Chapter 20: Carboxylic Acids
... Condensation of acids with amines (20-12) The reaction of carboxylic acids with amines is a simple acid/base reaction forming an ammonium salt. However, under more extreme conditions, when heat is applied it is possible to eliminate water from this salt and form amide. ...
... Condensation of acids with amines (20-12) The reaction of carboxylic acids with amines is a simple acid/base reaction forming an ammonium salt. However, under more extreme conditions, when heat is applied it is possible to eliminate water from this salt and form amide. ...
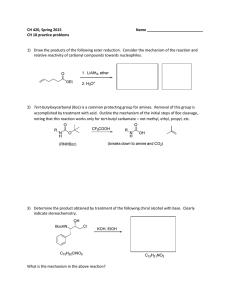
CH 420, Spring 2015 Name ___________________________ CH 18 practice problems
... 7) Rank the following compounds according to their relative acidity: cyclohexanol, phenol, pmethoxyphenol, p-nitrophenol. ...
... 7) Rank the following compounds according to their relative acidity: cyclohexanol, phenol, pmethoxyphenol, p-nitrophenol. ...
File
... (dehydration) but this reaction is favored using a large excess of concentrated acid (HCl and zinc chloride) • we can distinguish between 1°, 2° and 3° alcohols by their different rates of reaction using the Lucas Test • order of reactivity: – tertiary > secondary > primary ...
... (dehydration) but this reaction is favored using a large excess of concentrated acid (HCl and zinc chloride) • we can distinguish between 1°, 2° and 3° alcohols by their different rates of reaction using the Lucas Test • order of reactivity: – tertiary > secondary > primary ...
CHEMISTRY 3
... Lipids are triglycerides and their structure consists of a molecule of glycerol bonded through three ester linkages to three “fatty” acids. ...
... Lipids are triglycerides and their structure consists of a molecule of glycerol bonded through three ester linkages to three “fatty” acids. ...
CHEM1102 Worksheet 7: Reactions of Carbonyls and Acid
... Grignard reagents (RMgBr) are excellent nucleophiles, and are a very good way to form new carbon-carbon bonds. ...
... Grignard reagents (RMgBr) are excellent nucleophiles, and are a very good way to form new carbon-carbon bonds. ...
KS4 Unit 3 Organic Learning Objectives
... Esters have the functional group –COO–. They are volatile compounds with distinctive smells and are used as flavourings and perfumes. ...
... Esters have the functional group –COO–. They are volatile compounds with distinctive smells and are used as flavourings and perfumes. ...
Carboxylic Acid Derivatives
... groups: R- and H- are poor leaving groups.] A generalized reaction mechanism under basic conditions, BAC2 (basic, acyl, bimolecular) looks like this, where Y is a leaving group – one of the boxed groups above: trigonal O R C ...
... groups: R- and H- are poor leaving groups.] A generalized reaction mechanism under basic conditions, BAC2 (basic, acyl, bimolecular) looks like this, where Y is a leaving group – one of the boxed groups above: trigonal O R C ...
C h e m g u i d e ... ALCOHOLS: ESTERIFICATION
... 4. Esters can be also be made by reacting alcohols with acyl chlorides such as ethanoyl chloride, CH3COCl. a) Suggest a disadvantage of making, say, ethyl ethanoate using this reaction. b) What advantage(s) does the method have over the reaction between ethanol and ethanoic acid? c) Write the equati ...
... 4. Esters can be also be made by reacting alcohols with acyl chlorides such as ethanoyl chloride, CH3COCl. a) Suggest a disadvantage of making, say, ethyl ethanoate using this reaction. b) What advantage(s) does the method have over the reaction between ethanol and ethanoic acid? c) Write the equati ...
Addition/elimination under acidic conditions
... 2. Although the stability of the starting materials and products are about the same in the reaction above, the equilibrium lies far to the right. Explain how this is possible. How could this reaction be forced back to the left? (Hint: What is the byproduct of the reaction?) ...
... 2. Although the stability of the starting materials and products are about the same in the reaction above, the equilibrium lies far to the right. Explain how this is possible. How could this reaction be forced back to the left? (Hint: What is the byproduct of the reaction?) ...
C h e m g u id e –... ACID ANHYDRIDES: INTRODUCTION
... 1. a) van der Waals dispersion forces and dipole-dipole interactions. b) It reacts with moisture in the air or your nose to produce ethanoic acid. c) Ethanoic anhydride reacts with water and doesn’t simply dissolve in it. 2. Ethanoyl chloride reacts with molecules containing a hydrogen atom attached ...
... 1. a) van der Waals dispersion forces and dipole-dipole interactions. b) It reacts with moisture in the air or your nose to produce ethanoic acid. c) Ethanoic anhydride reacts with water and doesn’t simply dissolve in it. 2. Ethanoyl chloride reacts with molecules containing a hydrogen atom attached ...
4.5 Topic Checklist Carbonyl Compounds
... basis of a simple chemical test to distinguish between aldehydes and ketones (e.g. Fehling’s solution and Tollens’ reagent) appreciate the hazards of synthesis using HCN/KCN know that aldehydes can be reduced to primary alcohols and ketones to secondary alcohols using reducing agents such as NaBH4. ...
