following seven elements are diatomic: hydrogen, H2(g); nitrogen, N2(g); oxygen O2(g); fluorine, F2(g); chlorine Cl2(g); bromine, Br2(l); and ...
A guided tour of the periodic table The periodic table groups
... What is the mass number? ………………………………………………………………………………………..……………. If fluorine has 9 p and 10 n, then the mass number of flurorine = ……………………………………. If oxygen has 8p and 8 n, then the mass number of oxygen = …………………………………. Atoms of an element always have the same atomic number, but they can have dif ...
... What is the mass number? ………………………………………………………………………………………..……………. If fluorine has 9 p and 10 n, then the mass number of flurorine = ……………………………………. If oxygen has 8p and 8 n, then the mass number of oxygen = …………………………………. Atoms of an element always have the same atomic number, but they can have dif ...
specimen
... 3 Chemists have developed models for bonding and structure. These models are used to explain different properties of metals and non-metals. (a) (i) Draw a labelled diagram to show the currently accepted model for metallic bonding. ...
... 3 Chemists have developed models for bonding and structure. These models are used to explain different properties of metals and non-metals. (a) (i) Draw a labelled diagram to show the currently accepted model for metallic bonding. ...
sch3u unit 1 test: matter
... 26. ___ Barium hydroxide produced in a double displacement reaction will precipitate out. 27. ___ Hydrogen is in the activity series because it classifies as a metal. SECTION B: THINKING/INQUIRY (30 marks) 1. Draw the following Lewis symbols/Lewis structures (2 marks each) a) oxygen atom b) chloride ...
... 26. ___ Barium hydroxide produced in a double displacement reaction will precipitate out. 27. ___ Hydrogen is in the activity series because it classifies as a metal. SECTION B: THINKING/INQUIRY (30 marks) 1. Draw the following Lewis symbols/Lewis structures (2 marks each) a) oxygen atom b) chloride ...