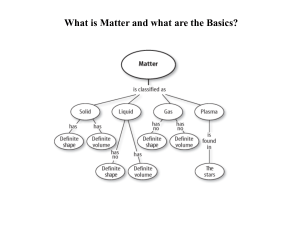
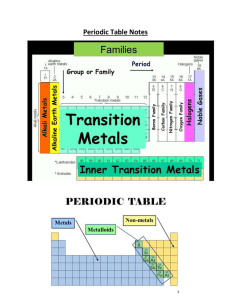
Chapter 5 - Effingham County Schools
... Atoms are composed of three types of particles. Electrons, protons and neutrons. A proton is positively charged. A neutron is an uncharged particle. Electrons are negatively charged. The protons and neutrons of an atom are grouped together in the atom’s center which is called the nucleus. Electrons ...
... Atoms are composed of three types of particles. Electrons, protons and neutrons. A proton is positively charged. A neutron is an uncharged particle. Electrons are negatively charged. The protons and neutrons of an atom are grouped together in the atom’s center which is called the nucleus. Electrons ...
Unit 2: Atomic Structure and Nuclear Chemistry
... Unit 2B: Electron Configuration and the Periodic Table Unit Introduction In this unit students will describe how the arrangement of elements in the periodic table and their electron configurations are related. They will describe how the location of an element in the periodic table can be used to pre ...
... Unit 2B: Electron Configuration and the Periodic Table Unit Introduction In this unit students will describe how the arrangement of elements in the periodic table and their electron configurations are related. They will describe how the location of an element in the periodic table can be used to pre ...
Atomic Structure and the Periodic Table
... nucleus of an atom. Neutron – neutrally charged particle found in the nucleus of an atom. Atomic number – the number of protons in the nucleus of an atom. ...
... nucleus of an atom. Neutron – neutrally charged particle found in the nucleus of an atom. Atomic number – the number of protons in the nucleus of an atom. ...
Periodic Table
... 3. Metalloids (Semimetals) = have properties of both metals and non-metals III. Trends or Patterns in the Periodic Table A. Certain properties of elements in the periodic table follow a predictable ...
... 3. Metalloids (Semimetals) = have properties of both metals and non-metals III. Trends or Patterns in the Periodic Table A. Certain properties of elements in the periodic table follow a predictable ...
Biochemistry I (CHE 418 / 5418)
... • Protons and neutrons are located in the nucleus. • Electrons orbit the nucleus in specific orbits with each orbit corresponding to a different energy level. – Ground state (most stable state) when electrons are in energy levels as near as possible to the nucleus – Excited state when electrons is p ...
... • Protons and neutrons are located in the nucleus. • Electrons orbit the nucleus in specific orbits with each orbit corresponding to a different energy level. – Ground state (most stable state) when electrons are in energy levels as near as possible to the nucleus – Excited state when electrons is p ...
atomic number - Thomas C. Cario Middle School
... The periodic table is a chart containing information about the atoms that make up all matter. An element is a substance made up of only one type of atom. The atomic number of an atom is equal to the number of protons in its nucleus. The number of electrons surrounding the nucleus of an atom is equal ...
... The periodic table is a chart containing information about the atoms that make up all matter. An element is a substance made up of only one type of atom. The atomic number of an atom is equal to the number of protons in its nucleus. The number of electrons surrounding the nucleus of an atom is equal ...
Chemistry--Chapter 5: Atomic Structure and the Periodic Table
... and arranged the periodic table in order according to atomic number B. The Modern Periodic Table 1. The periodic law states that when the elements are arranged in order of increasing atomic number, there is a periodic repetition of their physical and chemical properties. 2. Elements in the same colu ...
... and arranged the periodic table in order according to atomic number B. The Modern Periodic Table 1. The periodic law states that when the elements are arranged in order of increasing atomic number, there is a periodic repetition of their physical and chemical properties. 2. Elements in the same colu ...
Classifying Atoms
... The periodic table, a tool used to organize information about the elements, appears on pages 698–699 of the Appendix. Of the more than 100 known elements listed there, 92 occur naturally on Earth in significant amounts. The rest are synthetic elements produced by scientists. In each row of the perio ...
