chm 103 general chemistry
... c. React an amine with a carboxylic acid to eliminate water and generate an amide Amide type (a) is the reaction product of ammonia with a carboxylic acid, for example: CH3COOH + NH3 CH3CONH2 + H2O Amide type (b) is the reaction product of a primary amine, such as methyl amine, with a carboxyl ...
... c. React an amine with a carboxylic acid to eliminate water and generate an amide Amide type (a) is the reaction product of ammonia with a carboxylic acid, for example: CH3COOH + NH3 CH3CONH2 + H2O Amide type (b) is the reaction product of a primary amine, such as methyl amine, with a carboxyl ...
Lecture 9a - University of California, Los Angeles
... -OH (pKa~16-18 (alcohol), pKa~8-12 (phenols)), -NHx (pKa~35), -C≡CH (pKa~25), -SH (pKa~9-12), -COOH (pKa~3-5) Some functional groups react with the reagent because they contain electrophilic atoms: -CHO, -COR, -CONR2, -COOR, -C≡N, -NO2, -SO2R, epoxides (ring opening) If more than one of these fu ...
... -OH (pKa~16-18 (alcohol), pKa~8-12 (phenols)), -NHx (pKa~35), -C≡CH (pKa~25), -SH (pKa~9-12), -COOH (pKa~3-5) Some functional groups react with the reagent because they contain electrophilic atoms: -CHO, -COR, -CONR2, -COOR, -C≡N, -NO2, -SO2R, epoxides (ring opening) If more than one of these fu ...
Alcohol Worksheet Key
... 4-chlorophenol is more acidic than 3-chlorophenol due to the inductive effect of the chlorine at position 4. The negative charge is not delocalized onto postions 3 or 5, so electron withdrawing groups at position 3 and 5 will have very little effect on the acidity of the phenol. ...
... 4-chlorophenol is more acidic than 3-chlorophenol due to the inductive effect of the chlorine at position 4. The negative charge is not delocalized onto postions 3 or 5, so electron withdrawing groups at position 3 and 5 will have very little effect on the acidity of the phenol. ...
Carboxylic Acid Derivatives
... 4-Dimethylaminopyridine (DMAP), used in the example below, is a particularly effective catalyst of the acylation process. ...
... 4-Dimethylaminopyridine (DMAP), used in the example below, is a particularly effective catalyst of the acylation process. ...
Organic Tutorial 1st Year HT01
... consider Michael addition and the factors which determine 1,2 or 1,4 addition ...
... consider Michael addition and the factors which determine 1,2 or 1,4 addition ...
C3 Knowledge Test – Higher Tier 1. Why was Mendeleev`s periodic
... Write a word and symbol equation for a group 7 element with a group 1 element. How and why does reactivity change within the halogen group? Name five ways transition elements differ to group 1 metals? What causes water to be ‘hard’? What forms when hard water reacts with soap? Describe the hea ...
... Write a word and symbol equation for a group 7 element with a group 1 element. How and why does reactivity change within the halogen group? Name five ways transition elements differ to group 1 metals? What causes water to be ‘hard’? What forms when hard water reacts with soap? Describe the hea ...
H1- Functional Groups Theory Sheet Alcohol An alcohol group
... A functional group is a specific group of atoms or bonds that form part of an organic molecule. A certain type of functional group will undergo similar chemical reactions even when attached to different sized molecules; however neighbouring groups may affect this reactivity. A molecule can have mult ...
... A functional group is a specific group of atoms or bonds that form part of an organic molecule. A certain type of functional group will undergo similar chemical reactions even when attached to different sized molecules; however neighbouring groups may affect this reactivity. A molecule can have mult ...
Benzocaine Synthesis via Esterification
... (along with the stir bar) into a 200 mL beaker containing 30 mL of ice water. While stirring this mixture, slowly add approximately 10 mL of a 10% Na2CO3 solution. Gas evolution will be observed as the acid is neutralized; the final pH should be approximately 8. Vacuum filter the resulting precipita ...
... (along with the stir bar) into a 200 mL beaker containing 30 mL of ice water. While stirring this mixture, slowly add approximately 10 mL of a 10% Na2CO3 solution. Gas evolution will be observed as the acid is neutralized; the final pH should be approximately 8. Vacuum filter the resulting precipita ...
questions on the dehydration of alcohols
... Chemguide – questions ALCOHOLS: DEHYDRATION 1. a) Ethanol can be converted into ethene by passing the vapour over hot aluminium oxide, and collecting the ethene over water. Write the equation for the reaction. b) Ethanol can also be converted into ethene by heating it at 170°C with excess concentrat ...
... Chemguide – questions ALCOHOLS: DEHYDRATION 1. a) Ethanol can be converted into ethene by passing the vapour over hot aluminium oxide, and collecting the ethene over water. Write the equation for the reaction. b) Ethanol can also be converted into ethene by heating it at 170°C with excess concentrat ...
Organic Chemistry Review
... What is the functional group of a carboxylic acid? What is the ending given to carboxylic acid names? What is the name of these organics? ...
... What is the functional group of a carboxylic acid? What is the ending given to carboxylic acid names? What is the name of these organics? ...
