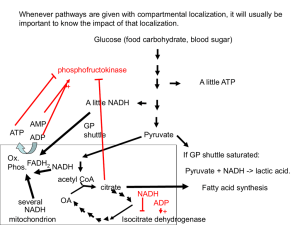
C483 Final Exam Study Guide The final will be held in CH 001 at 8
... molecule that you store in your liver. Circle the pathways/cycles below that are part of this overall transformation. Cross out any that are not. Gluconeogenesis, pentose phosphate pathway, glycogen synthesis, glycolysis, citric acid cycle B. Trace the metabolic path of this glutamate molecule throu ...
... molecule that you store in your liver. Circle the pathways/cycles below that are part of this overall transformation. Cross out any that are not. Gluconeogenesis, pentose phosphate pathway, glycogen synthesis, glycolysis, citric acid cycle B. Trace the metabolic path of this glutamate molecule throu ...
Biomacromolecules ppt
... • Nucleic acids are composed of long chains of nucleotides • The function of nucleic acids is to contain and transfer genetic ...
... • Nucleic acids are composed of long chains of nucleotides • The function of nucleic acids is to contain and transfer genetic ...
macromolecules notes
... -is made of glucose + glucose -formula = C12H22O11 H2O 3. Polysaccharides (Complex carbohydrates) a. Glycogen -used by animals to store extra sugar -sometimes called ‘animal starch’ -made entirely of α-glucose with α(1→4) glycosidic bonds -is highly branched molecule in the form of granules Glycogen ...
... -is made of glucose + glucose -formula = C12H22O11 H2O 3. Polysaccharides (Complex carbohydrates) a. Glycogen -used by animals to store extra sugar -sometimes called ‘animal starch’ -made entirely of α-glucose with α(1→4) glycosidic bonds -is highly branched molecule in the form of granules Glycogen ...
The ingredients of life. - Waterford Public Schools
... Each macromolecule performs it’s own tasks. ...
... Each macromolecule performs it’s own tasks. ...
Lect 1 (Metabolic Pathways) Lect 2 (Enzymes) Lect 3 (Glucose
... Glucokinase: hexokinase converts glucose > G6P. Has a low affinity for glucose & a maximal Km at 10Km. only found in liver cells. G6P is used to keep [blood glucose] low. Converted to glucose by Glucose-6Phosphorylase. G6P can’t enter/leave the cell due to Pi group. When [blood glucose] high, G6P ca ...
... Glucokinase: hexokinase converts glucose > G6P. Has a low affinity for glucose & a maximal Km at 10Km. only found in liver cells. G6P is used to keep [blood glucose] low. Converted to glucose by Glucose-6Phosphorylase. G6P can’t enter/leave the cell due to Pi group. When [blood glucose] high, G6P ca ...
Macromolecules Notes
... building blocks of polymers. Ex: Glucose is a monomer, starch is a polymer: many glucose bonded together make starch. ...
... building blocks of polymers. Ex: Glucose is a monomer, starch is a polymer: many glucose bonded together make starch. ...
Summary of lesson
... In this part of the lesson, you will learn about the metabolic pathways that cells use to turn sugar into usable, cellular energy and watch simulations of those pathways. 1. Read the background information on pages 1.2 and 1.3 to start learning about glycolysis. Move to pages 1.5 – 1.7. Answer quest ...
... In this part of the lesson, you will learn about the metabolic pathways that cells use to turn sugar into usable, cellular energy and watch simulations of those pathways. 1. Read the background information on pages 1.2 and 1.3 to start learning about glycolysis. Move to pages 1.5 – 1.7. Answer quest ...
Biochemistry Note
... Macromolecules – Large organic molecules that contain dozens of carbon atoms and many functional groups. There are four major groups of biologically important macromolecules: ...
... Macromolecules – Large organic molecules that contain dozens of carbon atoms and many functional groups. There are four major groups of biologically important macromolecules: ...
