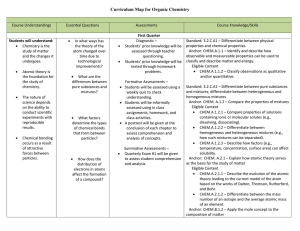
Organic Chemistry Curriculum Map - Belle Vernon Area School District
... (e.g., number of valence electrons, potential types of bonds, reactivity. Anchor: CHEM.A.2.2 – Describe the behavior of electrons in atoms. Eligible Content CHEM.A.2.2.4 – Relate the existence of quantized energy levels to atomic emission spectra. Standard: 3.2.C.A2 – Explain how atoms combine to ...
... (e.g., number of valence electrons, potential types of bonds, reactivity. Anchor: CHEM.A.2.2 – Describe the behavior of electrons in atoms. Eligible Content CHEM.A.2.2.4 – Relate the existence of quantized energy levels to atomic emission spectra. Standard: 3.2.C.A2 – Explain how atoms combine to ...
CfE Higher Chemistry Unit 1: Chemical Changes and Structure
... the substance. This is known as the 'kinetic model' of matter. In any sample of solution, liquid or gas there is a range of kinetic energies known as an energy distribution. The collision theory of reactions suggests that, for a chemical reaction to occur, particles must collide. Simple collision is ...
... the substance. This is known as the 'kinetic model' of matter. In any sample of solution, liquid or gas there is a range of kinetic energies known as an energy distribution. The collision theory of reactions suggests that, for a chemical reaction to occur, particles must collide. Simple collision is ...
Dr. Spencer`s PPT
... Paris, and although Lavoisier's father wanted him to be a lawyer, Lavoisier was fascinated by science. From the beginning of his scientific career, Lavoisier recognized the importance of accurate measurements. He wrote the first modern chemistry (1789) textbook so that it is not surprising that Lavo ...
... Paris, and although Lavoisier's father wanted him to be a lawyer, Lavoisier was fascinated by science. From the beginning of his scientific career, Lavoisier recognized the importance of accurate measurements. He wrote the first modern chemistry (1789) textbook so that it is not surprising that Lavo ...
View PDF
... Rutherford fired positively charged particles at metal foil and concluded that most of the mass of an atom was a. in the electrons. c. evenly spread throughout the atom. b. ...
... Rutherford fired positively charged particles at metal foil and concluded that most of the mass of an atom was a. in the electrons. c. evenly spread throughout the atom. b. ...
Atoms: The Building Blocks of Matter - Milton
... Section 1 covers the history and development of atomic theory, from Democritus to Dalton to the modern era. Section 2 covers the experiments that led to the discovery of the electron and the nucleus as well as the principal properties of these subatomic particles. ...
... Section 1 covers the history and development of atomic theory, from Democritus to Dalton to the modern era. Section 2 covers the experiments that led to the discovery of the electron and the nucleus as well as the principal properties of these subatomic particles. ...
Ch 1,2,4,25 pT
... a. Electrons are negatively charged and are the heaviest subatomic particle. b. Protons are positively charged and the lightest subatomic particle. c. Neutrons have no charge and are the lightest subatomic particle. d. The mass of a neutron nearly equals the mass of a proton. The particles that are ...
... a. Electrons are negatively charged and are the heaviest subatomic particle. b. Protons are positively charged and the lightest subatomic particle. c. Neutrons have no charge and are the lightest subatomic particle. d. The mass of a neutron nearly equals the mass of a proton. The particles that are ...
File
... (Isotopes, Actanides, Isobars, none of these) 13. The difference in atomic weights or mass numbers of the atoms of the same element is due to __________. (Different number of electrons in them, different number of protons in them, different number of neutrons in them, none of these) 14. A tritium nu ...
... (Isotopes, Actanides, Isobars, none of these) 13. The difference in atomic weights or mass numbers of the atoms of the same element is due to __________. (Different number of electrons in them, different number of protons in them, different number of neutrons in them, none of these) 14. A tritium nu ...
