strong force
... nucleus plus the masses of Z electrons The atomic masses of different isotopes are different The periodic table contains an average value of the atomic mass for each element based on the natural abundance of each isotope The value listed in the periodic table is the mass in grams of 1 mole [Avogadro ...
... nucleus plus the masses of Z electrons The atomic masses of different isotopes are different The periodic table contains an average value of the atomic mass for each element based on the natural abundance of each isotope The value listed in the periodic table is the mass in grams of 1 mole [Avogadro ...
The nucleus
... Binding energy B = (mass of nucleons) - (mass of nucleus) e.g. m n + m p – m D = B > 0 the potential energy of the nucleus is negative m n + m p + ( – E int ) = m D so B = E int the binding energy per nucleon B/A varies with A and is in the interval 5 MeV - 10 MeV (Krane) a A>62: B/A decreases with ...
... Binding energy B = (mass of nucleons) - (mass of nucleus) e.g. m n + m p – m D = B > 0 the potential energy of the nucleus is negative m n + m p + ( – E int ) = m D so B = E int the binding energy per nucleon B/A varies with A and is in the interval 5 MeV - 10 MeV (Krane) a A>62: B/A decreases with ...
Atomic and Nuclear Terms
... • The increases in binding energy per nucleon are much larger for fusion than for fission reactions, because the graph increases more steeply for light nuclei, so fusion gives out more energy per nucleon involved in the reaction than fission. • The stars are powered by fusion reactions. • Each secon ...
... • The increases in binding energy per nucleon are much larger for fusion than for fission reactions, because the graph increases more steeply for light nuclei, so fusion gives out more energy per nucleon involved in the reaction than fission. • The stars are powered by fusion reactions. • Each secon ...
entc 4390 medical imaging
... A common form of isomeric transition is gamma decay (g) in which the energy is released as a packet of energy (a quantum or photon) termed a gamma (g) ray An isomeric transition that competes with gamma decay is internal conversion, in which an electron from an extranuclear shell carries the energy ...
... A common form of isomeric transition is gamma decay (g) in which the energy is released as a packet of energy (a quantum or photon) termed a gamma (g) ray An isomeric transition that competes with gamma decay is internal conversion, in which an electron from an extranuclear shell carries the energy ...
Matter and Energy
... Most of the mass in an atom exists in the nucleus. Protons and neutrons are composed of the sub atomic particle known as quarks. A nucleus, must always have and equal number of protons and neutrons, with the exception of a hydrogen molecule. ...
... Most of the mass in an atom exists in the nucleus. Protons and neutrons are composed of the sub atomic particle known as quarks. A nucleus, must always have and equal number of protons and neutrons, with the exception of a hydrogen molecule. ...
I. Ch. 21.1 Nuclear Radiation
... The process continues until unstable isotopes of one element are changed, or transformed, into stable isotopes of a different element. ...
... The process continues until unstable isotopes of one element are changed, or transformed, into stable isotopes of a different element. ...
Atomic and Nuclear Terms
... • The increases in binding energy per nucleon are much larger for fusion than for fission reactions, because the graph increases more steeply for light nuclei, so fusion gives out more energy per nucleon involved in the reaction than fission. • The stars are powered by fusion reactions. • Each secon ...
... • The increases in binding energy per nucleon are much larger for fusion than for fission reactions, because the graph increases more steeply for light nuclei, so fusion gives out more energy per nucleon involved in the reaction than fission. • The stars are powered by fusion reactions. • Each secon ...
Introduction_to_Geochemistry_Pre-Lecture_Quiz
... Introduction to Geochemistry – Pre-lecture Quiz (1) True or False? (a) The diameter of an atom is less than the diameter of its nucleus. (b) The relative atomic mass of an atom is the mass of an atom relative to an atom of 12C. (c) p-orbitals can contain a maximum of 10 electrons. (d) The first ioni ...
... Introduction to Geochemistry – Pre-lecture Quiz (1) True or False? (a) The diameter of an atom is less than the diameter of its nucleus. (b) The relative atomic mass of an atom is the mass of an atom relative to an atom of 12C. (c) p-orbitals can contain a maximum of 10 electrons. (d) The first ioni ...
AP Chem
... even number of neutrons. The least stable situation is when both numbers are odd. There are only four (or five) stable odd/odd nuclei. Nuclides with a mass number over 200 usually undergo alpha decay. They emit a particle consisting of two protons and two neutrons. Nuclides with too many neutrons un ...
... even number of neutrons. The least stable situation is when both numbers are odd. There are only four (or five) stable odd/odd nuclei. Nuclides with a mass number over 200 usually undergo alpha decay. They emit a particle consisting of two protons and two neutrons. Nuclides with too many neutrons un ...
nuclear chemistry - Magoffin County Schools
... from the ATOMIC NUMBER. • Use the NEW ATOMIC NUMBER to locate the new element on the Periodic Table: ...
... from the ATOMIC NUMBER. • Use the NEW ATOMIC NUMBER to locate the new element on the Periodic Table: ...
File
... All matter is made of elements 92 naturally occurring elements on Earth Elements are made up of atoms. ...
... All matter is made of elements 92 naturally occurring elements on Earth Elements are made up of atoms. ...
Nuclear Reactions
... represents the number of protons in the nucleus • The number of protons determines what the element is. • Nucleus is held together by “strong force” ...
... represents the number of protons in the nucleus • The number of protons determines what the element is. • Nucleus is held together by “strong force” ...
FUSION AND FISSION
... • The fusion of two nuclei lighter than iron or nickel generally releases energy. • The fusion of nuclei heavier than them absorbs energy. Result: gain or loss of energy ...
... • The fusion of two nuclei lighter than iron or nickel generally releases energy. • The fusion of nuclei heavier than them absorbs energy. Result: gain or loss of energy ...
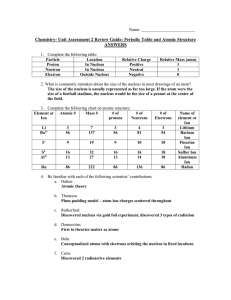
Chem Unit 2 Review Guide ANSWERS
... Atomic mass unit. Equal to the mass of a proton or neutron. 14.) What is a radioactive isotope? An unstable atom which decay (break down) and give off radioactive energy. 15.) What makes an atom unstable? An imbalance in the ratio of protons to neutrons. The farther this ratio gets from 1:1, the mor ...
... Atomic mass unit. Equal to the mass of a proton or neutron. 14.) What is a radioactive isotope? An unstable atom which decay (break down) and give off radioactive energy. 15.) What makes an atom unstable? An imbalance in the ratio of protons to neutrons. The farther this ratio gets from 1:1, the mor ...
13.4 The nucleus 3 - Nuclear fission and nuclear fusion
... Nuclear power stations, heat from the same chain reaction is used to make steam to generate electricity. The chain reaction is controlled by using uranium that has not been enriched so much, and by using control rods of materials that absorb neutrons. The atomic bomb exploding over Nagasaki In nucle ...
... Nuclear power stations, heat from the same chain reaction is used to make steam to generate electricity. The chain reaction is controlled by using uranium that has not been enriched so much, and by using control rods of materials that absorb neutrons. The atomic bomb exploding over Nagasaki In nucle ...