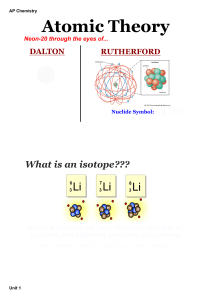
Midterm Review.ppt - Chemistry R: 4(AE)
... considered in order from top to bottom, the electronegativity of each successive element 1. decreases 2. increases 3. remains the same ...
... considered in order from top to bottom, the electronegativity of each successive element 1. decreases 2. increases 3. remains the same ...
Worksheet 1, UNIT THREE
... __________________________________________ than the last one. 3. As you move from left to right across a period on the periodic table the size of an atom will __________________________________________. 4. This happens because as you move across a period each element has one more _______________ in ...
... __________________________________________ than the last one. 3. As you move from left to right across a period on the periodic table the size of an atom will __________________________________________. 4. This happens because as you move across a period each element has one more _______________ in ...
Organic Functional Groups: Halocarbons
... • The halogens are in the second column from the right. • Astatine (At) is not included: it is unstable and extremely rare. • Halogens each have seven valence electrons – three more than carbon. • Halogens are very hungry to gain an 8th electron. When they do, the eight electrons collectively drop i ...
... • The halogens are in the second column from the right. • Astatine (At) is not included: it is unstable and extremely rare. • Halogens each have seven valence electrons – three more than carbon. • Halogens are very hungry to gain an 8th electron. When they do, the eight electrons collectively drop i ...
Unit 3 Practice Test
... A. hydrogen bond B. dipole-dipole force C. London dispersion force D. all of the above _______________________________________________________ 27. Using the theoretical model of non-metals, the reaction to form chloride from chlorine gas is A. Cl (g) + e− C. 2Cl−(aq) ...
... A. hydrogen bond B. dipole-dipole force C. London dispersion force D. all of the above _______________________________________________________ 27. Using the theoretical model of non-metals, the reaction to form chloride from chlorine gas is A. Cl (g) + e− C. 2Cl−(aq) ...
SNC2D – Science 10 Tuesday April 26th, 2010 Mr. Sourlis and Mr
... 6. What is the correct chemical formula for Calcium Nitride? a. CaN b. Ca2N c. Ca2N3 d. Ca3N2 e. Ca(NO3)2 7. Which of the following metals does NOT have more than one possible ion charge? a. Mn b. Ag c. Fe d. Cu e. Sn 8. What is the name of the compound PbSe? a. Lead (II) Selenide b. Lead (IV) Selen ...
... 6. What is the correct chemical formula for Calcium Nitride? a. CaN b. Ca2N c. Ca2N3 d. Ca3N2 e. Ca(NO3)2 7. Which of the following metals does NOT have more than one possible ion charge? a. Mn b. Ag c. Fe d. Cu e. Sn 8. What is the name of the compound PbSe? a. Lead (II) Selenide b. Lead (IV) Selen ...
Slide 1
... considered in order from top to bottom, the electronegativity of each successive element 1. decreases 2. increases 3. remains the same ...
... considered in order from top to bottom, the electronegativity of each successive element 1. decreases 2. increases 3. remains the same ...
Activity 17 Follow-up
... Activity 17 Analysis 3. Was it possible for an atom to make more than one bond? Explain, and give an ...
... Activity 17 Analysis 3. Was it possible for an atom to make more than one bond? Explain, and give an ...
Year 10 Chemistry Exam June 2012
... 5. What information does the group the element is in the periodic table tell you about the electronic configuration? __________________________________________________________________________ __________________________________________________________________________ 1 mark 6. For the ionic substance ...
... 5. What information does the group the element is in the periodic table tell you about the electronic configuration? __________________________________________________________________________ __________________________________________________________________________ 1 mark 6. For the ionic substance ...
Unit A Remediation Review
... 12. What are five clues that will allow you to conclude that a chemical change has occurred? 13. Describe what occurs in the following reaction types, the general equation and an example for each: a) Formation b) Decomposition c) Single Replacement d) Double Replacement e) Combustion 14. Write a bal ...
... 12. What are five clues that will allow you to conclude that a chemical change has occurred? 13. Describe what occurs in the following reaction types, the general equation and an example for each: a) Formation b) Decomposition c) Single Replacement d) Double Replacement e) Combustion 14. Write a bal ...
Metals and non-metals III IMPORTANT POINTS Non-metals
... 1. a. Magnesium, chromium and sodium are all metals, hence, they react with oxygen to form basic oxides b. Chromium, as it is a transition metal. Metals have high density and coloured compounds are formed by transition metals. c. Bromine - the formula is Br2, that is, two atoms of bromine. d. Bromin ...
... 1. a. Magnesium, chromium and sodium are all metals, hence, they react with oxygen to form basic oxides b. Chromium, as it is a transition metal. Metals have high density and coloured compounds are formed by transition metals. c. Bromine - the formula is Br2, that is, two atoms of bromine. d. Bromin ...
CHEM1411,chapter 1-2-3 exercises 1. In 1828, the diameter of the
... 5. A piece of a metal alloy with a mass of 114 g was placed into a graduated cylinder that contained 25.0 mL of water, raising the water level to 42.5 mL. What is the density of the metal? A) 0.154 g/cm3 B) 0.592 g/cm3 C) 2.68 g/cm3 D) 6.51 g/cm3 E) 7.25 g/cm3 ...
... 5. A piece of a metal alloy with a mass of 114 g was placed into a graduated cylinder that contained 25.0 mL of water, raising the water level to 42.5 mL. What is the density of the metal? A) 0.154 g/cm3 B) 0.592 g/cm3 C) 2.68 g/cm3 D) 6.51 g/cm3 E) 7.25 g/cm3 ...
