Boundless Study Slides
... • Ions are formed when the number of protons in an atom does not equal the number of electrons. If more protons are present, the ion is positive and is known as a cation; if more electrons are present, the ion is negative and referred to as an anion. • Ions are highly reactive species. They are gene ...
... • Ions are formed when the number of protons in an atom does not equal the number of electrons. If more protons are present, the ion is positive and is known as a cation; if more electrons are present, the ion is negative and referred to as an anion. • Ions are highly reactive species. They are gene ...
Chemistry Midterm Exam Review
... ____ 46. A change in the force of gravity on an object will affect its a. mass. c. weight. b. density. d. kinetic energy. ____ 47. Which of these is a measure of the amount of material? a. density c. volume b. weight d. mass ____ 48. Which of these statements about mass is true? a. Mass is expressed ...
... ____ 46. A change in the force of gravity on an object will affect its a. mass. c. weight. b. density. d. kinetic energy. ____ 47. Which of these is a measure of the amount of material? a. density c. volume b. weight d. mass ____ 48. Which of these statements about mass is true? a. Mass is expressed ...
ch final rvw
... Earth is made to illustrate which of the following? a. that atoms are indivisible b. that atoms are very small c. that atoms are very large d. that in a copper penny, there is one atom for every person on Earth The range in size of most atomic radii is approximately ____. a. 2 to 5 cm c. 5 10 m to 2 ...
... Earth is made to illustrate which of the following? a. that atoms are indivisible b. that atoms are very small c. that atoms are very large d. that in a copper penny, there is one atom for every person on Earth The range in size of most atomic radii is approximately ____. a. 2 to 5 cm c. 5 10 m to 2 ...
Fall Practice Final
... a. the first b. the second c. the third d. All the electrons are equally difficult to remove. ____ 99. Which of the following elements has the lowest electronegativity? a. lithium c. bromine b. carbon d. fluorine ____ 100. Which statement is true about electronegativity? a. Electronegativity is the ...
... a. the first b. the second c. the third d. All the electrons are equally difficult to remove. ____ 99. Which of the following elements has the lowest electronegativity? a. lithium c. bromine b. carbon d. fluorine ____ 100. Which statement is true about electronegativity? a. Electronegativity is the ...
Chemistry MCQS 12 class
... 26. The amount of energy required to remove an electron from an atom of an element in the ...
... 26. The amount of energy required to remove an electron from an atom of an element in the ...
By Martin S. Silberberg
... 66 The behavior of several real gases with increasing external pressure Figure 5.21 67 The effect of intermolecular attractions on measured gas pressure Figure 5.22 68 The effect of molecular volume on measured gas volume Figure 5.23 69 Using gas laws to determine a balanced equation Sample and Foll ...
... 66 The behavior of several real gases with increasing external pressure Figure 5.21 67 The effect of intermolecular attractions on measured gas pressure Figure 5.22 68 The effect of molecular volume on measured gas volume Figure 5.23 69 Using gas laws to determine a balanced equation Sample and Foll ...
enjoy chemistry
... The elements present in Group 18 have their valence shell orbitals completely filled and, therefore, react with a few elements only under certain conditions. Therefore, they are now known as noble gases. (ii)Noble gases are mostly chemically inert. Their inertness to chemical reactivity is attribute ...
... The elements present in Group 18 have their valence shell orbitals completely filled and, therefore, react with a few elements only under certain conditions. Therefore, they are now known as noble gases. (ii)Noble gases are mostly chemically inert. Their inertness to chemical reactivity is attribute ...
Science 9 Year End Review The following information includes all
... What did the early chemists use to represent elements? ...
... What did the early chemists use to represent elements? ...
PS 2 - Purdyphysicalscience
... Use the atomic number and the mass number to calculate the number of protons, neutrons, and/or electrons for a given isotope of an element Predict the charge that a representative element will acquire according to the arrangement of electrons in its outer energy level Compare fission and fusion (inc ...
... Use the atomic number and the mass number to calculate the number of protons, neutrons, and/or electrons for a given isotope of an element Predict the charge that a representative element will acquire according to the arrangement of electrons in its outer energy level Compare fission and fusion (inc ...
Matter Flashcards 5 - Henrico County Public Schools
... forms is water (H2 O). The number of oxygen atoms is __________. SOL 5.4 ...
... forms is water (H2 O). The number of oxygen atoms is __________. SOL 5.4 ...
Major 01 - KFUPM Faculty List
... should know. Thus look for the unknown symbol and you get A as the correct choice. B. He: you should know that this denotes helium (from helios, Greek for the sun, because it was first found in the sun spectroscopically). C. Na: you should know that this denotes sodium (from the German name Natrium) ...
... should know. Thus look for the unknown symbol and you get A as the correct choice. B. He: you should know that this denotes helium (from helios, Greek for the sun, because it was first found in the sun spectroscopically). C. Na: you should know that this denotes sodium (from the German name Natrium) ...
Document
... flame test. When it was dissolved in water and reacted with NaOCl, a purple color appeared. The unknown is: a. b. c. d. ...
... flame test. When it was dissolved in water and reacted with NaOCl, a purple color appeared. The unknown is: a. b. c. d. ...
Brilliant Preparatory Section, Sitamarhi
... viii. All the reactants and products should be written as molecules including the elements like hydrogen, oxygen, nitrogen, fluorine chlorine, bromine and iodine as H2, O2, N2, F2, Cl2, Br2 and I2. ...