Chem*3560 Lecture 35: Integration of metabolism in animals
... Amino acids in blood may be derived from dietary protein. If this source is insufficient, breakdown of tissue proteins occurs, mostly from body muscle mass (which is why starvation dieting is ill-advised). The liver has very active protein synthesis and degradation, and also makes plasma proteins to ...
... Amino acids in blood may be derived from dietary protein. If this source is insufficient, breakdown of tissue proteins occurs, mostly from body muscle mass (which is why starvation dieting is ill-advised). The liver has very active protein synthesis and degradation, and also makes plasma proteins to ...
Chapter 7 – How Cells Release Stored Energy
... Depends upon membranes and ATP synthase ETC makes this E transport possible Cells generate most of their E this way ...
... Depends upon membranes and ATP synthase ETC makes this E transport possible Cells generate most of their E this way ...
Liver glycogen constitutes a reserve of glucose for the
... The reserve is not large. In fact, the reserve is virtually exhausted in 24 hours. In such an animal the continuing requirement for glucose is satisfied by gluconeogenesis, which is the synthesis of glucose from non-carbohydrate precursors: lactate, propionate, glycerol, pyruvate, gluconeogenic amin ...
... The reserve is not large. In fact, the reserve is virtually exhausted in 24 hours. In such an animal the continuing requirement for glucose is satisfied by gluconeogenesis, which is the synthesis of glucose from non-carbohydrate precursors: lactate, propionate, glycerol, pyruvate, gluconeogenic amin ...
Exam 3 - Chemistry Courses: About
... Draw the structure of -ketoglutarate below. Use the structure of the compound to explain why isocitrate dehydrogenase and -ketoglutarate dehydrogenase have such different mechanisms of action even though they both catalyze an oxidative decarboxylation reaction. ...
... Draw the structure of -ketoglutarate below. Use the structure of the compound to explain why isocitrate dehydrogenase and -ketoglutarate dehydrogenase have such different mechanisms of action even though they both catalyze an oxidative decarboxylation reaction. ...
Biology 1408 - Lone Star College
... Enzymes are not permanently changed by the reactions they promote. All enzymes are proteins. Enzymes usually speed up chemical reactions. Enzyme activity is regulated. Enzymes are highly specific for the jobs they carry out. ...
... Enzymes are not permanently changed by the reactions they promote. All enzymes are proteins. Enzymes usually speed up chemical reactions. Enzyme activity is regulated. Enzymes are highly specific for the jobs they carry out. ...
Here
... Glycolysis: first step in aerobic or anaerobic respiration. Anaerobic: without oxygen Aerobic: with oxygen Oxidation: Reaction when an atom or molecule loses electrons Reduction: Reaction when an atom or molecule gains electrons, Fermentation: Glycolysis is followed by the conversion of pyruvic acid ...
... Glycolysis: first step in aerobic or anaerobic respiration. Anaerobic: without oxygen Aerobic: with oxygen Oxidation: Reaction when an atom or molecule loses electrons Reduction: Reaction when an atom or molecule gains electrons, Fermentation: Glycolysis is followed by the conversion of pyruvic acid ...
Carbohydrate Metabolism
... E. Because food remains for a short time in the mouth, digestion of starch and glycogen may be incomplete and gives a partial digestion products called: starch dextrins (amylodextrin, erythrodextrin and achrodextrin). F. Therefore, digestion of starch and glycogen in the mouth gives 3 maltose, isoma ...
... E. Because food remains for a short time in the mouth, digestion of starch and glycogen may be incomplete and gives a partial digestion products called: starch dextrins (amylodextrin, erythrodextrin and achrodextrin). F. Therefore, digestion of starch and glycogen in the mouth gives 3 maltose, isoma ...
Exercise 5
... construct a molecule of oxaloacetate (see page 553). Do not label these carbons. Perform the sequence of reactions in the citric acid cycle (reactions 1 through 8 on page 553). Do not worry about the electron carriers NAD+ and FAD; what you are doing is tracing the fate of the carbon atoms in the or ...
