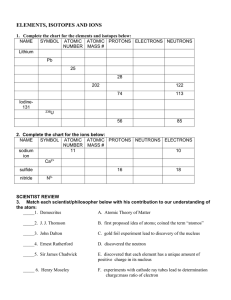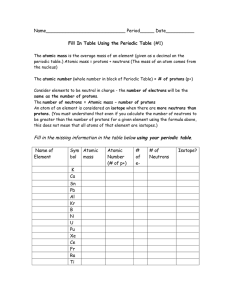
Periodic Table Fill in Table 1
... The atomic mass is the average mass of an element (given as a decimal on the periodic table.) Atomic mass = protons + neutrons (The mass of an atom comes from the nucleus) The atomic number (whole number in block of Periodic Table) = # of protons (p+) Consider elements to be neutral in charge - the ...
... The atomic mass is the average mass of an element (given as a decimal on the periodic table.) Atomic mass = protons + neutrons (The mass of an atom comes from the nucleus) The atomic number (whole number in block of Periodic Table) = # of protons (p+) Consider elements to be neutral in charge - the ...
Final Exam Review Part 1
... 2. What group number are the Noble Gases found in? __________ 3. Choose a color then color the group which represents the most reactive metals on the above periodic table: Color used: ________ 4. What is the group name of the most reactive metals you colored in question 3? ___________ 5. Choose a co ...
... 2. What group number are the Noble Gases found in? __________ 3. Choose a color then color the group which represents the most reactive metals on the above periodic table: Color used: ________ 4. What is the group name of the most reactive metals you colored in question 3? ___________ 5. Choose a co ...
Darlington High School EDI Lesson Plan Teacher: L. Grooms
... and charge and explain how these particles affect the properties of an atom. PS 2.3 Explain the trends of the periodic table based on the elements’ valence electrons and atomic number. PS 2.4 Use the atomic number and atom mass to determine the number of protons, neutrons and/or electrons for a give ...
... and charge and explain how these particles affect the properties of an atom. PS 2.3 Explain the trends of the periodic table based on the elements’ valence electrons and atomic number. PS 2.4 Use the atomic number and atom mass to determine the number of protons, neutrons and/or electrons for a give ...
Periodic Trends
... the periodic table • Radius of the atom – Distance between the nucleus and outermost electrons ...
... the periodic table • Radius of the atom – Distance between the nucleus and outermost electrons ...
notes - van Maarseveen
... o Negatively charged ___________________________ are found outside the nucleus. The electrons are much smaller than the protons and neutrons. Atomic number refers to the number of __________________ in the nucleus of an atom Each element has a different atomic number The total number of protons + ne ...
... o Negatively charged ___________________________ are found outside the nucleus. The electrons are much smaller than the protons and neutrons. Atomic number refers to the number of __________________ in the nucleus of an atom Each element has a different atomic number The total number of protons + ne ...
Chemistry Fall-2016 Final
... AL. one of the Group B (Groups 3-12) elements that is usually displayed in the main body of the periodic table ...
... AL. one of the Group B (Groups 3-12) elements that is usually displayed in the main body of the periodic table ...
Elements Unit Test
... 9. A Dutch scientist by the name of Neils Bohr was the first scientist to figure out that electrons that orbit the nucleus of an atom (sometimes called shells) can only hold a certain number of electrons. Which of the following sets of numbers describe the number of electrons in the first three shel ...
... 9. A Dutch scientist by the name of Neils Bohr was the first scientist to figure out that electrons that orbit the nucleus of an atom (sometimes called shells) can only hold a certain number of electrons. Which of the following sets of numbers describe the number of electrons in the first three shel ...
levels of organization and the atom
... unable to cut”. It has two areas: a nucleus and an electron cloud. It contains subatomic particles that are even smaller. ...
... unable to cut”. It has two areas: a nucleus and an electron cloud. It contains subatomic particles that are even smaller. ...
Atomic Structure
... An atom is the smallest building block of matter. Atoms are made of neutrons, protons and electrons. The nucleus of an atom is extremely small in comparison to the atom. If an atom was the size of the Houston Astrodome, then its nucleus would be the size of a pea. Scientists use the Periodic Table i ...
... An atom is the smallest building block of matter. Atoms are made of neutrons, protons and electrons. The nucleus of an atom is extremely small in comparison to the atom. If an atom was the size of the Houston Astrodome, then its nucleus would be the size of a pea. Scientists use the Periodic Table i ...
