* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Classification of Matter
Organic chemistry wikipedia , lookup
Crystallization wikipedia , lookup
Registration, Evaluation, Authorisation and Restriction of Chemicals wikipedia , lookup
Nuclear transmutation wikipedia , lookup
Stoichiometry wikipedia , lookup
Spinodal decomposition wikipedia , lookup
Safety data sheet wikipedia , lookup
Water splitting wikipedia , lookup
History of molecular theory wikipedia , lookup
Hypervalent molecule wikipedia , lookup
Water pollution wikipedia , lookup
Fluorochemical industry wikipedia , lookup
Periodic table wikipedia , lookup
Electrolysis of water wikipedia , lookup
Gas chromatography wikipedia , lookup
Drug discovery wikipedia , lookup
Inorganic chemistry wikipedia , lookup
Freshwater environmental quality parameters wikipedia , lookup
Chemical thermodynamics wikipedia , lookup
Chemical element wikipedia , lookup
Extended periodic table wikipedia , lookup
Abundance of the chemical elements wikipedia , lookup
Gas chromatography–mass spectrometry wikipedia , lookup
History of chemistry wikipedia , lookup
Atomic theory wikipedia , lookup
IUPAC nomenclature of inorganic chemistry 2005 wikipedia , lookup
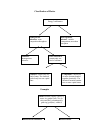
Classification of Matter Matter: anything with mass. Energy is not matter Homogeneous substance: same composition throughout Solutions: homogeneous mixtures Heterogeneous mixture: variable composition; more than one phase Pure Substances: contain only one kind of substance Elements: contain only one kind of atom. The chemical symbol only has one capital letter Compounds: contain two or more elements bonded together chemically. The chemical formula contains two or more capital letters Examples Matter: banana, sugar(C12H22O11), water, air, granite rock, CO2, O2, salt water(H2O), aluminum(Al), gold ring, gold(Au), sulfur(S) Solutions: homogeneous mixtures: air, salt water, gold ring Elements: O2, aluminum(Al), gold(Au), sulfur(S) Pure Substances: sugar(C12H22O11), water(H2O), CO2, O2, aluminum(Al), gold(Au), sulfur(S) Compounds: sugar(C12H22O11), water (H2O), CO2, Elements Versus Compounds Just what is the difference between a compound and an element? There are over 114 known elements: these are listed in the periodic table, and they make up all matter in the universe: rocks, stars, dust and living beings. Each element is made up of only one kind of atom, meaning that each atom has a specific number of protons. As mentioned, elements have to react chemically to form compounds. As atoms bond, energy is either released or absorbed. The reverse can also take place. When a compound decomposes, we often see its two components released, and if one of them is a gas, the leftover solid or liquid will weigh less than the original solid or liquid that decomposed. Example: 2 HgO(s)--> 2Hg(s) + O2(g) 432 g --> 400 g + 32g Also the original solid (HgO) and the product (Hg) are not the same colour. HgO is red and Hg is shiny and silvery. We have gas escaping (as suggested by the loss in solid mass: 432 vs. 400g) and a solid that is different from the original (difference in colour); the combination of these two observations suggest a chemical change.(see later notes) If we had iodine going from a shiny solid to a purple gas, we would again observe an apparent loss in solid mass, but the solid left behind would not be of a different colour. Also in this physical change(sublimation) the iodine gas would turn back into shiny iodine crystals upon cooling. In the chemical reaction involving HgO, the oxygen gas escaping would not turn back into red HgO powder.