Atoms and the Periodic Table Notes
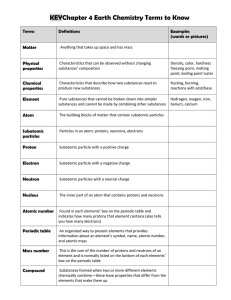
... Atoms of the same __________________ with ___________________ numbers Company,ofInc. ____________________________. ...
... Atoms of the same __________________ with ___________________ numbers Company,ofInc. ____________________________. ...
Atomic terms - ATOMIC NUMBER: The number of protons in the
... --- When atoms are arranged in order of their atomic weight, some of their chemical and physical properties repeat at regular intervals (periods) --- Some of the physical and chemical properties of atoms could be calculated based on atomic weight - Mendeleev was able to predict the properties of pre ...
... --- When atoms are arranged in order of their atomic weight, some of their chemical and physical properties repeat at regular intervals (periods) --- Some of the physical and chemical properties of atoms could be calculated based on atomic weight - Mendeleev was able to predict the properties of pre ...
ATOMS AND THE PERIODIC TABLE chapter three
... (METALOIDS). The elements that are between the metals and nonmetals are known as: SEMICONDUCTORS (METALOIDS) They may exhibit metallic and nonmetallic properties. B Si Ge ...
... (METALOIDS). The elements that are between the metals and nonmetals are known as: SEMICONDUCTORS (METALOIDS) They may exhibit metallic and nonmetallic properties. B Si Ge ...
File
... Substances formed when two or more different elements chemically combine—these have properties that differ from the elements that make them up ...
... Substances formed when two or more different elements chemically combine—these have properties that differ from the elements that make them up ...
document
... Periodic table – table where elements are organized by increasing atomic number (number of protons) In the late 1800’s, Dmitri Mendeleev devised first periodic table based on atomic mass – however, some elements were out of order. In 1913, Henry G. J. Moseley arranged elements by atomic number and i ...
... Periodic table – table where elements are organized by increasing atomic number (number of protons) In the late 1800’s, Dmitri Mendeleev devised first periodic table based on atomic mass – however, some elements were out of order. In 1913, Henry G. J. Moseley arranged elements by atomic number and i ...
Chemistry Test Study Guide
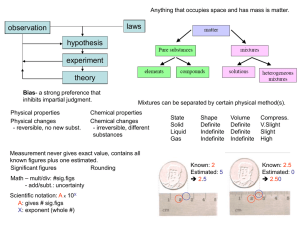
... 21. A mixture is created when two pure substances are combined so that each of the pure substances retains its own properties. 22. Where is the majority of the mass of an atom located? In the nucleus.(Protons and Neutrons) 23. If an atom loses electron’s, will it have a positive or negative charge? ...
... 21. A mixture is created when two pure substances are combined so that each of the pure substances retains its own properties. 22. Where is the majority of the mass of an atom located? In the nucleus.(Protons and Neutrons) 23. If an atom loses electron’s, will it have a positive or negative charge? ...
File
... What element is it and where is it on the PT? What is the atomic # of the element? This is equal to the # of protons. Is the atom neutral? Does it have a charge? If not, the # of electrons is equal to the # of protons. If I am given the mass number, I just have to subtract the # of protons from ...
... What element is it and where is it on the PT? What is the atomic # of the element? This is equal to the # of protons. Is the atom neutral? Does it have a charge? If not, the # of electrons is equal to the # of protons. If I am given the mass number, I just have to subtract the # of protons from ...
Intro to Element Note Answers
... Whose idea is closer to what we believe today? Democritus Whose idea was widely accepted for ~2000 years? Aristotle ...
... Whose idea is closer to what we believe today? Democritus Whose idea was widely accepted for ~2000 years? Aristotle ...
The topic that fascinated me the most in my Science lessons this
... power of predicting the existence and properties of elements yet to be discovered. Dimitri Mendeleev placed the 65 known elements of his time into a grid table and observed gaps in the table. Based on the position of gaps in the table, he predicted the existence of undiscovered elements and their pr ...
... power of predicting the existence and properties of elements yet to be discovered. Dimitri Mendeleev placed the 65 known elements of his time into a grid table and observed gaps in the table. Based on the position of gaps in the table, he predicted the existence of undiscovered elements and their pr ...
Atomic Structure: SOL Review #1 Name: Historical Developments 1
... The electrons are not “singly before pairing.” Electrons repel each other, so they do want not pair until there is no more “space” left in the sublevel. Periodic Table and Periodic Trends 27) Which elements would have similar properties to Na? ...
... The electrons are not “singly before pairing.” Electrons repel each other, so they do want not pair until there is no more “space” left in the sublevel. Periodic Table and Periodic Trends 27) Which elements would have similar properties to Na? ...
UNIT 5 REVIEW PROBLEMS
... 8. Determine which element would have characteristics of both a metal and a nonmetal: a. b. c. d. ...
... 8. Determine which element would have characteristics of both a metal and a nonmetal: a. b. c. d. ...
CLASS TEST NAME Class IIB Date ______ 1 .Which atomic
... 21. The electrons ______________________________________________________ around the nucleus in shells. The first shell, which is _______________________________ the nucleus, can hold ________electrons, whereas the 2nd and 3rd shells can hold ...
