carboxylic acids
... carboxylic acid and alcohol – heated under reflux with conc. Sulfuric acid as catalyst. Excess acid is then neutralised with sodium carbonate, and anhydrous magnesium sulfate is added to remove excess water. Because the esters are more volatile than the rest of the mixture they can be removed by fra ...
... carboxylic acid and alcohol – heated under reflux with conc. Sulfuric acid as catalyst. Excess acid is then neutralised with sodium carbonate, and anhydrous magnesium sulfate is added to remove excess water. Because the esters are more volatile than the rest of the mixture they can be removed by fra ...
C h e m g u i d e ... CARBOXYLIC ACIDS: PREPARATION
... presence of an acid is 2Cr3+ + 7H2O Cr2O72- + 14H+ + 6e(i) Work out the electron-half-equation for the conversion of propan-1-ol to propanoic acid. (ii) Combine that with the electron-half-equation given above to work out the ionic equation for the overall reaction. e) Although it would be less comm ...
... presence of an acid is 2Cr3+ + 7H2O Cr2O72- + 14H+ + 6e(i) Work out the electron-half-equation for the conversion of propan-1-ol to propanoic acid. (ii) Combine that with the electron-half-equation given above to work out the ionic equation for the overall reaction. e) Although it would be less comm ...
Regents Unit 15b: Aldehydes, Ketones, Carboxylic Acids, & Esters
... used as sutures in internal surgery. ...
... used as sutures in internal surgery. ...
Diet and Exercise – Healthy Diet
... (Conc phosphoric acid or conc sulphuric acid) This is an elimination reaction Alcohol is heated under reflux with the catalyst for 40 minutes. ...
... (Conc phosphoric acid or conc sulphuric acid) This is an elimination reaction Alcohol is heated under reflux with the catalyst for 40 minutes. ...
Unit C Lesson 6 Carboxylic Acids And Esters
... Naming and Drawing Esters: Identify the root Identify the part of the ester that contains the C = O group. The root of the name of the ester is based on the name of the acid. Determine the name of the parent acid. Identify the suffix Remove the –oic acid from the name of the parent acid and repl ...
... Naming and Drawing Esters: Identify the root Identify the part of the ester that contains the C = O group. The root of the name of the ester is based on the name of the acid. Determine the name of the parent acid. Identify the suffix Remove the –oic acid from the name of the parent acid and repl ...
Word - chemmybear.com
... b) the molecule has some polarity c) they ionize completely in water solution ...
... b) the molecule has some polarity c) they ionize completely in water solution ...
CARBONYL COMPOUNDS ALDEHYDES AND KETONES
... carboxylic acid, forming a carboxylate anion, which does not react with an electron-rich nucleophile. ...
... carboxylic acid, forming a carboxylate anion, which does not react with an electron-rich nucleophile. ...
reactions of the carbonyl group in aldehydes and ketones
... atoms attracted to an electron-deficient centre, where it donates a pair of electrons to form a new covalent bond A curly arrow is a symbol used in reaction mechanisms to show the movement of an electron pair in the braking or forming of a covalent bond ...
... atoms attracted to an electron-deficient centre, where it donates a pair of electrons to form a new covalent bond A curly arrow is a symbol used in reaction mechanisms to show the movement of an electron pair in the braking or forming of a covalent bond ...
Chemistry of Nitrogen-containing Organic
... Reactions of amides 2 • DEHYDRATION Heating an amide with phosphorus(V) oxide dehydrates the compund. Using FSF, construct an equation for the dehydration of an amide with 4 carbon ...
... Reactions of amides 2 • DEHYDRATION Heating an amide with phosphorus(V) oxide dehydrates the compund. Using FSF, construct an equation for the dehydration of an amide with 4 carbon ...
carboxylic acid - Career Launcher
... primary (RNH2) and secondary amines (R2NH) The reaction with tertiary amines (R3N) gives an unstable species that cannot be isolated HCl is neutralized by the amine or an added base ...
... primary (RNH2) and secondary amines (R2NH) The reaction with tertiary amines (R3N) gives an unstable species that cannot be isolated HCl is neutralized by the amine or an added base ...
INTRODUCING ACYL CHLORIDES (acid
... ammonia and amines. All of these particular cases contain a very electronegative element with an active lone pair of electrons - either oxygen or nitrogen. Note: You can find details of all these reactions from the acyl chlorides menu (link below). If you are interested in exploring the general mech ...
