Atoms, Molecules and Ions
... Dalton’s Atomic Theory (1808) (Listed on p 203) 1. Elements are composed of extremely small particles called atoms. 2. All atoms of a given element are identical, having the same size, mass and chemical properties. 3. The atoms of one element are different from the atoms of all other elements. 4. C ...
... Dalton’s Atomic Theory (1808) (Listed on p 203) 1. Elements are composed of extremely small particles called atoms. 2. All atoms of a given element are identical, having the same size, mass and chemical properties. 3. The atoms of one element are different from the atoms of all other elements. 4. C ...
Chapter 1 D Study Guide
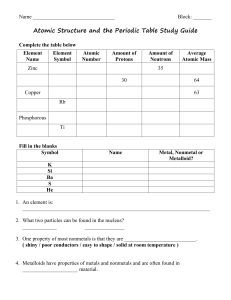
... 2. Protons and neutrons are found in the nucleus of an atom. 3. Electrons move around the nucleus in electron rings or shells or energy levels. 4. Atomic number is equal to the number of protons, and is unique to each element 5. The number of protons is equal to the number of electrons in a balanced ...
... 2. Protons and neutrons are found in the nucleus of an atom. 3. Electrons move around the nucleus in electron rings or shells or energy levels. 4. Atomic number is equal to the number of protons, and is unique to each element 5. The number of protons is equal to the number of electrons in a balanced ...
Periodic Trends
... The Periodic Table is arranged by atomic number and the resulting patterns of electron configurations, element properties, bond type, and what we call the periodic trends. The trends include atomic radii, ionic radii, ionization energy, and electronegativity. Atomic radii (AR) are the size of the at ...
... The Periodic Table is arranged by atomic number and the resulting patterns of electron configurations, element properties, bond type, and what we call the periodic trends. The trends include atomic radii, ionic radii, ionization energy, and electronegativity. Atomic radii (AR) are the size of the at ...
Chap 7: Around the Room Review
... 3. The atomic number tells us __________. 4. Nitrogen’s atomic number is 7. An isotope of nitrogen containing 7 neutrons would be nitrogen_____. 5. How does the size of a negative ion compare to the size of the atom that formed it? 6. The modern periodic table is organized by _____. 7. Elements in a ...
... 3. The atomic number tells us __________. 4. Nitrogen’s atomic number is 7. An isotope of nitrogen containing 7 neutrons would be nitrogen_____. 5. How does the size of a negative ion compare to the size of the atom that formed it? 6. The modern periodic table is organized by _____. 7. Elements in a ...
Section 3 The Periodic Table
... Elements that are in the same group have the same number of electrons in their outer energy level. ...
... Elements that are in the same group have the same number of electrons in their outer energy level. ...
Atoms - Science with Mrs. Schulte
... Also arranged in rows and columns, where elements share properties with the other elements in their groups ...
... Also arranged in rows and columns, where elements share properties with the other elements in their groups ...
Modern Atomic Theory and The Periodic Table
... _____________________________ arranged the first modern periodic table. –This periodic table now contains information regarding the chemical symbol, atomic number, average atomic mass, physical state of each element, group’s numbers, electron configurations, as well as many other useful characteris ...
... _____________________________ arranged the first modern periodic table. –This periodic table now contains information regarding the chemical symbol, atomic number, average atomic mass, physical state of each element, group’s numbers, electron configurations, as well as many other useful characteris ...
Atom The smallest piece of matter that still has the properties of the
... Sub-atomic particle with negative charge; much smaller than protons and neutrons Located at the center of the atom. Consists of protons and neutrons. Electrons surround the nucleus. Electron found in outermost shell of an atom; determines atoms chemical properties The number of protons in an atom. T ...
... Sub-atomic particle with negative charge; much smaller than protons and neutrons Located at the center of the atom. Consists of protons and neutrons. Electrons surround the nucleus. Electron found in outermost shell of an atom; determines atoms chemical properties The number of protons in an atom. T ...
Extra Credit Test Review
... 12.One atom has 17 protons, 18 neutrons, and 17 electrons. Another atom has 17 protons, 19 neutrons and 17 electrons. Are these the same element? Yes No Explain: __________________________________________________________________ 13.Today we use Mendeleev’s arrangement, elements are arranged by incre ...
... 12.One atom has 17 protons, 18 neutrons, and 17 electrons. Another atom has 17 protons, 19 neutrons and 17 electrons. Are these the same element? Yes No Explain: __________________________________________________________________ 13.Today we use Mendeleev’s arrangement, elements are arranged by incre ...
Atomic Structure and Nuclear Chemistry OEQs
... Do you believe that there are smaller subatomic particles than the ones that are currently believed to be the smallest? Explain. The Periodic Table of Elements is an organized table that contains all of the elements known-to-date. How the Periodic Table developed and what are its key features? ...
... Do you believe that there are smaller subatomic particles than the ones that are currently believed to be the smallest? Explain. The Periodic Table of Elements is an organized table that contains all of the elements known-to-date. How the Periodic Table developed and what are its key features? ...
Chemistry Presentation: Part One
... • A theory is a good, logical idea about something but it hasn’t been proven to be true ...
... • A theory is a good, logical idea about something but it hasn’t been proven to be true ...
Periodic Table
... go down the group. Atomic number gets larger as you go across the row from _________to _________ (period) and down the _______. Some of the elements are named after ________, _______, countries, and their Latin or Greek names. Hydrogen and Helium are separate from the other elements in their group. ...
