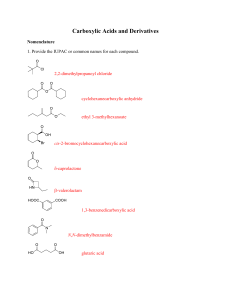
carboxylic acids esters amides (R
... So, remember...anhydrides react just about like acid chlorides. ...
... So, remember...anhydrides react just about like acid chlorides. ...
2009_outline_4
... 2. Oxidation of Alcohols and Aldehydes 3. Oxidation of Alkylbenzenes 4. Oxidation of Methyl Ketones (Haloform reaction) 5. Hydrolysis of Nitriles 6. Carbonation of Grignard Reagents (RMgX + CO2) C. Reactions 1. Salt formation 2. Ester formation 3. Reduction III. Acid Halides A. Physical Properties a ...
... 2. Oxidation of Alcohols and Aldehydes 3. Oxidation of Alkylbenzenes 4. Oxidation of Methyl Ketones (Haloform reaction) 5. Hydrolysis of Nitriles 6. Carbonation of Grignard Reagents (RMgX + CO2) C. Reactions 1. Salt formation 2. Ester formation 3. Reduction III. Acid Halides A. Physical Properties a ...
19-3 Esters and Anhydrides (PPT)
... With less basic nucleophiles, especially under acidic conditions, substitution through the addition-elimination mechanism may occur. In the esterification of a carboxylic acid, an alcohol and a carboxylic acid react in the presence of acid to form an ester and water. ...
... With less basic nucleophiles, especially under acidic conditions, substitution through the addition-elimination mechanism may occur. In the esterification of a carboxylic acid, an alcohol and a carboxylic acid react in the presence of acid to form an ester and water. ...
Exam 1
... Acid catalyzed ester & amide hydrolysis, Fisher esterification, trans esterification Hydroxide promoted ester hydrolysis Nucleophilic addition to aldehyde/ketone with Grignard reagents Determine reaction mechanisms from reaction outcome (see Mechanism work sheet). Be able to recognize the following ...
... Acid catalyzed ester & amide hydrolysis, Fisher esterification, trans esterification Hydroxide promoted ester hydrolysis Nucleophilic addition to aldehyde/ketone with Grignard reagents Determine reaction mechanisms from reaction outcome (see Mechanism work sheet). Be able to recognize the following ...
Carboxylic Acid
... You know Alkanes and Benzenes, and Alkynes and Alkenes, Amines and Alcohols, Aldehydes and Ketones...... But do you recall, the most famous functional group of all Carboxylic Acid, Has a carbonyl and a hydroxyl It loves to donate protons Then an anion is formed Of all the other acids It’s the most c ...
... You know Alkanes and Benzenes, and Alkynes and Alkenes, Amines and Alcohols, Aldehydes and Ketones...... But do you recall, the most famous functional group of all Carboxylic Acid, Has a carbonyl and a hydroxyl It loves to donate protons Then an anion is formed Of all the other acids It’s the most c ...
Answer keys
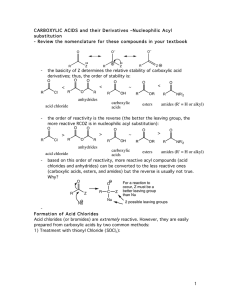
... 3. Rank the compounds in order of increasing reactivity in nucleophilic acyl substitution. ...
... 3. Rank the compounds in order of increasing reactivity in nucleophilic acyl substitution. ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.