Chapter 20: Carboxylic Acids and Nitriles
... Must enhance reactivity Convert OH into a better leaving group Specific reagents can produce acid chlorides, anhydrides, esters, amides ...
... Must enhance reactivity Convert OH into a better leaving group Specific reagents can produce acid chlorides, anhydrides, esters, amides ...
Carboxylic Acid Derivatives and Nucleophilic Acyl Substitution
... • Reaction with thionyl chloride, SOCl2 ...
... • Reaction with thionyl chloride, SOCl2 ...
Chapter 21 Carboxylic Acid Derivatives
... • For 1 amide, drop -ic or -oic acid from the carboxylic acid name, add -amide. • For 2 and 3 amides, the alkyl groups bonded to nitrogen are named with Nto indicate their position. O CH3 ...
... • For 1 amide, drop -ic or -oic acid from the carboxylic acid name, add -amide. • For 2 and 3 amides, the alkyl groups bonded to nitrogen are named with Nto indicate their position. O CH3 ...
In the preparation of the esters given in this experiment
... a. Calculate the mole percent composition of the mixture. Assume that the response of the detector (area per mole) is the same for each component. b. Calculate the weight percent composition of the mixture, using the same assumptions as in part a. 6. When tert-pentyl bromide is treated with 80% etha ...
... a. Calculate the mole percent composition of the mixture. Assume that the response of the detector (area per mole) is the same for each component. b. Calculate the weight percent composition of the mixture, using the same assumptions as in part a. 6. When tert-pentyl bromide is treated with 80% etha ...
Acid Basics
... Acids are rated on their strength, or ability to give up protons, on the pH scale. pH stands for power of hydrogen and works like this: the pH scale goes from 1 (strongest acid) to 14 (weakest acid) with 7 being neutral. Water is exactly 7. By the way, strong acids are weak bases and vice versa. Fin ...
... Acids are rated on their strength, or ability to give up protons, on the pH scale. pH stands for power of hydrogen and works like this: the pH scale goes from 1 (strongest acid) to 14 (weakest acid) with 7 being neutral. Water is exactly 7. By the way, strong acids are weak bases and vice versa. Fin ...
Chapter 21: Carboxylic acid Derivatives I. Introduction
... Esters and acid chlorides react through the addition elimination mechanism, generating aldehydes, that are quickly reduced to the primary alcohols. ...
... Esters and acid chlorides react through the addition elimination mechanism, generating aldehydes, that are quickly reduced to the primary alcohols. ...
Organic Chemistry Fifth Edition
... Reduction with LiAlH4 (Section 19.13) With ammonia and amines (Sections 19.11) ...
... Reduction with LiAlH4 (Section 19.13) With ammonia and amines (Sections 19.11) ...
Slide 1
... Next week is the final week of practicals Make sure you are up to date with your reports by next week – all reports Final reports must be handed up no later than one week after completion of final practical – otherwise marks will be forfeited ...
... Next week is the final week of practicals Make sure you are up to date with your reports by next week – all reports Final reports must be handed up no later than one week after completion of final practical – otherwise marks will be forfeited ...
Reactions to know from Chapters 17, 18, 19
... Starting with either an aldehyde or a ketone, you can see that the hemiacetals formed are characterized by having a carbon bonded to an OH- group and an OR- group. Here, the oxygen of the alcohol attacks and bonds with the carbonyl carbon of the aldehyde or ketone If the alcohol group and the ...
... Starting with either an aldehyde or a ketone, you can see that the hemiacetals formed are characterized by having a carbon bonded to an OH- group and an OR- group. Here, the oxygen of the alcohol attacks and bonds with the carbonyl carbon of the aldehyde or ketone If the alcohol group and the ...
Worksheet 1 - Oregon State chemistry
... What is meant by a condensation reaction? Give an example. Water is lost during a condensation reaction. Examples include: the formation of an ester from an alcohol and a carboxylic acid, the formation of an amide from an amine and a carboxylic acid. ...
... What is meant by a condensation reaction? Give an example. Water is lost during a condensation reaction. Examples include: the formation of an ester from an alcohol and a carboxylic acid, the formation of an amide from an amine and a carboxylic acid. ...
Experiment 7
... A) Methyl salicylate is an ester having the signature fragrance of wintergreen, and is synthesized from salicylic acid and methanol. B) Synthesize a custom ester of your choice & describe the fragrance. You’ll need to choose a starting carboxylic acid & alcohol to prepare the desired fragrance. 1. A ...
... A) Methyl salicylate is an ester having the signature fragrance of wintergreen, and is synthesized from salicylic acid and methanol. B) Synthesize a custom ester of your choice & describe the fragrance. You’ll need to choose a starting carboxylic acid & alcohol to prepare the desired fragrance. 1. A ...
Preparation of alkyl halides There are lots of ways to make alkyl
... You use some kind of base in each of these cases (either triethylamine or pyridine) so that you can neutralize the acid that is formed during the reaction. The key feature of these reactions is that you are converting OH into a much better leaving group as well. 2. Preparation o ...
... You use some kind of base in each of these cases (either triethylamine or pyridine) so that you can neutralize the acid that is formed during the reaction. The key feature of these reactions is that you are converting OH into a much better leaving group as well. 2. Preparation o ...
Aldehid dan Keton
... Nucleophilic Addition of H2O: Hydration Aldehydes and ketones react reversibly with water to give ...
