chapter 2: reactions of organic compounds
... • Halogens in HCl, HBr or HI can substitute the OH group of alcohol or the reverse. • For Ex: CH3-CH2-OH + HCl CH3-CH2Cl + H2O • The reverse reaction: CH3-CH2Cl + OHCH3-CH2-OH + Cl(from water) ...
... • Halogens in HCl, HBr or HI can substitute the OH group of alcohol or the reverse. • For Ex: CH3-CH2-OH + HCl CH3-CH2Cl + H2O • The reverse reaction: CH3-CH2Cl + OHCH3-CH2-OH + Cl(from water) ...
102 Lecture Ch17
... • Aldehydes can be oxidized to carboxylic acids with most oxidizing agents, such as Tollens’reagent (AgNO3/NH3) - alcohols do not react with Tollens CrO3 OH ...
... • Aldehydes can be oxidized to carboxylic acids with most oxidizing agents, such as Tollens’reagent (AgNO3/NH3) - alcohols do not react with Tollens CrO3 OH ...
4.4 Formation of Esters from Carboxylic Acids and Alcohols
... (One might wonder how the ester gets formed in the first place, given that it is uphill from the carboxylic acid and alcohol molecules. In fact in biological systems the carboxylic acids are not the reactive molecule itself. The carboxylic acid is activated (energy level raised) by attaching a group ...
... (One might wonder how the ester gets formed in the first place, given that it is uphill from the carboxylic acid and alcohol molecules. In fact in biological systems the carboxylic acids are not the reactive molecule itself. The carboxylic acid is activated (energy level raised) by attaching a group ...
F324 summary - Macmillan Academy
... Reactions of carboxylic acids • Carboxylic acids react reversibly with alcohols, in the presence of an acid catalyst, to form esters – used in perfumes and flavourings. • Esters may be hydrolysed in the presence of: • hot alkalis to form the corresponding alcohol and the carboxylate salt • hot acid ...
... Reactions of carboxylic acids • Carboxylic acids react reversibly with alcohols, in the presence of an acid catalyst, to form esters – used in perfumes and flavourings. • Esters may be hydrolysed in the presence of: • hot alkalis to form the corresponding alcohol and the carboxylate salt • hot acid ...
Making esters from carboxylic acids and alcohols
... Doing the reactions On a test tube scale Carboxylic acids and alcohols are often warmed together in the presence of a few drops of concentrated sulphuric acid in order to observe the smell of the esters formed. You would normally use small quantities of everything heated in a test tube stood in a ho ...
... Doing the reactions On a test tube scale Carboxylic acids and alcohols are often warmed together in the presence of a few drops of concentrated sulphuric acid in order to observe the smell of the esters formed. You would normally use small quantities of everything heated in a test tube stood in a ho ...
www.xtremepapers.net
... effective if electronegative elements such as chlorine are present. Thus the chloroethanoic acids become increasingly more acidic as more chlorine atoms are present in the molecule. The reagent of preference is SOCl2, since both by-products are gases. Other possibilities are PCl3 and PCl5. The react ...
... effective if electronegative elements such as chlorine are present. Thus the chloroethanoic acids become increasingly more acidic as more chlorine atoms are present in the molecule. The reagent of preference is SOCl2, since both by-products are gases. Other possibilities are PCl3 and PCl5. The react ...
www.xtremepapers.net
... effective if electronegative elements such as chlorine are present. Thus the chloroethanoic acids become increasingly more acidic as more chlorine atoms are present in the molecule. The reagent of preference is SOCl2, since both by-products are gases. Other possibilities are PCl3 and PCl5. The react ...
... effective if electronegative elements such as chlorine are present. Thus the chloroethanoic acids become increasingly more acidic as more chlorine atoms are present in the molecule. The reagent of preference is SOCl2, since both by-products are gases. Other possibilities are PCl3 and PCl5. The react ...
File - Dr KHALID SHADID
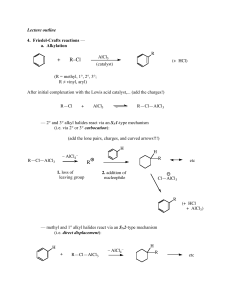
... elimination leads to regeneration of the carbon-oxygen double bond and to a substitution product. The overall process in the case of acyl substitution occurs, therefore, by a nucleophilic addition-elimination mechanism. ...
... elimination leads to regeneration of the carbon-oxygen double bond and to a substitution product. The overall process in the case of acyl substitution occurs, therefore, by a nucleophilic addition-elimination mechanism. ...
Ch. 16: Solutions - Quynh Nguyen Official Website
... (directly connected to an oxygen) first, use “yl” Then name the part that came from a carboxylic acid, use the “oate” suffix ...
... (directly connected to an oxygen) first, use “yl” Then name the part that came from a carboxylic acid, use the “oate” suffix ...
aldehydesketonescarb..
... contain dipole-dipole forces and are able to hydrogen bond. • How does the carboxyl group affect the physical properties of these compounds? • They are weak acids that will ionize slightly in water. • The H on the –OH is the acidic hydrogen and leaves when the acid is ionized. ...
... contain dipole-dipole forces and are able to hydrogen bond. • How does the carboxyl group affect the physical properties of these compounds? • They are weak acids that will ionize slightly in water. • The H on the –OH is the acidic hydrogen and leaves when the acid is ionized. ...
