Atomic Size
... As you move down a group in the periodic table, atomic size generally increases. • WHY? ...
... As you move down a group in the periodic table, atomic size generally increases. • WHY? ...
CH.2
... differentiate between alpha, beta, and gamma radiation with respect to penetrating power, shielding, and composition. (B2) differentiate between the major atom components (proton, neutron and electron) in terms of location, size, and charge. (B2) distinguish between a group and a period. (B2) ...
... differentiate between alpha, beta, and gamma radiation with respect to penetrating power, shielding, and composition. (B2) differentiate between the major atom components (proton, neutron and electron) in terms of location, size, and charge. (B2) distinguish between a group and a period. (B2) ...
What is Matter? Anything that can be smelled, tasted, touched… Has
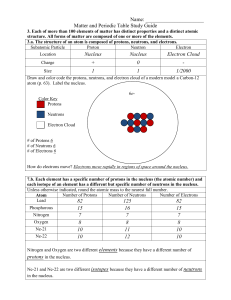
... Smallest part or piece of matter that still has the properties of matter Proton: Positively charged particle; largest of three particles; symbol is P+ Neutron: Neutrally charged particle; has both positive and negative charges; slightly smaller than proton; symbol is N= Electron: Negatively charge p ...
... Smallest part or piece of matter that still has the properties of matter Proton: Positively charged particle; largest of three particles; symbol is P+ Neutron: Neutrally charged particle; has both positive and negative charges; slightly smaller than proton; symbol is N= Electron: Negatively charge p ...
17 review for test - Blair Community Schools
... What happens to metallic properties as one goes across the table? ...
... What happens to metallic properties as one goes across the table? ...
Chemical Basis of Life
... both hydrogen and oxygen are gases at room temperature 2 - Some compounds are quite simple others are very complex, the important thing to remember is that a compound is always in a fixed ratio ...
... both hydrogen and oxygen are gases at room temperature 2 - Some compounds are quite simple others are very complex, the important thing to remember is that a compound is always in a fixed ratio ...
What is Matter? Anything that can be smelled, tasted, touched… Has
... Smallest part or piece of matter that still has the properties of matter : Positively charged particle; largest of three particles; symbol is : Neutrally charged particle; has both positive and negative charges; slightly smaller than proton; symbol is : Negatively charge particle; 2000 times smaller ...
... Smallest part or piece of matter that still has the properties of matter : Positively charged particle; largest of three particles; symbol is : Neutrally charged particle; has both positive and negative charges; slightly smaller than proton; symbol is : Negatively charge particle; 2000 times smaller ...
What is Matter
... both positive and negative charges; slightly smaller than proton; symbol is N= Electron: Negatively charge particle; 2000 times smaller than proton; symbol is e- ...
... both positive and negative charges; slightly smaller than proton; symbol is N= Electron: Negatively charge particle; 2000 times smaller than proton; symbol is e- ...
HONORS CHEMISTRY Quarter 2 Exam Topics Know the following
... Know experimental observations and their key contributions to the atomic theory. Be able to draw and label the model of the atom for each scientist mentioned above. Know Dalton’s postulates. Distinguish atoms based upon their number of subatomic particles. Distinguish between isotopes. Underst ...
... Know experimental observations and their key contributions to the atomic theory. Be able to draw and label the model of the atom for each scientist mentioned above. Know Dalton’s postulates. Distinguish atoms based upon their number of subatomic particles. Distinguish between isotopes. Underst ...
CHAPTER6_MEET_THE_ELEMENTS
... Atomic number – is the number of protons in the nucleus of an atom. This number is ABOVE the element’s symbol. An element’s position in the periodic table is determined by it’s atomic number. The atomic number (number of protons) makes each element unique from the others. Vertical columns are calle ...
... Atomic number – is the number of protons in the nucleus of an atom. This number is ABOVE the element’s symbol. An element’s position in the periodic table is determined by it’s atomic number. The atomic number (number of protons) makes each element unique from the others. Vertical columns are calle ...
Chemistry Test Review – 8th Science Vocabulary: Element atom
... electron subatomic particles atomic number atomic mass unit atomic mass mass number valence electron valence shell octet rule period shell/energy level group/family metals nonmetals metalloids Bohr model of the atom Lewis Dot Diagrams of an atom Representative elements Transition elements Dalton’s A ...
... electron subatomic particles atomic number atomic mass unit atomic mass mass number valence electron valence shell octet rule period shell/energy level group/family metals nonmetals metalloids Bohr model of the atom Lewis Dot Diagrams of an atom Representative elements Transition elements Dalton’s A ...
atomic theory quiz II review
... We will have a quiz over the information you have learned about the atomic theory. Please actively study (sing, dance, act, draw, write) the following info: ...
... We will have a quiz over the information you have learned about the atomic theory. Please actively study (sing, dance, act, draw, write) the following info: ...
VOCABULARY name, date, hour: Fill in the number of each term
... ___ substance that is a mixture of two or more metals ___ columns of the periodic table; also known as groups ___ number of protons carried by the nucleus of an atom ___ element with an imbalance in the number of neutrons and protons ___ uncharged particle found in the nucleus of an atom ___ physica ...
... ___ substance that is a mixture of two or more metals ___ columns of the periodic table; also known as groups ___ number of protons carried by the nucleus of an atom ___ element with an imbalance in the number of neutrons and protons ___ uncharged particle found in the nucleus of an atom ___ physica ...