... Nucleophilic Addition of H2O: Hydration Aldehydes and ketones react reversibly with water to give ...
Solid phase reactions II
... Reduction of carboxylic acids (Weinreb amide) Oxidation of alcohols ...
... Reduction of carboxylic acids (Weinreb amide) Oxidation of alcohols ...
study note 3 33
... where X is a side group. When X is H the amino acid is called “glycine”, when X is CH3 the name is “alanine”. In what way are amino acids Amino acids are the building blocks of proteins (a.k.a. important? How do they combine? polypeptides). Amino acids combine via a peptide bond. A peptide bond form ...
... where X is a side group. When X is H the amino acid is called “glycine”, when X is CH3 the name is “alanine”. In what way are amino acids Amino acids are the building blocks of proteins (a.k.a. important? How do they combine? polypeptides). Amino acids combine via a peptide bond. A peptide bond form ...
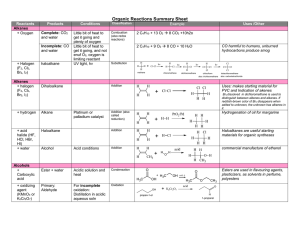
Organic Reactions Summary Sheet
... 2 C4H10 + 13 O2 8 CO2 +10h2o 2 C4H10 + 9 O2 8 CO + 10 H2O ...
... 2 C4H10 + 13 O2 8 CO2 +10h2o 2 C4H10 + 9 O2 8 CO + 10 H2O ...
New aniline photocage for carboxylic acids
... Wang et al. have recently reported an interesting example of photocage for alcohols, based on m-hydroxyaniline ethers.[5] We decided to further develop this type of protecting group and expand their application for the protection of carboxylic acids. A systematic study of photochemical reactivity wa ...
... Wang et al. have recently reported an interesting example of photocage for alcohols, based on m-hydroxyaniline ethers.[5] We decided to further develop this type of protecting group and expand their application for the protection of carboxylic acids. A systematic study of photochemical reactivity wa ...
Chemistry - Choithram School
... LONG ANSWER TYPE Give reason for the following : i)Haloalkanes react with KCN to form alkyl cyanides as main product while AgCN forms isocyanides as the major product. ii) Haloarenes are much less reactive than haloalkanes towards nucleophilic substitution reactions. iii)Reaction of alkyl chlorides ...
... LONG ANSWER TYPE Give reason for the following : i)Haloalkanes react with KCN to form alkyl cyanides as main product while AgCN forms isocyanides as the major product. ii) Haloarenes are much less reactive than haloalkanes towards nucleophilic substitution reactions. iii)Reaction of alkyl chlorides ...
Organic Chemistry - Unit 2
... aryl halide – this is a halocarbon in which there is a halogen substituent on an arene ring. (Substituted benzene) In all of these compounds, the halogen is considered to be the functional group. Very few halocarbons exist in nature but they are easily produced in substitution reactions. Alcohols Al ...
... aryl halide – this is a halocarbon in which there is a halogen substituent on an arene ring. (Substituted benzene) In all of these compounds, the halogen is considered to be the functional group. Very few halocarbons exist in nature but they are easily produced in substitution reactions. Alcohols Al ...
2015 CH 420 Take Home Quiz 3 March 24
... an arrow to the more nucleophilic nitrogen atom in the hydrazine substrate. In addition, circle the most electrophilic carbonyl in the 1,3-diketone substrate. ...
... an arrow to the more nucleophilic nitrogen atom in the hydrazine substrate. In addition, circle the most electrophilic carbonyl in the 1,3-diketone substrate. ...
Carboxylic Derivatives - University of Nebraska Omaha
... either aqueous acid or aqueous base. • Hydrolysis in aqueous acid is the reverse of Fischer esterification. • The key step in the mechanism of hydrolysis is formation of a tetrahedral carbonyl addition intermediate followed by its collapse (see the next screen for details). ...
... either aqueous acid or aqueous base. • Hydrolysis in aqueous acid is the reverse of Fischer esterification. • The key step in the mechanism of hydrolysis is formation of a tetrahedral carbonyl addition intermediate followed by its collapse (see the next screen for details). ...
Organic Reactions 2.1- 2.3 - mccormack-sch4u-2013
... • Halogens in HCl, HBr or HI can substitute the OH group of alcohol or the reverse. • For Ex: CH3-CH2-OH + HCl CH3-CH2Cl + H2O • The reverse reaction: CH3-CH2Cl + OHCH3-CH2-OH + Cl(from water) ...
... • Halogens in HCl, HBr or HI can substitute the OH group of alcohol or the reverse. • For Ex: CH3-CH2-OH + HCl CH3-CH2Cl + H2O • The reverse reaction: CH3-CH2Cl + OHCH3-CH2-OH + Cl(from water) ...
Acidic and Basic Character of Carboxylic Acids
... Electron-withdrawing substituents increase the acidity of carboxylic acids. The inductive effect of electron-withdrawing groups close to the carboxy group causes an increase in acidity. Three electron-withdrawing groups on the -carbon sometimes results in acidity near that of some inorganic acids. ...
... Electron-withdrawing substituents increase the acidity of carboxylic acids. The inductive effect of electron-withdrawing groups close to the carboxy group causes an increase in acidity. Three electron-withdrawing groups on the -carbon sometimes results in acidity near that of some inorganic acids. ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.