... ammonia and amines. All of these particular cases contain a very electronegative element with an active lone pair of electrons - either oxygen or nitrogen. Note: You can find details of all these reactions from the acyl chlorides menu (link below). If you are interested in exploring the general mech ...
OrganicChem10 RxPaths SOLUTIONS (2014)
... E. ethanoic acid (from D) + ethanamine (from B) to N-ethylethanamide (condensation) CH3COOH + H2NCH2CH3 CH3CONCH2CH3 + H2O ...
... E. ethanoic acid (from D) + ethanamine (from B) to N-ethylethanamide (condensation) CH3COOH + H2NCH2CH3 CH3CONCH2CH3 + H2O ...
Acid-Base Theories Arrhenius Acids and Bases • An acid is a
... Acid-Base Theories Arrhenius Acids and Bases ...
... Acid-Base Theories Arrhenius Acids and Bases ...
Carboxylic Acid Derivatives 1. Background and Properties
... and halides consist of alkyl and/or aryl groups bonded to hydroxyl, alkoxyl, amino and halo substituents respectively. If these same functional groups are attached to an acyl group (RCO–) their properties are substantially changed, and they are designated ...
... and halides consist of alkyl and/or aryl groups bonded to hydroxyl, alkoxyl, amino and halo substituents respectively. If these same functional groups are attached to an acyl group (RCO–) their properties are substantially changed, and they are designated ...
CHE 322
... 3. (8) Give the complete mechanism that shows why the reaction of butanal with a cyclic 2° amine followed by heating with acid produces an enamine that is nucleophilic at butanal’s former α-carbon. ...
... 3. (8) Give the complete mechanism that shows why the reaction of butanal with a cyclic 2° amine followed by heating with acid produces an enamine that is nucleophilic at butanal’s former α-carbon. ...
ch18 by dr. dina
... Acid Chlorides Synthesis of Acid Chlorides Acid chlorides are made from carboxylic acids by reaction with thionyl chloride, phosphorus trichloride or phosphorus pentachloride These reagents work because they turn the hydroxyl group of the carboxylic acid into an excellent leaving group ...
... Acid Chlorides Synthesis of Acid Chlorides Acid chlorides are made from carboxylic acids by reaction with thionyl chloride, phosphorus trichloride or phosphorus pentachloride These reagents work because they turn the hydroxyl group of the carboxylic acid into an excellent leaving group ...
6.1.3 revision guide carboxylic acids and esters
... CH3CH2CO2CH3 + NaOH CH3CH2CO2- Na+ + CH3OH methyl propanoate sodium propanoate methanol The carboxylic acid salt product is the anion of the carboxylic acid. The anion is resistant to attack by weak nucleophiles such as alcohols, so the reaction is not reversible. ...
... CH3CH2CO2CH3 + NaOH CH3CH2CO2- Na+ + CH3OH methyl propanoate sodium propanoate methanol The carboxylic acid salt product is the anion of the carboxylic acid. The anion is resistant to attack by weak nucleophiles such as alcohols, so the reaction is not reversible. ...
Polyesters are condensation polymers.
... Note that sometimes other molecules (HCl for eg) are lost in other condensation reactions. It is the elimination of a molecule which makes it a condensation reaction, not the loss of water. ...
... Note that sometimes other molecules (HCl for eg) are lost in other condensation reactions. It is the elimination of a molecule which makes it a condensation reaction, not the loss of water. ...
organic chemistry ii
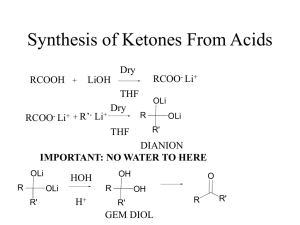
... Chapter 22: Carboxylic Acids and their Derivatives –Nucleophilic Acyl Substitution The common features of these classes of compounds is that they can all be hydrolyzed into carboxylic acids Syntheses of Carboxylic Acids: (i) Oxidation of aldehydes and 1o alcohols (remember 1o alcohols will only oxid ...
... Chapter 22: Carboxylic Acids and their Derivatives –Nucleophilic Acyl Substitution The common features of these classes of compounds is that they can all be hydrolyzed into carboxylic acids Syntheses of Carboxylic Acids: (i) Oxidation of aldehydes and 1o alcohols (remember 1o alcohols will only oxid ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.