... The periodic table, a tool used to organize information about the elements, appears on pages 698–699 of the Appendix. Of the more than 100 known elements listed there, 92 occur naturally on Earth in significant amounts. The rest are synthetic elements produced by scientists. In each row of the perio ...
Atomic Theory
... Isotopes • An isotopes are atoms with the same number of protons but different numbers of neutrons. • Most elements have several naturally occuring isotopes ...
... Isotopes • An isotopes are atoms with the same number of protons but different numbers of neutrons. • Most elements have several naturally occuring isotopes ...
Introduction_to_Geochemistry_Pre-Lecture_Quiz
... (b) The relative atomic mass of an atom is the mass of an atom relative to an atom of 12C. (c) p-orbitals can contain a maximum of 10 electrons. (d) The first ionisation energy of an element is the energy input (in kg mol-1) required to detach the loosest electron from atoms of that element. (e) All ...
... (b) The relative atomic mass of an atom is the mass of an atom relative to an atom of 12C. (c) p-orbitals can contain a maximum of 10 electrons. (d) The first ionisation energy of an element is the energy input (in kg mol-1) required to detach the loosest electron from atoms of that element. (e) All ...
Chemistry Timeline
... Joseph John Thomson Polonium) Four elements Invented a good atomic theory (list all points) Matter is made up of “atomos” For each person include in a TYPED paragraph: 1. The name of the scientist with birth and death dates (as known). In other words, WHEN. 2. A complete explanation of their ...
... Joseph John Thomson Polonium) Four elements Invented a good atomic theory (list all points) Matter is made up of “atomos” For each person include in a TYPED paragraph: 1. The name of the scientist with birth and death dates (as known). In other words, WHEN. 2. A complete explanation of their ...
Blue File
... Over the years Many Scientists have put forward ‘models’ that they have arrived at from their research of what they understand an atom to look like……. Firstly came : ‘J.J Thompson’s ‘Plum Pudding ‘ model which showed that ‘atoms were tiny balls of positive charge with tiny negative particles stuck i ...
... Over the years Many Scientists have put forward ‘models’ that they have arrived at from their research of what they understand an atom to look like……. Firstly came : ‘J.J Thompson’s ‘Plum Pudding ‘ model which showed that ‘atoms were tiny balls of positive charge with tiny negative particles stuck i ...
CHAPTER 1: INTRODUCTION TO CHEMISTRY
... All groups except for the ones in the transition metals and metalloids all have similar characteristics ...
... All groups except for the ones in the transition metals and metalloids all have similar characteristics ...
element - Mrs. Phillips` Physical Science Webpage
... • Classification systems are methods of grouping things together in a way that makes sense, so that like are with like – makes it easier to find things if you know where to look. • The periodic table was developed by Dmitri Mendeleev in 1869, as a way of classifying elements according to their prope ...
... • Classification systems are methods of grouping things together in a way that makes sense, so that like are with like – makes it easier to find things if you know where to look. • The periodic table was developed by Dmitri Mendeleev in 1869, as a way of classifying elements according to their prope ...
Matter and the Periodic Table
... The Periodic Table In 1869 Dmitri Mendeleev (18341907) succeeded in organizing the 62 elements known at that time into a system of rows and columns on the basis of increasing mass and similar chemical and physical properties. Since the organization exhibited a periodic repetition of similar propert ...
... The Periodic Table In 1869 Dmitri Mendeleev (18341907) succeeded in organizing the 62 elements known at that time into a system of rows and columns on the basis of increasing mass and similar chemical and physical properties. Since the organization exhibited a periodic repetition of similar propert ...
Periodic Table Quiz
... 3. An unknown sample is malleable and ductile. Where on the periodic table would the sample most likely be found? a. To the right of the zig-zag line. b. Bordering right on the zig-zag line. c. To the left of the zig-zag line. d. There are no elements that are both malleable and ductile found on the ...
... 3. An unknown sample is malleable and ductile. Where on the periodic table would the sample most likely be found? a. To the right of the zig-zag line. b. Bordering right on the zig-zag line. c. To the left of the zig-zag line. d. There are no elements that are both malleable and ductile found on the ...
