A. Acid Halides
... This particular alcoholysis reaction is also called a transesterification reaction because one ester is converted to another ester (useful in converting liquid ester to solid ester; for identification by ...
... This particular alcoholysis reaction is also called a transesterification reaction because one ester is converted to another ester (useful in converting liquid ester to solid ester; for identification by ...
Annexure `CD-01` L T P/S SW/FW TOTAL CREDIT UNITS 3 1 2 0 5
... strength. Preparation of carboxylic acids. Reactions of carboxylic acids. Hell-Volhard-Zelinsky reaction. Reduction of carboxylic acids. Mechanism of decarboxylation, methods of formation and chemical reactions of unsaturated monocarboxylic acids. Structure and nomenclature of acid chlorides, este ...
... strength. Preparation of carboxylic acids. Reactions of carboxylic acids. Hell-Volhard-Zelinsky reaction. Reduction of carboxylic acids. Mechanism of decarboxylation, methods of formation and chemical reactions of unsaturated monocarboxylic acids. Structure and nomenclature of acid chlorides, este ...
Carbonyl Compounds
... Esters do not have any free OH groups and therefore they are unable to form hydrogen bonds, either with other ester molecules or with water. This means that: the boiling points of esters are lower than the boiling points of carboxylic acids of similar Mr esters are almost insoluble in water Este ...
... Esters do not have any free OH groups and therefore they are unable to form hydrogen bonds, either with other ester molecules or with water. This means that: the boiling points of esters are lower than the boiling points of carboxylic acids of similar Mr esters are almost insoluble in water Este ...
Synthesis of an Ester: Fischer Esterification The ester is synthesised
... As a result, they are partially extracted into the aqueous layer during work-up, reducing the amount recovered. ...
... As a result, they are partially extracted into the aqueous layer during work-up, reducing the amount recovered. ...
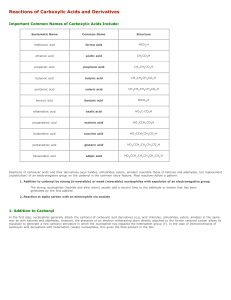
Functional Groups - SISIBChemistry2012
... ammonia. This is due to the alkyl group stabilizing, by acting as electron donating groups, which stabilize the cation (+ charge). Examine the Kb values. - However in amides, the lone pair of electrons on the nitrogen is involved in delocalized π-bonding with the electrons in the –C=O double bond. T ...
... ammonia. This is due to the alkyl group stabilizing, by acting as electron donating groups, which stabilize the cation (+ charge). Examine the Kb values. - However in amides, the lone pair of electrons on the nitrogen is involved in delocalized π-bonding with the electrons in the –C=O double bond. T ...
REVISED Review 3 - Bonham Chemistry
... * 34. The drugs Prilosec and Nexium both contain the same active ingredient. Nexium contains a single enantiomer but Prilosec is a racemic mixture. Which drug will be more effective if you receive a 20 mg dose of each? How much more effective? ...
... * 34. The drugs Prilosec and Nexium both contain the same active ingredient. Nexium contains a single enantiomer but Prilosec is a racemic mixture. Which drug will be more effective if you receive a 20 mg dose of each? How much more effective? ...
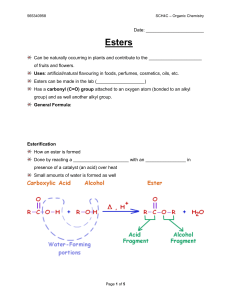
Esters - Mr. Lee`s Science
... Uses: artificial/natural flavouring in foods, perfumes, cosmetics, oils, etc. Esters can be made in the lab (___________________) Has a carbonyl (C=O) group attached to an oxygen atom (bonded to an alkyl group) and as well another alkyl group. General Formula: ...
