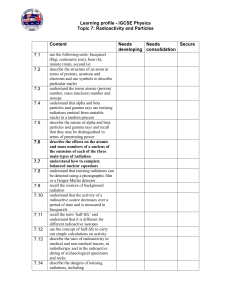
Final Exam
... heard, but as the train gets closer, the whistle gets louder. This is an example of _______. A. a sonic boom B. interference C. refraction D. the Doppler effect ...
... heard, but as the train gets closer, the whistle gets louder. This is an example of _______. A. a sonic boom B. interference C. refraction D. the Doppler effect ...
nuclear chemistry - Wood County Schools
... your textbook and is for classroom or individual use only. ...
... your textbook and is for classroom or individual use only. ...
Atomic Structure Study Guide
... high voltage vacuum tube – was made up of ____________ charged particles, which had a smaller mass/charge ratio than any known atom. This showed that ______________ particles existed, and therefore atoms were not indivisible. These particles are now known as _____________. His student, Rutherford, s ...
... high voltage vacuum tube – was made up of ____________ charged particles, which had a smaller mass/charge ratio than any known atom. This showed that ______________ particles existed, and therefore atoms were not indivisible. These particles are now known as _____________. His student, Rutherford, s ...
t 1/2
... emitted from the cathode are deflected by either an electric field or a magnetic field. The parallel plates connected to a battery provide the electric field. Two current-carrying coils (not shown) produce a magnetic field perpendicular to the electric field. The sizes of the deflections, as noted o ...
... emitted from the cathode are deflected by either an electric field or a magnetic field. The parallel plates connected to a battery provide the electric field. Two current-carrying coils (not shown) produce a magnetic field perpendicular to the electric field. The sizes of the deflections, as noted o ...
UNIT 2 CLASSIFICATION
... … place IN which a fission NUCLEAR reaction TAKES place. It contains …in THE form of rods to produce the appropriate result. The reactor consists OF a fuel, a moderator and A cooling system. An instrument …neutron WHICH strikes the nucleus of an atom of U-235. The nucleus … which collide WITH other ...
... … place IN which a fission NUCLEAR reaction TAKES place. It contains …in THE form of rods to produce the appropriate result. The reactor consists OF a fuel, a moderator and A cooling system. An instrument …neutron WHICH strikes the nucleus of an atom of U-235. The nucleus … which collide WITH other ...
Periodic Table
... Atoms of one element are converted into another element May involve protons, neutrons, or electrons Have large energy changes Reaction rates are not affected ...
... Atoms of one element are converted into another element May involve protons, neutrons, or electrons Have large energy changes Reaction rates are not affected ...
6.3 Nuclear Reactions
... after binding may be fractionally small. • For systems with high binding energies, however, the missing mass may be an easily measurable fraction. ...
... after binding may be fractionally small. • For systems with high binding energies, however, the missing mass may be an easily measurable fraction. ...
Nuclear Reactions - Socastee High School
... uranium, U-238, does not split easily, but the 235 isotope does. We induce fission by “tickling” the nucleus with a “thermal” neutron. The neutron has to have the right kinetic energy: – Too little kinetic energy means that the neutron will bounce off the nucleus; – Too much kinetic energy means tha ...
... uranium, U-238, does not split easily, but the 235 isotope does. We induce fission by “tickling” the nucleus with a “thermal” neutron. The neutron has to have the right kinetic energy: – Too little kinetic energy means that the neutron will bounce off the nucleus; – Too much kinetic energy means tha ...
Matter Unit - OG
... 1.) Are made up of only one type of atom. 2) Cannot be broken down into any simpler substances by normal physical or chemical means. 3) Periodic Table of Elements *Familiarize yourself w/ it *Know what those numbers mean! ...
... 1.) Are made up of only one type of atom. 2) Cannot be broken down into any simpler substances by normal physical or chemical means. 3) Periodic Table of Elements *Familiarize yourself w/ it *Know what those numbers mean! ...
Nuclear Fission vs Fusion
... (Spl“i”tting an atom into two new ones) In nuclear fission reactions (also called radioactive decay), a neutron is aimed at the nucleus of a large, unstable atom, like uranium, thorium, or other radioactive elements. The extra mass of the neutron causes the radioactive nucleus to split apart, formin ...
... (Spl“i”tting an atom into two new ones) In nuclear fission reactions (also called radioactive decay), a neutron is aimed at the nucleus of a large, unstable atom, like uranium, thorium, or other radioactive elements. The extra mass of the neutron causes the radioactive nucleus to split apart, formin ...