Ch2 lecture outline - OnCourse Systems For Education
... EX: Naturally occurring chromium consists of four isotopes. IT is 4.31 % Cr-50 of mass 49.946 amu; 83.76 % Cr-52 of mass 51.941 amu; 9.55 % Cr-53 of mass 52.941 amu; and 2.38 % Cr-54 of mass 53.939 amu. Calculate the average atomic mass of chromium. ...
... EX: Naturally occurring chromium consists of four isotopes. IT is 4.31 % Cr-50 of mass 49.946 amu; 83.76 % Cr-52 of mass 51.941 amu; 9.55 % Cr-53 of mass 52.941 amu; and 2.38 % Cr-54 of mass 53.939 amu. Calculate the average atomic mass of chromium. ...
Chapter 9 - Fayetteville State University
... 7) Periodic Law: States that elements arranged in order of the atomic number share similar chemical and physical properties. These arrangement are called groups, examples are the alkali metals (Li, Na, K, Rb, Cs), the halogens (F, Cl, Br, I). 8) Groups: A sequence of elements of increasing atomic nu ...
... 7) Periodic Law: States that elements arranged in order of the atomic number share similar chemical and physical properties. These arrangement are called groups, examples are the alkali metals (Li, Na, K, Rb, Cs), the halogens (F, Cl, Br, I). 8) Groups: A sequence of elements of increasing atomic nu ...
Atoms - misshoughton.net
... cannot be broken down into simpler parts by a chemical change. Compounds: pure substances made of more than one type of atom. Compounds are made of elements. NaCl (sodium chloride) is an example of a compound. ...
... cannot be broken down into simpler parts by a chemical change. Compounds: pure substances made of more than one type of atom. Compounds are made of elements. NaCl (sodium chloride) is an example of a compound. ...
Period Table, valence Electrons and Ion Notes
... Example: Na = 1s2 2s2 2p6 3s1 Add up the e-‘s found in the last energy level, in this case there is only 1 so Na has 1 valence e**You have to do this for the Transition metal every time** ...
... Example: Na = 1s2 2s2 2p6 3s1 Add up the e-‘s found in the last energy level, in this case there is only 1 so Na has 1 valence e**You have to do this for the Transition metal every time** ...
Periodic table
The periodic table is a tabular arrangement of the chemical elements, ordered by their atomic number (number of protons in the nucleus), electron configurations, and recurring chemical properties. The table also shows four rectangular blocks: s-, p- d- and f-block. In general, within one row (period) the elements are metals on the lefthand side, and non-metals on the righthand side.The rows of the table are called periods; the columns are called groups. Six groups (columns) have names as well as numbers: for example, group 17 elements are the halogens; and group 18, the noble gases. The periodic table can be used to derive relationships between the properties of the elements, and predict the properties of new elements yet to be discovered or synthesized. The periodic table provides a useful framework for analyzing chemical behavior, and is widely used in chemistry and other sciences.Although precursors exist, Dmitri Mendeleev is generally credited with the publication, in 1869, of the first widely recognized periodic table. He developed his table to illustrate periodic trends in the properties of the then-known elements. Mendeleev also predicted some properties of then-unknown elements that would be expected to fill gaps in this table. Most of his predictions were proved correct when the elements in question were subsequently discovered. Mendeleev's periodic table has since been expanded and refined with the discovery or synthesis of further new elements and the development of new theoretical models to explain chemical behavior.All elements from atomic numbers 1 (hydrogen) to 118 (ununoctium) have been discovered or reportedly synthesized, with elements 113, 115, 117, and 118 having yet to be confirmed. The first 94 elements exist naturally, although some are found only in trace amounts and were synthesized in laboratories before being found in nature. Elements with atomic numbers from 95 to 118 have only been synthesized in laboratories. It has been shown that einsteinium and fermium once occurred in nature but currently do not. Synthesis of elements having higher atomic numbers is being pursued. Numerous synthetic radionuclides of naturally occurring elements have also been produced in laboratories.