DOE Chemistry 1
... for use by DOE category A reactors. The subject areas, subject matter content, and level of detail of the Reactor Operator Fundamentals Manuals were determined from several sources. DOE Category A reactor training managers determined which materials should be included, and served as a primary refere ...
... for use by DOE category A reactors. The subject areas, subject matter content, and level of detail of the Reactor Operator Fundamentals Manuals were determined from several sources. DOE Category A reactor training managers determined which materials should be included, and served as a primary refere ...
Summer Work: Independent Packet: Basics Of Atomic Structure
... negatively charged electrons. They had reason to believe that electrons were found in specific areas (shells) outside the nucleus (although, this was being debated hotly by now). And, atoms were neutral in terms of charge, because the number of protons and electrons were equal. and by 1939, some of ...
... negatively charged electrons. They had reason to believe that electrons were found in specific areas (shells) outside the nucleus (although, this was being debated hotly by now). And, atoms were neutral in terms of charge, because the number of protons and electrons were equal. and by 1939, some of ...
幻灯片 1
... way different orbitals are filled is controlled by their energies (and hence their An atom consists of a very small positively charged nucleus, Electron and Nuclei different screening by other electrons) and by the Pauli exclusion principle. surrounded by negative electrons held by electrostatic att ...
... way different orbitals are filled is controlled by their energies (and hence their An atom consists of a very small positively charged nucleus, Electron and Nuclei different screening by other electrons) and by the Pauli exclusion principle. surrounded by negative electrons held by electrostatic att ...
File
... oxidized. Therefore, in order to prepare the pure metals, alkali metals must be produced in the absence of materials (H2O, O2) that are capable of oxidizing them. The method of preparation is electrochemical processes, specifically, electrolysis of molten chloride salts and reduction of alkali salts ...
... oxidized. Therefore, in order to prepare the pure metals, alkali metals must be produced in the absence of materials (H2O, O2) that are capable of oxidizing them. The method of preparation is electrochemical processes, specifically, electrolysis of molten chloride salts and reduction of alkali salts ...
Next Generation Sunshine State Standards Chapter 2
... B.C., humans discovered how to combine copper with tin to make bronze, a tough, hard alloy. The Bronze Age began its We begin our discussion of Earth materials with an overview decline when the ability to extract iron from minerals such as of mineralogy (mineral = mineral, ology = the study of), beh ...
... B.C., humans discovered how to combine copper with tin to make bronze, a tough, hard alloy. The Bronze Age began its We begin our discussion of Earth materials with an overview decline when the ability to extract iron from minerals such as of mineralogy (mineral = mineral, ology = the study of), beh ...
Preview Sample 2
... A. Mendeleev arranged the known elements in order of increasing relative atomic mass. B. He grouped elements with similar properties into columns and rows so that their properties varied in a regular pattern. C. He arranged the elements so that they were in increasing atomic number order. D. He was ...
... A. Mendeleev arranged the known elements in order of increasing relative atomic mass. B. He grouped elements with similar properties into columns and rows so that their properties varied in a regular pattern. C. He arranged the elements so that they were in increasing atomic number order. D. He was ...
9/10/10 1 Chemistry 121: Atomic and Molecular Chemistry
... (indicating 9 protons in the nucleus). Thus the number of neutrons in an atom of fluorine is 19 -9 = 10. The atomic number, number of neutrons, and mass number all must be positive integers Atoms of a given element do not all have the same mass. Most elements have two or more isotopes, atoms that ha ...
... (indicating 9 protons in the nucleus). Thus the number of neutrons in an atom of fluorine is 19 -9 = 10. The atomic number, number of neutrons, and mass number all must be positive integers Atoms of a given element do not all have the same mass. Most elements have two or more isotopes, atoms that ha ...
Chem 11 Review Answers - hrsbstaff.ednet.ns.ca
... In a period, the electron affinity increases as the atomic number increases. As you go across (left to right) the size of the atom decreases and the electrons are added closer to the nucleus where the attraction is greater – causing more energy to be released when the electron is added, increasing t ...