Ch 2-1 Properties of Matter
... 71) A gas may be released during a physical change. For example, bubbles form when water boils. 72) The wax appears to disappear because the products of the reaction—carbon dioxide and water vapor—are colorless. 79) a) yes; because the graph is a straight line, the proportion of iron to oxygen is a ...
... 71) A gas may be released during a physical change. For example, bubbles form when water boils. 72) The wax appears to disappear because the products of the reaction—carbon dioxide and water vapor—are colorless. 79) a) yes; because the graph is a straight line, the proportion of iron to oxygen is a ...
CHEMISTRY 1 FINAL EXAM REVIEW
... 3.) List each as being a physical change or a chemical change: butter melting, butter burning, sugar dissolving in water, a sandwich getting digested. 4.) List each as being a physical or a chemical property: copper sulfate is blue, iron is a solid, water o ...
... 3.) List each as being a physical change or a chemical change: butter melting, butter burning, sugar dissolving in water, a sandwich getting digested. 4.) List each as being a physical or a chemical property: copper sulfate is blue, iron is a solid, water o ...
Chemical Reactions
... • Substance formed from positive ion of a base and the negative ion of an acid – When an acid and a base are combined, they produce a salt and water • HCl + NaOH NaCl + H2O Acid ...
... • Substance formed from positive ion of a base and the negative ion of an acid – When an acid and a base are combined, they produce a salt and water • HCl + NaOH NaCl + H2O Acid ...
Snc2d Chapter 5 Practice Test
... 16. Potassium carbonate solution reacts with aluminum chloride solution producing two different compounds. ...
... 16. Potassium carbonate solution reacts with aluminum chloride solution producing two different compounds. ...
Writing Chemical Equations KClO3 O2 (g) + KCl (s) Balancing
... In this example we started with 2 atoms of hydrogen and we ended with 2 atoms of hydrogen. But we started with only one atom of oxygen and ended with 2 atoms of oxygen. We cannot change the numbers at the bottom of the symbols because that is how the formulas must be written. To balance the equation ...
... In this example we started with 2 atoms of hydrogen and we ended with 2 atoms of hydrogen. But we started with only one atom of oxygen and ended with 2 atoms of oxygen. We cannot change the numbers at the bottom of the symbols because that is how the formulas must be written. To balance the equation ...
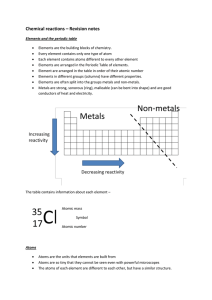
Chemical reactions revision
... Elements are the building blocks of chemistry. Every element contains only one type of atom Each element contains atoms different to every other element Elements are arranged in the Periodic Table of elements. Element are arranged in the table in order of their atomic number Elements in different gr ...
... Elements are the building blocks of chemistry. Every element contains only one type of atom Each element contains atoms different to every other element Elements are arranged in the Periodic Table of elements. Element are arranged in the table in order of their atomic number Elements in different gr ...
Chemical Bonds
... compounds An atom is chemically stable when it has a complete outer energy level ...
... compounds An atom is chemically stable when it has a complete outer energy level ...
Unit 9 The p-Block Elements
... The Group VII elements, the halogens, are reactive non-metals. They have the electronic structure ns2 np5 for their valence shells. Halogens exist as diatomic molecules, X2. The first element in the group, fluorine, is anomalous, just as lithium and beryllium are in Group I and II respectively. Asta ...
... The Group VII elements, the halogens, are reactive non-metals. They have the electronic structure ns2 np5 for their valence shells. Halogens exist as diatomic molecules, X2. The first element in the group, fluorine, is anomalous, just as lithium and beryllium are in Group I and II respectively. Asta ...
ATOMIC STRUCTURE - IGCSE STUDY BANK
... Except for the noble gases, the more electrons in the outer shell the more non-metallic and the more reactive the element. The most reactive non-metals only need to share/gain one or two electrons. The most reactive metals only have 1 or 2 electrons in the outer shell which tend to be easily los ...
... Except for the noble gases, the more electrons in the outer shell the more non-metallic and the more reactive the element. The most reactive non-metals only need to share/gain one or two electrons. The most reactive metals only have 1 or 2 electrons in the outer shell which tend to be easily los ...
6-Getting to Know the Periodic Table
... Metals, Alkaline Earth Metals, Halogens and Noble Gases. 2) Using red ink, show the lewis dot structure for groups 1-2, 13-18. 3) Using black ink OR pencil, write in the period number for each period, and group number for each group. 4) Answer each of the following questions: a. Which element has 27 ...
... Metals, Alkaline Earth Metals, Halogens and Noble Gases. 2) Using red ink, show the lewis dot structure for groups 1-2, 13-18. 3) Using black ink OR pencil, write in the period number for each period, and group number for each group. 4) Answer each of the following questions: a. Which element has 27 ...
Review Package
... a) Calcium metal reacts with hydrobromic acid to form aqueous calcium bromide and hydrogen gas. ...
... a) Calcium metal reacts with hydrobromic acid to form aqueous calcium bromide and hydrogen gas. ...
1.5.16(Chem) - mrcarlsonschemistryclass
... Cations and Anions • Cations are ions with a POSITIVE charge. • Anions are ions with a NEGATIVE charge. • Draw the funny way to remember cations and anions: ...
... Cations and Anions • Cations are ions with a POSITIVE charge. • Anions are ions with a NEGATIVE charge. • Draw the funny way to remember cations and anions: ...
Chemistry Unit Review
... b. When sugar (C12H22O11) and sulfuric acid (H2SO4) are combined, carbon, water, and sulfur dioxide are formed. ...
... b. When sugar (C12H22O11) and sulfuric acid (H2SO4) are combined, carbon, water, and sulfur dioxide are formed. ...