... construct a molecule of oxaloacetate (see page 553). Do not label these carbons. Perform the sequence of reactions in the citric acid cycle (reactions 1 through 8 on page 553). Do not worry about the electron carriers NAD+ and FAD; what you are doing is tracing the fate of the carbon atoms in the or ...
1. The table shows the number of carbon atoms contained in some
... The table shows the number of carbon atoms contained in some substances. Substance ...
... The table shows the number of carbon atoms contained in some substances. Substance ...
Exercise 5
... construct a molecule of oxaloacetate (see page 553). Do not label these carbons. Perform the sequence of reactions in the citric acid cycle (reactions 1 through 8 on page 553). Do not worry about the electron carriers NAD+ and FAD; what you are doing is tracing the fate of the carbon atoms in the or ...
... construct a molecule of oxaloacetate (see page 553). Do not label these carbons. Perform the sequence of reactions in the citric acid cycle (reactions 1 through 8 on page 553). Do not worry about the electron carriers NAD+ and FAD; what you are doing is tracing the fate of the carbon atoms in the or ...
Note sheet Chap 5, Sect 3
... Stage 1: Break down of glucose The primary fuel for cellular respiration is __glucose__, which comes from the breakdown of __carbohydrates__. Glucose is broken down in the __cytoplasm__ during a process called ___glycolysis___, which is an enzyme assisted __anaerobic_ process (means no oxygen needed ...
... Stage 1: Break down of glucose The primary fuel for cellular respiration is __glucose__, which comes from the breakdown of __carbohydrates__. Glucose is broken down in the __cytoplasm__ during a process called ___glycolysis___, which is an enzyme assisted __anaerobic_ process (means no oxygen needed ...
Slide 1
... Liver cells have a responsibility to support blood glucose levels by first releasing glucose from their internal glycogen stores, and if necessary synthesizing glucose from amino acids. They will shut down glycolysis and rely on other energy sources for their own needs under these conditions. Liver ...
... Liver cells have a responsibility to support blood glucose levels by first releasing glucose from their internal glycogen stores, and if necessary synthesizing glucose from amino acids. They will shut down glycolysis and rely on other energy sources for their own needs under these conditions. Liver ...
Chapter 5: The Structure and Function of Macromolecules
... POLYMERIZATION REACTIONS: chemical reactions that link 2 or more small molecules ( larger molecules ( ...
... POLYMERIZATION REACTIONS: chemical reactions that link 2 or more small molecules ( larger molecules ( ...
student-notes-copy-unit-review
... 1.) ________________________ compounds- Contain ______________ and __________________ atoms 2.) ________________________ compounds- Can have one or the other, but do not contain both carbon and hydrogen atoms A. Most of your body’s molecules are _______________________ compounds. a. ________________ ...
... 1.) ________________________ compounds- Contain ______________ and __________________ atoms 2.) ________________________ compounds- Can have one or the other, but do not contain both carbon and hydrogen atoms A. Most of your body’s molecules are _______________________ compounds. a. ________________ ...
Glucose

Glucose is a sugar with the molecular formula C6H12O6. The name ""glucose"" (/ˈɡluːkoʊs/) comes from the Greek word γλευκος, meaning ""sweet wine, must"". The suffix ""-ose"" is a chemical classifier, denoting a carbohydrate. It is also known as dextrose or grape sugar. With 6 carbon atoms, it is classed as a hexose, a sub-category of monosaccharides. α-D-glucose is one of the 16 aldose stereoisomers. The D-isomer (D-glucose) occurs widely in nature, but the L-isomer (L-glucose) does not. Glucose is made during photosynthesis from water and carbon dioxide, using energy from sunlight. The reverse of the photosynthesis reaction, which releases this energy, is a very important source of power for cellular respiration. Glucose is stored as a polymer, in plants as starch and in animals as glycogen.