... In a period, the electron affinity increases as the atomic number increases. As you go across (left to right) the size of the atom decreases and the electrons are added closer to the nucleus where the attraction is greater – causing more energy to be released when the electron is added, increasing t ...
2 Atoms and Molecules
... In Chapter 1, we defined elements as homogeneous pure substances made up of identical atoms. At least 115 different elements are known to exist. This leads to the conclusion that a minimum of 115 different kinds of atoms exist. Eighty-eight of the elements are naturally occurring and therefore are f ...
... In Chapter 1, we defined elements as homogeneous pure substances made up of identical atoms. At least 115 different elements are known to exist. This leads to the conclusion that a minimum of 115 different kinds of atoms exist. Eighty-eight of the elements are naturally occurring and therefore are f ...
cont. - Appoquinimink High School
... • While the element iron is defined as being made up of neutral atoms with 26 protons and 26 electrons, not every iron atom has the same number of neutrons. • Atoms that have the same number of protons but different numbers of neutrons are called isotopes. (cont.) © 2004 Key Curriculum Press. ...
... • While the element iron is defined as being made up of neutral atoms with 26 protons and 26 electrons, not every iron atom has the same number of neutrons. • Atoms that have the same number of protons but different numbers of neutrons are called isotopes. (cont.) © 2004 Key Curriculum Press. ...
Atom Building - Campbell County Schools
... • Neils Bohr hypothesized that the atom was like a tiny solar system, with electrons circling the nucleus in well defined orbits like planets • The speculated paths of the electrons were called orbitals, shells, or energy ...
... • Neils Bohr hypothesized that the atom was like a tiny solar system, with electrons circling the nucleus in well defined orbits like planets • The speculated paths of the electrons were called orbitals, shells, or energy ...
Chapter 3: Elements, Compounds and the Periodic Table
... Mendeleev (Russian) & Meyer (German) in 1869 Noted repeating (periodic) properties ...
... Mendeleev (Russian) & Meyer (German) in 1869 Noted repeating (periodic) properties ...
Periodic table
The periodic table is a tabular arrangement of the chemical elements, ordered by their atomic number (number of protons in the nucleus), electron configurations, and recurring chemical properties. The table also shows four rectangular blocks: s-, p- d- and f-block. In general, within one row (period) the elements are metals on the lefthand side, and non-metals on the righthand side.The rows of the table are called periods; the columns are called groups. Six groups (columns) have names as well as numbers: for example, group 17 elements are the halogens; and group 18, the noble gases. The periodic table can be used to derive relationships between the properties of the elements, and predict the properties of new elements yet to be discovered or synthesized. The periodic table provides a useful framework for analyzing chemical behavior, and is widely used in chemistry and other sciences.Although precursors exist, Dmitri Mendeleev is generally credited with the publication, in 1869, of the first widely recognized periodic table. He developed his table to illustrate periodic trends in the properties of the then-known elements. Mendeleev also predicted some properties of then-unknown elements that would be expected to fill gaps in this table. Most of his predictions were proved correct when the elements in question were subsequently discovered. Mendeleev's periodic table has since been expanded and refined with the discovery or synthesis of further new elements and the development of new theoretical models to explain chemical behavior.All elements from atomic numbers 1 (hydrogen) to 118 (ununoctium) have been discovered or reportedly synthesized, with elements 113, 115, 117, and 118 having yet to be confirmed. The first 94 elements exist naturally, although some are found only in trace amounts and were synthesized in laboratories before being found in nature. Elements with atomic numbers from 95 to 118 have only been synthesized in laboratories. It has been shown that einsteinium and fermium once occurred in nature but currently do not. Synthesis of elements having higher atomic numbers is being pursued. Numerous synthetic radionuclides of naturally occurring elements have also been produced in laboratories.























