Procedure Notes
... due to the fact that the reaction is in equilibrium and esters are not very stable. The addition of water and heat from perspiration can cause the reaction to favor the reactants. Carboxylic acids tend to be associated with a foul smelling odor that one would not want to wear on their body. • Esters ...
... due to the fact that the reaction is in equilibrium and esters are not very stable. The addition of water and heat from perspiration can cause the reaction to favor the reactants. Carboxylic acids tend to be associated with a foul smelling odor that one would not want to wear on their body. • Esters ...
Organic Chemistry Chapter 1
... • The acidity can be explained by either the stability of the acids or the stability of the conjugate base using inductive effect or resonance effect • The stability of the acids: Inductive effect • The stability of the conjugate base: Both ...
... • The acidity can be explained by either the stability of the acids or the stability of the conjugate base using inductive effect or resonance effect • The stability of the acids: Inductive effect • The stability of the conjugate base: Both ...
Carboxylic Acid Derivatives and Nitriles
... with an alkyl halide. This reaction is of very limited scope (it only works with primary alkyl halides, but it does work particularly well with allylic and benzylic compounds), but it has some real synthetic potential, as will be discussed later. O O O ...
... with an alkyl halide. This reaction is of very limited scope (it only works with primary alkyl halides, but it does work particularly well with allylic and benzylic compounds), but it has some real synthetic potential, as will be discussed later. O O O ...
Ch 21 Carboxylic Acid Derivatives
... Ch 21 Carboxylic Acid Derivatives and Nu Acyl Subst’n Acid Derivatives and their Names - Acid Halides have a Cl or Br instead of OH. Replace “ic acid” with “yl halide”, such as propionyl chloride (a common name) and propanoyl bromide (a systematic name). Replace “carboxylic acid” with “carbonyl hali ...
... Ch 21 Carboxylic Acid Derivatives and Nu Acyl Subst’n Acid Derivatives and their Names - Acid Halides have a Cl or Br instead of OH. Replace “ic acid” with “yl halide”, such as propionyl chloride (a common name) and propanoyl bromide (a systematic name). Replace “carboxylic acid” with “carbonyl hali ...
Chapter 20: Carboxylic Acids and Nitriles
... I don’t like this description -- it is better, I believe, shown as on the next slide ...
... I don’t like this description -- it is better, I believe, shown as on the next slide ...
Chapter 19: Carboxylic Acid Derivatives
... Nucleophilic acyl substitution reactions of esters (Table 19.4). Esters are less reactive toward nucleophilic acyl substitution than acid chlorides or acid anhydrides. 1. Aminolysis (Ch.19.11): Esters react with ammonia, 1° and 2° amines to give amides ...
... Nucleophilic acyl substitution reactions of esters (Table 19.4). Esters are less reactive toward nucleophilic acyl substitution than acid chlorides or acid anhydrides. 1. Aminolysis (Ch.19.11): Esters react with ammonia, 1° and 2° amines to give amides ...
19_03_05rw
... Second stage is restoration of C=O by elimination. Complicating features of each stage involve acid-base chemistry. ...
... Second stage is restoration of C=O by elimination. Complicating features of each stage involve acid-base chemistry. ...
Slide 1
... below), a small molecule (often water) is produced. • Each monomer must also have two functional groups. • This can involve two different functional groups on the same monomer or more frequently, as in the examples below, two different monomers which have two identical groups on them. • One group of ...
... below), a small molecule (often water) is produced. • Each monomer must also have two functional groups. • This can involve two different functional groups on the same monomer or more frequently, as in the examples below, two different monomers which have two identical groups on them. • One group of ...
- professional publication
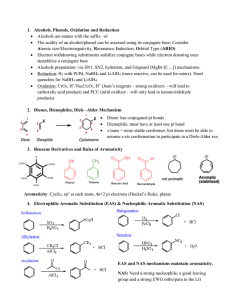
... Effect of Substituent Groups, Determination of Orientation, Determination of Relative Reactivity, Classification of Substituent Groups, Mechanism of Nitration, Sulphonation, Halogenation, Friedel Craft’s Alkylation and Friedel Craft’s Acylation, Reactivity and Orientation, Activating and Deactivatin ...
... Effect of Substituent Groups, Determination of Orientation, Determination of Relative Reactivity, Classification of Substituent Groups, Mechanism of Nitration, Sulphonation, Halogenation, Friedel Craft’s Alkylation and Friedel Craft’s Acylation, Reactivity and Orientation, Activating and Deactivatin ...
Unit 4_Carbonyl and carboxylic acid questions
... b. Draw the structures of the products formed when K and L are reduced with sodium ...
... b. Draw the structures of the products formed when K and L are reduced with sodium ...
Key Practice Exam 3
... alkanes, ketones and carboxylic acids. Provide rationale for your ranking. Carboxylic acid > alcohol > ketones > alkanes The ranking is based on the ability of these compounds to form hydrogen bonds (their attractive intermolecular forces). Carboxylic acids can form two hydrogen bonds (they are dimm ...
... alkanes, ketones and carboxylic acids. Provide rationale for your ranking. Carboxylic acid > alcohol > ketones > alkanes The ranking is based on the ability of these compounds to form hydrogen bonds (their attractive intermolecular forces). Carboxylic acids can form two hydrogen bonds (they are dimm ...
Fragrant Esters Esters are prepared in a reaction between a
... can prepare esters that produce different scents. Sometimes only small changes are needed to get a completely different fragrance. For example experiment 1 (see below) yields an ester that smells like apple. A slightly longer chain produces an orange scent. Having a branched chain leads to banana oi ...
... can prepare esters that produce different scents. Sometimes only small changes are needed to get a completely different fragrance. For example experiment 1 (see below) yields an ester that smells like apple. A slightly longer chain produces an orange scent. Having a branched chain leads to banana oi ...
Chapter 20 - people.vcu.edu
... o What would happen if you put the following reagents into a vessel? ...
... o What would happen if you put the following reagents into a vessel? ...
Chem 400 Review Chem 350 JJ.S17
... Atomic size/Electronegativity, Resonance, Induction, Orbital Type (ARIO) Electron withdrawing substituents stabilize conjugate bases while electron donating ones destabilize a conjugate base Alcohols preparation: via SN1, SN2, hydration, and Grignard (MgBr-[C…]) mechanisms Reduction: H2 with P ...
... Atomic size/Electronegativity, Resonance, Induction, Orbital Type (ARIO) Electron withdrawing substituents stabilize conjugate bases while electron donating ones destabilize a conjugate base Alcohols preparation: via SN1, SN2, hydration, and Grignard (MgBr-[C…]) mechanisms Reduction: H2 with P ...
26-3: Carboxylic Acids and Esters
... “-oic acid” to root word. 2. Name any branches as necessary. ...
... “-oic acid” to root word. 2. Name any branches as necessary. ...
International Arab Baccalaureate
... hydrolysis a molecule is cleaved into two parts by the addition of a molecule of water. Question 2: Amides can be hydrolyzed under acidic or basic conditions. Describe the reaction of hydrolysis and give the obtained compounds in acidic medium. ...
... hydrolysis a molecule is cleaved into two parts by the addition of a molecule of water. Question 2: Amides can be hydrolyzed under acidic or basic conditions. Describe the reaction of hydrolysis and give the obtained compounds in acidic medium. ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.