... basis of a simple chemical test to distinguish between aldehydes and ketones (e.g. Fehling’s solution and Tollens’ reagent) appreciate the hazards of synthesis using HCN/KCN know that aldehydes can be reduced to primary alcohols and ketones to secondary alcohols using reducing agents such as NaBH4. ...
review sheet
... Which carboxylic acid is most acidic? (Which one forms the most stable anion?) Why? Which of the carboxylic acid derivatives (acid chloride, acid anhydride, ester, amide) is the most stable? Which is the most reactive? Why are acid chlorides highly reactive? What is the mechanism for nucleophilic ac ...
... Which carboxylic acid is most acidic? (Which one forms the most stable anion?) Why? Which of the carboxylic acid derivatives (acid chloride, acid anhydride, ester, amide) is the most stable? Which is the most reactive? Why are acid chlorides highly reactive? What is the mechanism for nucleophilic ac ...
Chapter 21: Carboxylic Acid Derivatives
... however this reaction is easy since the leaving group Cl- is a weaker base than NH2300 ...
... however this reaction is easy since the leaving group Cl- is a weaker base than NH2300 ...
Document
... Reaction of acid chlorides • A good example of this mechanism is reaction of acid chlorides with amines or alcohols – the first step is as we have seen previously. ...
... Reaction of acid chlorides • A good example of this mechanism is reaction of acid chlorides with amines or alcohols – the first step is as we have seen previously. ...
Carboxylic Acid Derivatives: Nucleophilic Acyl Substitution
... intermediate (Mechanism 20.4) as in acid-catalyzed hydrolysis. Unlike acid-catalyzed hydrolysis, saponification is irreversible because the carboxylic acid is deprotonated under the reaction conditions. ...
... intermediate (Mechanism 20.4) as in acid-catalyzed hydrolysis. Unlike acid-catalyzed hydrolysis, saponification is irreversible because the carboxylic acid is deprotonated under the reaction conditions. ...
Carboxylic Acid Derivatives
... A similar procedure is used to make amides from acyl chlorides and amines (the amine must have at least one hydrogen attached to the nitrogen). ...
... A similar procedure is used to make amides from acyl chlorides and amines (the amine must have at least one hydrogen attached to the nitrogen). ...
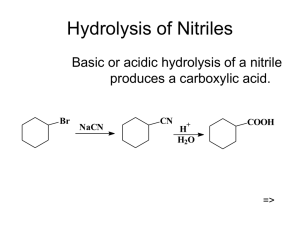
Hydrolysis of Nitriles
... An activated form of the carboxylic acid. Chloride is a good leaving group, so undergoes acyl substitution easily. To synthesize acid chlorides use thionyl chloride or oxalyl chloride with the acid. O ...
... An activated form of the carboxylic acid. Chloride is a good leaving group, so undergoes acyl substitution easily. To synthesize acid chlorides use thionyl chloride or oxalyl chloride with the acid. O ...
carboxylic acids - La Salle University
... Grignard reagents react with dry ice (solid CO2) followed by aqueous acid workup to give carboxylic acids. CO2 can be thought of as a being a dicarbonyl compound : O=C=O Note that the carboxylic acid contains one extra C atom compared to the original halide from which the Grignard reagent was prepar ...
... Grignard reagents react with dry ice (solid CO2) followed by aqueous acid workup to give carboxylic acids. CO2 can be thought of as a being a dicarbonyl compound : O=C=O Note that the carboxylic acid contains one extra C atom compared to the original halide from which the Grignard reagent was prepar ...
Answers
... forced back to the left? (Hint: What is the byproduct of the reaction?) This equilibrium is possible through the addition of acid to the electrophile, making it a better electrophile. This means it does not need as strong of a nucleophile to react. The reaction could be forced back to the left is wa ...
... forced back to the left? (Hint: What is the byproduct of the reaction?) This equilibrium is possible through the addition of acid to the electrophile, making it a better electrophile. This means it does not need as strong of a nucleophile to react. The reaction could be forced back to the left is wa ...
CHE 312 Answers in BOLD RED EXAM 1 KEY (Ch. 16
... The most commonly available and useful acid derivatives for use in other reactions are the A. esters B. acyl halides C. amides D. anhydrides ...
... The most commonly available and useful acid derivatives for use in other reactions are the A. esters B. acyl halides C. amides D. anhydrides ...
Name: Chem 22 Final exam Spring `00 What product is formed when
... e) addtion of a hydride ion and a proton more or less at the same time 18. Which of the following describes “reductive amination?” a) an aldehyde or a ketone + a tertiary amine + H2/zeolite b) an aldehyde or a ketone + ammonia or a primary or a secondary amine + ...
... e) addtion of a hydride ion and a proton more or less at the same time 18. Which of the following describes “reductive amination?” a) an aldehyde or a ketone + a tertiary amine + H2/zeolite b) an aldehyde or a ketone + ammonia or a primary or a secondary amine + ...
Derivatives of Carboxylic Acids
... • Carboxylic acids and their derivatives can be expressed as variation of a single formula in which electronegative atom (O, N, or halogen) is bonded to an acyl group (RC=O) • Carboxylic acid -OH is attached to acyl group. • Ester -OR, alkoxy is attaches to acyl group ...
... • Carboxylic acids and their derivatives can be expressed as variation of a single formula in which electronegative atom (O, N, or halogen) is bonded to an acyl group (RC=O) • Carboxylic acid -OH is attached to acyl group. • Ester -OR, alkoxy is attaches to acyl group ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.