Chapter 11 and 12-2 Review/Study Guide for Test
... 13. Explain the difference between a group and a period on the periodic table of elements. Groups = columns (there are 18 total) and Periods = Rows (there are 7). Also, a group shares similar properties. 14. Why are neither the alkali metals nor the alkaline-earth metals found uncombined in nature? ...
... 13. Explain the difference between a group and a period on the periodic table of elements. Groups = columns (there are 18 total) and Periods = Rows (there are 7). Also, a group shares similar properties. 14. Why are neither the alkali metals nor the alkaline-earth metals found uncombined in nature? ...
2.9 Use the helium-4 isotope to define atomic number and mass
... 2.34 Give two examples of each of the following: (a) a diatomic molecule containing atoms of the same element, (b) a diatomic molecule containing atoms of different elements, (c) polyatomic molecule containing atoms of the same element, (d) a polyatomic molecule containing atoms of different element ...
... 2.34 Give two examples of each of the following: (a) a diatomic molecule containing atoms of the same element, (b) a diatomic molecule containing atoms of different elements, (c) polyatomic molecule containing atoms of the same element, (d) a polyatomic molecule containing atoms of different element ...
Intro to Chapter 5 Development of the Periodic Table
... Development of the Periodic Table Light and Electromagnetic Spectrum Electromagnetic Radiation and Atomic Spectra Particlelike Properties of Electromagnetic Radiation: The Planck Equation Wavelike Properties of Matter: The de Broglie Equation Quantum Mechanics and the Heisenberg Uncertainty Principl ...
... Development of the Periodic Table Light and Electromagnetic Spectrum Electromagnetic Radiation and Atomic Spectra Particlelike Properties of Electromagnetic Radiation: The Planck Equation Wavelike Properties of Matter: The de Broglie Equation Quantum Mechanics and the Heisenberg Uncertainty Principl ...
Study Guide Matter: Building Blocks of the Universe
... * Know the atomic particles: electron, neutron, and proton. where are they in the atom? What is their charge? What is their mass? How are electrons arranged in the electron cloud? * Know the four forces in the atom: strong, electromagnetic, weak, & gravity What are each of the forces responsible for ...
... * Know the atomic particles: electron, neutron, and proton. where are they in the atom? What is their charge? What is their mass? How are electrons arranged in the electron cloud? * Know the four forces in the atom: strong, electromagnetic, weak, & gravity What are each of the forces responsible for ...
Atomic Structure PPT Notes Sheet
... In a neutral atom, the number of ____________= the number of ____________. The atomic mass is equal to the number of __________ and the number of ___________. An __________________ represents the area in an atom where an electron is likely to be found. Each energy levels can hold only a ____________ ...
... In a neutral atom, the number of ____________= the number of ____________. The atomic mass is equal to the number of __________ and the number of ___________. An __________________ represents the area in an atom where an electron is likely to be found. Each energy levels can hold only a ____________ ...
Matter and Periodic Table Matter- anything that has mass and takes
... Nucleus-center of the atom; contains protons and neutrons A. Proton - positive charge - Identifies the atom - Atomic # = # of protons B. Neutron- no charge - Mass atom = protons + neutrons Electrons are located in the electron cloud. Electrons- negative charge - Responsible for all bonding - Found ...
... Nucleus-center of the atom; contains protons and neutrons A. Proton - positive charge - Identifies the atom - Atomic # = # of protons B. Neutron- no charge - Mass atom = protons + neutrons Electrons are located in the electron cloud. Electrons- negative charge - Responsible for all bonding - Found ...
Darlington High School EDI Lesson Plan Teacher: L. Grooms
... and charge and explain how these particles affect the properties of an atom. PS 2.3 Explain the trends of the periodic table based on the elements’ valence electrons and atomic number. PS 2.4 Use the atomic number and atom mass to determine the number of protons, neutrons and/or electrons for a give ...
... and charge and explain how these particles affect the properties of an atom. PS 2.3 Explain the trends of the periodic table based on the elements’ valence electrons and atomic number. PS 2.4 Use the atomic number and atom mass to determine the number of protons, neutrons and/or electrons for a give ...