... 21. The electrons ______________________________________________________ around the nucleus in shells. The first shell, which is _______________________________ the nucleus, can hold ________electrons, whereas the 2nd and 3rd shells can hold ...
1. Define each of the following terms: a.Alkaline earth metals
... m.Chemical property = a property that describes the behavior of a substance n.Physical property = a property that describes the appearance of a substance o.Electron = a negatively charged subatomic particle found in the orbits of an atom ...
... m.Chemical property = a property that describes the behavior of a substance n.Physical property = a property that describes the appearance of a substance o.Electron = a negatively charged subatomic particle found in the orbits of an atom ...
Atomic terms Example: Helium has an atomic number of 2. Every
... Example: Helium has an atomic number of 2. Every helium atom has two protons in its nucleus. - MASS NUMBER: The number of protons PLUS the number of neutrons in the atomic nucleus, Atoms of the same element may have DIFFERENT mass numbers. - ISOTOPES: are atoms of the same element with different mas ...
... Example: Helium has an atomic number of 2. Every helium atom has two protons in its nucleus. - MASS NUMBER: The number of protons PLUS the number of neutrons in the atomic nucleus, Atoms of the same element may have DIFFERENT mass numbers. - ISOTOPES: are atoms of the same element with different mas ...
The periodic table is the most significant tool that chemist use for
... Atomic size - The atomic radius increases going down a group. This can be explain by adding more and more energy level. As one moves left to right within a period , the radii of the atoms decrease. As more and more protons are added to the nucleus, the nuclear charge of the elements increases. This ...
... Atomic size - The atomic radius increases going down a group. This can be explain by adding more and more energy level. As one moves left to right within a period , the radii of the atoms decrease. As more and more protons are added to the nucleus, the nuclear charge of the elements increases. This ...
Comprehensive Science 3 Module 4 Practice Test
... Different elements are made up of different atoms Atoms of different elements combine to make different compounds 5. The elements listed at the far right side of the periodic table are _______. Metalloids Nonmetals Metals Transitional Metals ...
... Different elements are made up of different atoms Atoms of different elements combine to make different compounds 5. The elements listed at the far right side of the periodic table are _______. Metalloids Nonmetals Metals Transitional Metals ...
Study Guide Answers
... 21. A mixture is created when two pure substances are combined so that each of the pure substances retains its own properties. 22. Where is the majority of the mass of an atom located? In the nucleus. 23. If an atom loses electron’s, will it have a positive or negative charge? Explain. Positive char ...
... 21. A mixture is created when two pure substances are combined so that each of the pure substances retains its own properties. 22. Where is the majority of the mass of an atom located? In the nucleus. 23. If an atom loses electron’s, will it have a positive or negative charge? Explain. Positive char ...
Note taker: ATOMS AND THE PERIODIC TABLE
... THOMPSON: •As it turns out, the atom can be divided into ________________________________________. •Thompson and Millikan are given credit for the first discoveries relating to _______________. ...
... THOMPSON: •As it turns out, the atom can be divided into ________________________________________. •Thompson and Millikan are given credit for the first discoveries relating to _______________. ...
Science 10 Chem notes
... outermost orbit that react with neighboring atoms to form compounds. Observations that supported theory: electricity passed through a gaseous element emits only certain wavelengths of light. ...
... outermost orbit that react with neighboring atoms to form compounds. Observations that supported theory: electricity passed through a gaseous element emits only certain wavelengths of light. ...
Physical Science Notes–Ch. 17-Glencoe
... From this information, he was able to ____________________________________________ of new elements that had not yet been discovered. ...
... From this information, he was able to ____________________________________________ of new elements that had not yet been discovered. ...
Periodic table
The periodic table is a tabular arrangement of the chemical elements, ordered by their atomic number (number of protons in the nucleus), electron configurations, and recurring chemical properties. The table also shows four rectangular blocks: s-, p- d- and f-block. In general, within one row (period) the elements are metals on the lefthand side, and non-metals on the righthand side.The rows of the table are called periods; the columns are called groups. Six groups (columns) have names as well as numbers: for example, group 17 elements are the halogens; and group 18, the noble gases. The periodic table can be used to derive relationships between the properties of the elements, and predict the properties of new elements yet to be discovered or synthesized. The periodic table provides a useful framework for analyzing chemical behavior, and is widely used in chemistry and other sciences.Although precursors exist, Dmitri Mendeleev is generally credited with the publication, in 1869, of the first widely recognized periodic table. He developed his table to illustrate periodic trends in the properties of the then-known elements. Mendeleev also predicted some properties of then-unknown elements that would be expected to fill gaps in this table. Most of his predictions were proved correct when the elements in question were subsequently discovered. Mendeleev's periodic table has since been expanded and refined with the discovery or synthesis of further new elements and the development of new theoretical models to explain chemical behavior.All elements from atomic numbers 1 (hydrogen) to 118 (ununoctium) have been discovered or reportedly synthesized, with elements 113, 115, 117, and 118 having yet to be confirmed. The first 94 elements exist naturally, although some are found only in trace amounts and were synthesized in laboratories before being found in nature. Elements with atomic numbers from 95 to 118 have only been synthesized in laboratories. It has been shown that einsteinium and fermium once occurred in nature but currently do not. Synthesis of elements having higher atomic numbers is being pursued. Numerous synthetic radionuclides of naturally occurring elements have also been produced in laboratories.