... viii. All the reactants and products should be written as molecules including the elements like hydrogen, oxygen, nitrogen, fluorine chlorine, bromine and iodine as H2, O2, N2, F2, Cl2, Br2 and I2. ...
Chem Course Desc2. New
... 2.4 Use the Periodic table to identify metals, semimetals (metalloids), nonmetals, alkali metals, alkaline earth metals, halogens and transition metals. (C. S. 1. b, c ) 2.5 Relate the position of an element in the periodic table to its quantum electron configuration. ( C.S. 1.g ) ...
... 2.4 Use the Periodic table to identify metals, semimetals (metalloids), nonmetals, alkali metals, alkaline earth metals, halogens and transition metals. (C. S. 1. b, c ) 2.5 Relate the position of an element in the periodic table to its quantum electron configuration. ( C.S. 1.g ) ...
Chapters 1 to 5 - Lakeland Regional High School
... d. the electron must make a transition from a higher to a lower energy level. ____ 30. The French scientist Louis de Broglie theorized that a. electrons could have a dual wave-particle nature. b. light waves did not have a dual wave-particle nature. c. the natures of light and quantized electron orb ...
... d. the electron must make a transition from a higher to a lower energy level. ____ 30. The French scientist Louis de Broglie theorized that a. electrons could have a dual wave-particle nature. b. light waves did not have a dual wave-particle nature. c. the natures of light and quantized electron orb ...
Introduction to Inorganic Chemistry
... 2 The relative importance of different elements From an academic point of view, all elements might be said to be equally important. The chemistry of each has to be understood in detail if the chemistry of matter as a whole is to be understood in detail. From the point of view of the world at large, ...
... 2 The relative importance of different elements From an academic point of view, all elements might be said to be equally important. The chemistry of each has to be understood in detail if the chemistry of matter as a whole is to be understood in detail. From the point of view of the world at large, ...
Introduction to Inorganic Chemistry
... 2 The relative importance of different elements From an academic point of view, all elements might be said to be equally important. The chemistry of each has to be understood in detail if the chemistry of matter as a whole is to be understood in detail. From the point of view of the world at large, ...
... 2 The relative importance of different elements From an academic point of view, all elements might be said to be equally important. The chemistry of each has to be understood in detail if the chemistry of matter as a whole is to be understood in detail. From the point of view of the world at large, ...
Final Exam
... long, 2.50 cm wide, and 1.00 cm thick. What is a possible identity of the element? a. nickel, 8.90 g/cm3 b. aluminum, 2.70 g/cm3 c. titanium, 4.54 g/cm3 d. zirconium, 6.51 g/cm3 e. chromium, 7.20 g/cm3 ...
... long, 2.50 cm wide, and 1.00 cm thick. What is a possible identity of the element? a. nickel, 8.90 g/cm3 b. aluminum, 2.70 g/cm3 c. titanium, 4.54 g/cm3 d. zirconium, 6.51 g/cm3 e. chromium, 7.20 g/cm3 ...
chemistry - My Study materials – Kumar
... All matters in the universe exist in three states. There are two ways of classification of matter. 1. According to physical state as solid, liquid or gas. 2. According to its composition as element, compound or mixture. According to this law mass of an isolated system will remain constant over time. ...
... All matters in the universe exist in three states. There are two ways of classification of matter. 1. According to physical state as solid, liquid or gas. 2. According to its composition as element, compound or mixture. According to this law mass of an isolated system will remain constant over time. ...
Periodic table
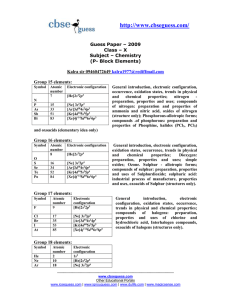
The periodic table is a tabular arrangement of the chemical elements, ordered by their atomic number (number of protons in the nucleus), electron configurations, and recurring chemical properties. The table also shows four rectangular blocks: s-, p- d- and f-block. In general, within one row (period) the elements are metals on the lefthand side, and non-metals on the righthand side.The rows of the table are called periods; the columns are called groups. Six groups (columns) have names as well as numbers: for example, group 17 elements are the halogens; and group 18, the noble gases. The periodic table can be used to derive relationships between the properties of the elements, and predict the properties of new elements yet to be discovered or synthesized. The periodic table provides a useful framework for analyzing chemical behavior, and is widely used in chemistry and other sciences.Although precursors exist, Dmitri Mendeleev is generally credited with the publication, in 1869, of the first widely recognized periodic table. He developed his table to illustrate periodic trends in the properties of the then-known elements. Mendeleev also predicted some properties of then-unknown elements that would be expected to fill gaps in this table. Most of his predictions were proved correct when the elements in question were subsequently discovered. Mendeleev's periodic table has since been expanded and refined with the discovery or synthesis of further new elements and the development of new theoretical models to explain chemical behavior.All elements from atomic numbers 1 (hydrogen) to 118 (ununoctium) have been discovered or reportedly synthesized, with elements 113, 115, 117, and 118 having yet to be confirmed. The first 94 elements exist naturally, although some are found only in trace amounts and were synthesized in laboratories before being found in nature. Elements with atomic numbers from 95 to 118 have only been synthesized in laboratories. It has been shown that einsteinium and fermium once occurred in nature but currently do not. Synthesis of elements having higher atomic numbers is being pursued. Numerous synthetic radionuclides of naturally occurring elements have also been produced in laboratories.