10-2 Intensive Chemistry Review for Chapters 3
... 21. Several people were working to organize the known elements into some sort of structure at the same time (mid-1800s). Why is Mendeleev given the major credit for discovering the periodic table? (What did he do that was so special – even audacious?!) 22. What is the periodic law? What does it mean ...
... 21. Several people were working to organize the known elements into some sort of structure at the same time (mid-1800s). Why is Mendeleev given the major credit for discovering the periodic table? (What did he do that was so special – even audacious?!) 22. What is the periodic law? What does it mean ...
Atomic Theory- 1. Matter is composed of tiny, indivisible particles
... Atomic Theory1. Matter is composed of tiny, indivisible particles called atoms. 2. An element is composed of one type of atom. Properties of atoms are identical to each other. 3. A compound contains two or more different elements. The relative number of atoms of each element in a compound is the sam ...
... Atomic Theory1. Matter is composed of tiny, indivisible particles called atoms. 2. An element is composed of one type of atom. Properties of atoms are identical to each other. 3. A compound contains two or more different elements. The relative number of atoms of each element in a compound is the sam ...
C1a - Mr Corfe
... Element + water → Element hydroxide + hydrogen REACTIVITY SERIES Most reactive least reactive caesium Cs rubidium Rb potassium K sodium Na lithium Li calcium Ca magnesium Mg aluminium Al zinc Zn iron Fe Gold Au silver Ag RULE: An metal is more reactive if it is further to the l ...
... Element + water → Element hydroxide + hydrogen REACTIVITY SERIES Most reactive least reactive caesium Cs rubidium Rb potassium K sodium Na lithium Li calcium Ca magnesium Mg aluminium Al zinc Zn iron Fe Gold Au silver Ag RULE: An metal is more reactive if it is further to the l ...
Science - Atomic Structure
... • Review (from grade 5): Structure of atoms: protons, neutron, electrons Molecules Compounds are formed by combining two or more elements and have properties different from the constituent elements. ...
... • Review (from grade 5): Structure of atoms: protons, neutron, electrons Molecules Compounds are formed by combining two or more elements and have properties different from the constituent elements. ...
Chapter 4 Atomic Structure I. History of the Atom A. Democritus (400
... 1. Orbital: A region of space around the nucleus where an electron is likely to be found D. Electron configuration: The arrangement of electrons in the orbitals of an atom 1. Ground state: All the electrons in an atom have the lowest possible energy a. Stable 2. Excited state: An electron moves to a ...
... 1. Orbital: A region of space around the nucleus where an electron is likely to be found D. Electron configuration: The arrangement of electrons in the orbitals of an atom 1. Ground state: All the electrons in an atom have the lowest possible energy a. Stable 2. Excited state: An electron moves to a ...
Periodic table
The periodic table is a tabular arrangement of the chemical elements, ordered by their atomic number (number of protons in the nucleus), electron configurations, and recurring chemical properties. The table also shows four rectangular blocks: s-, p- d- and f-block. In general, within one row (period) the elements are metals on the lefthand side, and non-metals on the righthand side.The rows of the table are called periods; the columns are called groups. Six groups (columns) have names as well as numbers: for example, group 17 elements are the halogens; and group 18, the noble gases. The periodic table can be used to derive relationships between the properties of the elements, and predict the properties of new elements yet to be discovered or synthesized. The periodic table provides a useful framework for analyzing chemical behavior, and is widely used in chemistry and other sciences.Although precursors exist, Dmitri Mendeleev is generally credited with the publication, in 1869, of the first widely recognized periodic table. He developed his table to illustrate periodic trends in the properties of the then-known elements. Mendeleev also predicted some properties of then-unknown elements that would be expected to fill gaps in this table. Most of his predictions were proved correct when the elements in question were subsequently discovered. Mendeleev's periodic table has since been expanded and refined with the discovery or synthesis of further new elements and the development of new theoretical models to explain chemical behavior.All elements from atomic numbers 1 (hydrogen) to 118 (ununoctium) have been discovered or reportedly synthesized, with elements 113, 115, 117, and 118 having yet to be confirmed. The first 94 elements exist naturally, although some are found only in trace amounts and were synthesized in laboratories before being found in nature. Elements with atomic numbers from 95 to 118 have only been synthesized in laboratories. It has been shown that einsteinium and fermium once occurred in nature but currently do not. Synthesis of elements having higher atomic numbers is being pursued. Numerous synthetic radionuclides of naturally occurring elements have also been produced in laboratories.