... Uses: artificial/natural flavouring in foods, perfumes, cosmetics, oils, etc. Esters can be made in the lab (___________________) Has a carbonyl (C=O) group attached to an oxygen atom (bonded to an alkyl group) and as well another alkyl group. General Formula: ...
org test 1
... 2. Why is Sulphuric acid not used during reaction of alcohol with KI? 3. Why is preparation of ethers by acid catalysed dehydration of 2° and 3° alcohols not a suitable method? 4. Of benzene and phenol, which is more easily nitrated and why? 5. Ethers possess a net dipole moment even if they are sym ...
... 2. Why is Sulphuric acid not used during reaction of alcohol with KI? 3. Why is preparation of ethers by acid catalysed dehydration of 2° and 3° alcohols not a suitable method? 4. Of benzene and phenol, which is more easily nitrated and why? 5. Ethers possess a net dipole moment even if they are sym ...
Chapter 1 Structure and Bonding
... 1) Assign number 1 to carboxy carbon and number longest chain including it 2) Replace –ane ending of an alkane with –oic acid ending (or –dioic acid if two) 3) Number and name any other substituents 4) Carboxylic acids have priority over any other functional group studied 5) Cyclic (cycloalkanecarbo ...
... 1) Assign number 1 to carboxy carbon and number longest chain including it 2) Replace –ane ending of an alkane with –oic acid ending (or –dioic acid if two) 3) Number and name any other substituents 4) Carboxylic acids have priority over any other functional group studied 5) Cyclic (cycloalkanecarbo ...
Study Guide for Exam 4 Chapter 17
... Know basic terms, especially those discussed in class and in bold face print in the text From their structural or line-angle formulas, write IUPAC names of carboxylic acids, carboxylic salts, anhydrides, esters, and amides From their names, draw condensed structural or line-angle formulas of c ...
... Know basic terms, especially those discussed in class and in bold face print in the text From their structural or line-angle formulas, write IUPAC names of carboxylic acids, carboxylic salts, anhydrides, esters, and amides From their names, draw condensed structural or line-angle formulas of c ...
ppt
... aqueous acid or aqueous base to give carboxylic acids. The corresponding primary amide is an intermediate in the reaction. Base-promoted mechanism (Fig. 20.8, p. 865) ...
... aqueous acid or aqueous base to give carboxylic acids. The corresponding primary amide is an intermediate in the reaction. Base-promoted mechanism (Fig. 20.8, p. 865) ...
Professor Marina Gatti
... Alkyl nucleophilic substitution and elimination reactions. Properties of haloalkanes. SN2 and SN1 mechanisms: factors influencing the reaction kinetics. E1 and E2 mechanisms. Competition between substitution and elimination. Alcohols, phenols and ethers. Nomenclature and acid-base properties of alco ...
... Alkyl nucleophilic substitution and elimination reactions. Properties of haloalkanes. SN2 and SN1 mechanisms: factors influencing the reaction kinetics. E1 and E2 mechanisms. Competition between substitution and elimination. Alcohols, phenols and ethers. Nomenclature and acid-base properties of alco ...
AMINO ACIDS Ethan Secor, John N. Gitua (Mentor)
... The titanium complex was prepared in purified tetrahydrofuran using titanium(IV) isopropoxide and butyl lithium at low temperatures maintained by mixing acetone and dry ice. The imine was added to this mixture. The reaction mixture was gradually warmed to room temperature. Carbon dioxide was introdu ...
... The titanium complex was prepared in purified tetrahydrofuran using titanium(IV) isopropoxide and butyl lithium at low temperatures maintained by mixing acetone and dry ice. The imine was added to this mixture. The reaction mixture was gradually warmed to room temperature. Carbon dioxide was introdu ...
Pre Ch15 HW
... regions of charge imbalance that result in functional groups (§15.1) 3. Structures and names of alkanes, alkenes, and alkynes (§15.2) 4. The distinctions among constitutional, optical, and geometric isomers (§15.2) 5. The importance of optical isomerism in organisms (§15.2) 6. The effect of restrict ...