... go down the group. Atomic number gets larger as you go across the row from _________to _________ (period) and down the _______. Some of the elements are named after ________, _______, countries, and their Latin or Greek names. Hydrogen and Helium are separate from the other elements in their group. ...
Chemistry Unit Study Guide Key
... formula for salt, sugar, and oxygen gas – Salt NaCl; Sugar C6H12O6; ...
... formula for salt, sugar, and oxygen gas – Salt NaCl; Sugar C6H12O6; ...
1. Of the three major categories of elements (metals, non
... They are called groups or families. 12. What are the horizontal rows on the periodic table called? They are called periods. 13. Explain the relationship between elements in the same group. They have similar chemical and physical properties because each one has the same number of valence electrons. ...
... They are called groups or families. 12. What are the horizontal rows on the periodic table called? They are called periods. 13. Explain the relationship between elements in the same group. They have similar chemical and physical properties because each one has the same number of valence electrons. ...
Page 233 - ClassZone
... he called atoms. Democritus said that all atoms were made of the same material. The objects of the world differed because each was made of atoms of different sizes and shapes. How does the modern view of atoms differ from this ancient view? How is it similar? ...
... he called atoms. Democritus said that all atoms were made of the same material. The objects of the world differed because each was made of atoms of different sizes and shapes. How does the modern view of atoms differ from this ancient view? How is it similar? ...
Exam 1 Review Questions
... Covalent compounds contain both metal and nonmetal atoms. Ionic compounds are made of molecules. Dmitri Mendeleev published the first modern atomic theory in 1805. Fluorine is found as a metal in its pure form. Francium chloride FrCl is a covalent compound. Graphite is a compound containing carbon a ...
... Covalent compounds contain both metal and nonmetal atoms. Ionic compounds are made of molecules. Dmitri Mendeleev published the first modern atomic theory in 1805. Fluorine is found as a metal in its pure form. Francium chloride FrCl is a covalent compound. Graphite is a compound containing carbon a ...
Fall Final Exam Review Questions
... 39. List an element that has properties similar to the element Carbon. 40. Draw the Lewis Dot structures for the following: Potassium, Carbon, Iodine and Xenon? 41. What are properties of metals and where are they generally located on a periodic table? 42. What are properties of nonmetals and where ...
... 39. List an element that has properties similar to the element Carbon. 40. Draw the Lewis Dot structures for the following: Potassium, Carbon, Iodine and Xenon? 41. What are properties of metals and where are they generally located on a periodic table? 42. What are properties of nonmetals and where ...
File
... • Atomic Number-the number of protons in the nucleus of an atom of a particular element. • Chemical Formula-an expression of the elements in a compound and their ratios in which the elements are denoted by their chemical ...
... • Atomic Number-the number of protons in the nucleus of an atom of a particular element. • Chemical Formula-an expression of the elements in a compound and their ratios in which the elements are denoted by their chemical ...
BC1 Atoms Unit Standards
... of the protons, neutrons, and electrons in an atom of an element. 2a For a given element, determine the number of protons 2b When given a number of protons, identify the element name and symbol 2c Identify the number of neutrons in an atom from atomic number and mass number 2d Identify the number of ...
... of the protons, neutrons, and electrons in an atom of an element. 2a For a given element, determine the number of protons 2b When given a number of protons, identify the element name and symbol 2c Identify the number of neutrons in an atom from atomic number and mass number 2d Identify the number of ...
File
... 7. Noble gases- any of the gaseous elements helium, neon, argon, krypton, xenon, and radon occupying Group 0 (18) of the periodic table. ...
... 7. Noble gases- any of the gaseous elements helium, neon, argon, krypton, xenon, and radon occupying Group 0 (18) of the periodic table. ...
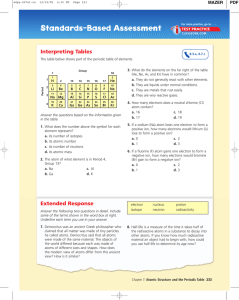
Periodic table
The periodic table is a tabular arrangement of the chemical elements, ordered by their atomic number (number of protons in the nucleus), electron configurations, and recurring chemical properties. The table also shows four rectangular blocks: s-, p- d- and f-block. In general, within one row (period) the elements are metals on the lefthand side, and non-metals on the righthand side.The rows of the table are called periods; the columns are called groups. Six groups (columns) have names as well as numbers: for example, group 17 elements are the halogens; and group 18, the noble gases. The periodic table can be used to derive relationships between the properties of the elements, and predict the properties of new elements yet to be discovered or synthesized. The periodic table provides a useful framework for analyzing chemical behavior, and is widely used in chemistry and other sciences.Although precursors exist, Dmitri Mendeleev is generally credited with the publication, in 1869, of the first widely recognized periodic table. He developed his table to illustrate periodic trends in the properties of the then-known elements. Mendeleev also predicted some properties of then-unknown elements that would be expected to fill gaps in this table. Most of his predictions were proved correct when the elements in question were subsequently discovered. Mendeleev's periodic table has since been expanded and refined with the discovery or synthesis of further new elements and the development of new theoretical models to explain chemical behavior.All elements from atomic numbers 1 (hydrogen) to 118 (ununoctium) have been discovered or reportedly synthesized, with elements 113, 115, 117, and 118 having yet to be confirmed. The first 94 elements exist naturally, although some are found only in trace amounts and were synthesized in laboratories before being found in nature. Elements with atomic numbers from 95 to 118 have only been synthesized in laboratories. It has been shown that einsteinium and fermium once occurred in nature but currently do not. Synthesis of elements having higher atomic numbers is being pursued. Numerous synthetic radionuclides of naturally occurring elements have also been produced in laboratories.