Chemistry Notes
... Gain valence electrons when they bond Properties of metalloids Elements that touch stair-step line between metals and nonmetals Have characteristics of metals and nonmetals Family/Group – columns of elements Same physical and chemical properties Same number of valence electrons Period – ro ...
... Gain valence electrons when they bond Properties of metalloids Elements that touch stair-step line between metals and nonmetals Have characteristics of metals and nonmetals Family/Group – columns of elements Same physical and chemical properties Same number of valence electrons Period – ro ...
Properties of matter student notes[1]
... Electrons = Invisible, ______________________charged particles. Nucleus = Positively charged _________________________of an atom Protons = _______________________ charged particles in the nucleus Neutrons = _____________________ particles in the nucleus ...
... Electrons = Invisible, ______________________charged particles. Nucleus = Positively charged _________________________of an atom Protons = _______________________ charged particles in the nucleus Neutrons = _____________________ particles in the nucleus ...
Chapter 7 Review Sheet
... 5. It would require more energy to remove an electron from Az than from any other element. 6. Jq has one more valence electron than Dw but one less valence electron than Gt. 7. The sizes of the 3 isoelectronic species are: Cx+ < Hs < By–. 8. Ev and Kp both lose the same number of electrons when they ...
... 5. It would require more energy to remove an electron from Az than from any other element. 6. Jq has one more valence electron than Dw but one less valence electron than Gt. 7. The sizes of the 3 isoelectronic species are: Cx+ < Hs < By–. 8. Ev and Kp both lose the same number of electrons when they ...
Semester 1 Exam Review Part 1
... 10. Vertical column in the Periodic Table. 11. Elements in the same family have similar ___. 12. Horizontal row on the Periodic Table. 13. Elements on the right side of the Periodic Table. ...
... 10. Vertical column in the Periodic Table. 11. Elements in the same family have similar ___. 12. Horizontal row on the Periodic Table. 13. Elements on the right side of the Periodic Table. ...
Understanding the Atom GN
... When atoms of the same element have different numbers of neutrons they are called ____________________. Isotope – ________________________________________________________________________ Most elements have ______________________ isotopes. Mass Number - ________________________________________ ...
... When atoms of the same element have different numbers of neutrons they are called ____________________. Isotope – ________________________________________________________________________ Most elements have ______________________ isotopes. Mass Number - ________________________________________ ...
Thursday, October 31, 2013 D-day
... • Radioactive Elements- no naturally occurring stable isotope (what is an isotope?). – These elements loose neutrons and protons and emit them as particles. – All manmade elements are radioactive. ...
... • Radioactive Elements- no naturally occurring stable isotope (what is an isotope?). – These elements loose neutrons and protons and emit them as particles. – All manmade elements are radioactive. ...
Matter and the Periodic Table Study Guide Answer Key
... 3.b. Compounds are formed by combining two or more different elements and compounds have properties that are different from their constituent elements. 3.f. Use the periodic table to identify elements in simple compounds. ...
... 3.b. Compounds are formed by combining two or more different elements and compounds have properties that are different from their constituent elements. 3.f. Use the periodic table to identify elements in simple compounds. ...
History of the Atom and Periodic Table
... had a neutral charge and it is called the neutron. His discovery made us realize isotopes existed. Isotopes are atoms of the same element with a different number of neutrons. Proved Dalton’s Atomic theory was incorrect again by showing atoms of the same element can be different. ...
... had a neutral charge and it is called the neutron. His discovery made us realize isotopes existed. Isotopes are atoms of the same element with a different number of neutrons. Proved Dalton’s Atomic theory was incorrect again by showing atoms of the same element can be different. ...
Periodic table
The periodic table is a tabular arrangement of the chemical elements, ordered by their atomic number (number of protons in the nucleus), electron configurations, and recurring chemical properties. The table also shows four rectangular blocks: s-, p- d- and f-block. In general, within one row (period) the elements are metals on the lefthand side, and non-metals on the righthand side.The rows of the table are called periods; the columns are called groups. Six groups (columns) have names as well as numbers: for example, group 17 elements are the halogens; and group 18, the noble gases. The periodic table can be used to derive relationships between the properties of the elements, and predict the properties of new elements yet to be discovered or synthesized. The periodic table provides a useful framework for analyzing chemical behavior, and is widely used in chemistry and other sciences.Although precursors exist, Dmitri Mendeleev is generally credited with the publication, in 1869, of the first widely recognized periodic table. He developed his table to illustrate periodic trends in the properties of the then-known elements. Mendeleev also predicted some properties of then-unknown elements that would be expected to fill gaps in this table. Most of his predictions were proved correct when the elements in question were subsequently discovered. Mendeleev's periodic table has since been expanded and refined with the discovery or synthesis of further new elements and the development of new theoretical models to explain chemical behavior.All elements from atomic numbers 1 (hydrogen) to 118 (ununoctium) have been discovered or reportedly synthesized, with elements 113, 115, 117, and 118 having yet to be confirmed. The first 94 elements exist naturally, although some are found only in trace amounts and were synthesized in laboratories before being found in nature. Elements with atomic numbers from 95 to 118 have only been synthesized in laboratories. It has been shown that einsteinium and fermium once occurred in nature but currently do not. Synthesis of elements having higher atomic numbers is being pursued. Numerous synthetic radionuclides of naturally occurring elements have also been produced in laboratories.
















![Properties of matter student notes[1]](http://s1.studyres.com/store/data/009076956_1-3293fc3fecf578fd34e3f0f2700d471f-300x300.png)