... regions of charge imbalance that result in functional groups (§15.1) 3. Structures and names of alkanes, alkenes, and alkynes (§15.2) 4. The distinctions among constitutional, optical, and geometric isomers (§15.2) 5. The importance of optical isomerism in organisms (§15.2) 6. The effect of restrict ...
Honors Chemistry Organic Chemistry
... b. carcinogen in burned meat and cigarettes c. organic bases d. hydrogenation e. hydroxyl f. carboxyl g. carbonyl h. containing benzene or benzene-like structures i. from wood distillation / metabolized into formaldehyde j. benzene as a substituent k. reaction in the formation of esters l. phenol ...
... b. carcinogen in burned meat and cigarettes c. organic bases d. hydrogenation e. hydroxyl f. carboxyl g. carbonyl h. containing benzene or benzene-like structures i. from wood distillation / metabolized into formaldehyde j. benzene as a substituent k. reaction in the formation of esters l. phenol ...
Esters - Phillips Scientific Methods
... FYI- Back to Biology: The below molecule is produced in the first step of Glycolysis in Cellular Respiration (oxidation of glucose), as ATP ADP to start the process, and the phosphate bonds to glucose (see p. 750) 5. Phosphate esters are important biological molecules. Shown below is the structure ...
... FYI- Back to Biology: The below molecule is produced in the first step of Glycolysis in Cellular Respiration (oxidation of glucose), as ATP ADP to start the process, and the phosphate bonds to glucose (see p. 750) 5. Phosphate esters are important biological molecules. Shown below is the structure ...
solutions
... 11) Zaitsev’s rule enables one to predict the major product of a(n) __________ reaction. a) condensation b) saponification c) oxidation d) elimination ...
... 11) Zaitsev’s rule enables one to predict the major product of a(n) __________ reaction. a) condensation b) saponification c) oxidation d) elimination ...
CH19 Aldehydes and Ketones
... – Hydride (H-) and Grignard (R-) as Nucleophiles – Water addition (hydrate/diol/geminal diol formation) – Alcohol addition (acetal formation) – Amine addition (imine formation) ...
... – Hydride (H-) and Grignard (R-) as Nucleophiles – Water addition (hydrate/diol/geminal diol formation) – Alcohol addition (acetal formation) – Amine addition (imine formation) ...
Objective Reaction Type Structural Feature How to figure out how reactants react?
... sulfhydryl-containing enzymes such as those found in the eye are a prime target, the Army says. Effects of CS exposure include an extreme burning sensation in the eyes with a copious flow of tears, coughing, sneezing, a perception of chest tightness, and dizziness. Most of these effects subside with ...
... sulfhydryl-containing enzymes such as those found in the eye are a prime target, the Army says. Effects of CS exposure include an extreme burning sensation in the eyes with a copious flow of tears, coughing, sneezing, a perception of chest tightness, and dizziness. Most of these effects subside with ...
Table
... Alcohol+(O)aldehyde+(O)Carboxylic acid Oxidation reaction; add (O) Carboxylic acid+alcoholester+H2O Condensation Reaction Pathway to other compounds Ester+NaOH sodium salt of acid+ alcohol Hydrolysis; saponification Preparation Amines: RX+NH3 amine + HX RX+R2NH amine +HX Amide +H2Ocarboxyic ...
... Alcohol+(O)aldehyde+(O)Carboxylic acid Oxidation reaction; add (O) Carboxylic acid+alcoholester+H2O Condensation Reaction Pathway to other compounds Ester+NaOH sodium salt of acid+ alcohol Hydrolysis; saponification Preparation Amines: RX+NH3 amine + HX RX+R2NH amine +HX Amide +H2Ocarboxyic ...
Slides for Chapter 1-4 - Department of Chemistry and Physics
... Reaction is with inversion at reacting center Follows second order reaction kinetics Ingold nomenclature to describe characteristic step: ...
... Reaction is with inversion at reacting center Follows second order reaction kinetics Ingold nomenclature to describe characteristic step: ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.