Chem263_Nov 25_notes_2010
... Reactivity of Carboxylic acid Derivatives Nucleophilic acyl substitution reactions usually take place in two steps: addition of the nucleophile and elimination of a leaving group. Although both steps can affect the overall rate of the reaction, it is generally the first step that is rate-limiting. ...
... Reactivity of Carboxylic acid Derivatives Nucleophilic acyl substitution reactions usually take place in two steps: addition of the nucleophile and elimination of a leaving group. Although both steps can affect the overall rate of the reaction, it is generally the first step that is rate-limiting. ...
1.7AMIDES
... Because the N atom is an electron withdrawing group, the C-N and H-N bonds are polar. As a result the physical properties of amides are similar to carboxylic acids. 1) Primary amides have two N-H bonds so they have even stronger hydrogen bonds than carboxylic acid. Secondary amides also have one N-H ...
... Because the N atom is an electron withdrawing group, the C-N and H-N bonds are polar. As a result the physical properties of amides are similar to carboxylic acids. 1) Primary amides have two N-H bonds so they have even stronger hydrogen bonds than carboxylic acid. Secondary amides also have one N-H ...
Document
... Asyou perform the experiment, record your obsmwations ii~ Table 50.1. 1. CAUTION: The acids used in this experime~tt are extremdy corrosive. Label five medium test tubes with the nnmbers 1-5. Put the robes in a test-tube rack. To each of the tubes, add 1 mL of a carboxylic acid and 1 mL of an alcoho ...
... Asyou perform the experiment, record your obsmwations ii~ Table 50.1. 1. CAUTION: The acids used in this experime~tt are extremdy corrosive. Label five medium test tubes with the nnmbers 1-5. Put the robes in a test-tube rack. To each of the tubes, add 1 mL of a carboxylic acid and 1 mL of an alcoho ...
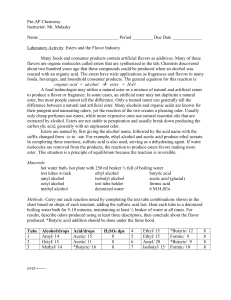
Pre-AP Chemistry
... only cheap perfumes use esters, while more expensive ones use natural essential oils that are extracted by alcohol. Esters are not stable to perspiration and usually break down producing the carboxylic acid, generally with an unpleasant odor. Esters are named by first giving the alcohol name, follow ...
... only cheap perfumes use esters, while more expensive ones use natural essential oils that are extracted by alcohol. Esters are not stable to perspiration and usually break down producing the carboxylic acid, generally with an unpleasant odor. Esters are named by first giving the alcohol name, follow ...
Organic Reactions Summary
... Adding groups (or atoms) to a chain by breaking a C=C bond Family Alkenes Alkenes ...
... Adding groups (or atoms) to a chain by breaking a C=C bond Family Alkenes Alkenes ...
synthesizing esters in the laboratory
... cucumber, and raspberry. Esters are easily made in the laboratory from their corresponding carboxylic acid and a condensation reaction with an alcohol. This reaction is condensed and catalyzed with concentrated (18M) sulfuric acid. The making of esters in the school laboratory is also an excellent w ...
... cucumber, and raspberry. Esters are easily made in the laboratory from their corresponding carboxylic acid and a condensation reaction with an alcohol. This reaction is condensed and catalyzed with concentrated (18M) sulfuric acid. The making of esters in the school laboratory is also an excellent w ...
اســـم المـــدرس: د
... ] Keton is the product of reaction between one equivalent of Grignard reagent and ester ...
... ] Keton is the product of reaction between one equivalent of Grignard reagent and ester ...
877-Alcohols Carboxylic acids and Esters Presentation
... The formula gives us clues about the structure of the molecule The oxygen and hydrogen in the alcohol functional group are bonded and so are written together ...
... The formula gives us clues about the structure of the molecule The oxygen and hydrogen in the alcohol functional group are bonded and so are written together ...
Organic Reactions 1
... Condensation is an organic reaction when two molecules combine, usually in the presence of a catalyst, with the elimination of water or some other simple molecule. Catalysts commonly used in condensation reactions include acids and bases. The combination of two identical molecules is known as self-c ...
... Condensation is an organic reaction when two molecules combine, usually in the presence of a catalyst, with the elimination of water or some other simple molecule. Catalysts commonly used in condensation reactions include acids and bases. The combination of two identical molecules is known as self-c ...
Organic Notes #5 - RX`ns - Winston Knoll Collegiate
... Condensation reactions can also be used to create polymers. These reactions produce compounds like polyesters and polyamides. These are often called synthetic polymers because they have been produced in a lab. Dacron, nylon and Teflon are just a few examples of polymers created using this process. N ...
... Condensation reactions can also be used to create polymers. These reactions produce compounds like polyesters and polyamides. These are often called synthetic polymers because they have been produced in a lab. Dacron, nylon and Teflon are just a few examples of polymers created using this process. N ...
MULTISTEP SYNTHESIS PROTECTING GROUPS
... 7. PROTECTING GROUP STRATEGY - Acetanilide is used as the starting material in these reactions, but at the end the amide group gets hydrolyzed into an amine group. Why not start the reaction sequence with aniline (below) rather than acetanilide, and save the last step? The answer is that the amino ...
... 7. PROTECTING GROUP STRATEGY - Acetanilide is used as the starting material in these reactions, but at the end the amide group gets hydrolyzed into an amine group. Why not start the reaction sequence with aniline (below) rather than acetanilide, and save the last step? The answer is that the amino ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.