CHE 145-381 – TEST #2 SPRING 2009 CHAPTERS 6, 7, 8 NAME
... You may use the Periodic Table. Please note that all quantities needed for the successful completion of this test have been given. Feel free to ask questions for clarification. 1. Indicate to which of the following types of chemical reactions each of the statements listed applies: synthesis, decompo ...
... You may use the Periodic Table. Please note that all quantities needed for the successful completion of this test have been given. Feel free to ask questions for clarification. 1. Indicate to which of the following types of chemical reactions each of the statements listed applies: synthesis, decompo ...
Practice: Chapter 21
... 15.Yes; both compounds have the molecular formula C5H8, but they have different carbon chains. 16.The aromatic compounds are 1-methyl-4-propylbenzene and anthracene because these compounds contain benzene rings as part of their molecular structures. 17. Halocarbons are named by use of the prefixes f ...
... 15.Yes; both compounds have the molecular formula C5H8, but they have different carbon chains. 16.The aromatic compounds are 1-methyl-4-propylbenzene and anthracene because these compounds contain benzene rings as part of their molecular structures. 17. Halocarbons are named by use of the prefixes f ...
organic quiz 2
... A student identifies three features of the aspartame molecule—a phenyl group attached to a carbon, an ester bond, and a carboxyl group. Match the structural feature to the numbered carbon that it is associated with. Record all three digits of your answer. ...
... A student identifies three features of the aspartame molecule—a phenyl group attached to a carbon, an ester bond, and a carboxyl group. Match the structural feature to the numbered carbon that it is associated with. Record all three digits of your answer. ...
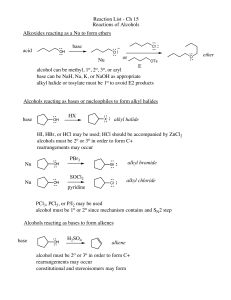
Reactions of Alcohols
... SN2 reaction between R-X and R-OWE NEED TO CONSIDER STERIC HINDERANCE. This might lead to E2! ...
... SN2 reaction between R-X and R-OWE NEED TO CONSIDER STERIC HINDERANCE. This might lead to E2! ...
Carboxylic Acids - University of Nebraska Omaha
... • However, at lower temperatures, most are quite resistant to decarboxylation. • Exceptions are carboxylic acids that have a carbonyl group beta to the carboxyl group. ...
... • However, at lower temperatures, most are quite resistant to decarboxylation. • Exceptions are carboxylic acids that have a carbonyl group beta to the carboxyl group. ...
This is the first exam with targeted syntheses that you
... Most of the reactions of aldehydes and ketones in these chapters are nucleophilic addition reactions. The oxygen in C=O polarizes the bond. Therefore, while electrophilic addition (electrophile first, followed by nucleophile) was favored for the comparatively non-polar, electron-rich alkene, carbony ...
... Most of the reactions of aldehydes and ketones in these chapters are nucleophilic addition reactions. The oxygen in C=O polarizes the bond. Therefore, while electrophilic addition (electrophile first, followed by nucleophile) was favored for the comparatively non-polar, electron-rich alkene, carbony ...
C h e m g u i d e ... ALCOHOLS: REPLACING THE -OH GROUP BY A HALOGEN
... 1. a) Describe what you would see if you added a small amount of phosphorus(V) chloride to an alcohol. b) This can only be used as a test for an alcohol if you first eliminate other compounds which also contain an -OH group. Give two completely different examples of something which would react with ...
... 1. a) Describe what you would see if you added a small amount of phosphorus(V) chloride to an alcohol. b) This can only be used as a test for an alcohol if you first eliminate other compounds which also contain an -OH group. Give two completely different examples of something which would react with ...
Section 7.5~7.6 - www .alexandria .k12 .mn .us
... 1) Why is it important to understand the acid-base behavior of organic molecules? 2) What are the differences in behavior between Lewis acids and Bronsted-Lowry acids? 2.5) In equation 3.53, is the amide ion ( NH2- ) functioning as an acid or a base? 3) How would you explain the concept of pKa to th ...
... 1) Why is it important to understand the acid-base behavior of organic molecules? 2) What are the differences in behavior between Lewis acids and Bronsted-Lowry acids? 2.5) In equation 3.53, is the amide ion ( NH2- ) functioning as an acid or a base? 3) How would you explain the concept of pKa to th ...
Synthetic Transformations of C=O Compounds Reaction Summary
... o Reacts with α,β-unsaturated aldehydes and ketones to give β-substituted carbonyl compounds. This process is called 1,4-addition or conjugate addition. O R ...
... o Reacts with α,β-unsaturated aldehydes and ketones to give β-substituted carbonyl compounds. This process is called 1,4-addition or conjugate addition. O R ...
Aldehydes and Ketones
... These carbonyl compounds generally have two reaction pathways – they react with strong nucleophiles (generally, strong nucleophiles have a formal negative charge) under neutral, generally anhydrous conditions, or with weak nucleophiles (those with lone pairs, but no charge) under mild acid catalysis ...
... These carbonyl compounds generally have two reaction pathways – they react with strong nucleophiles (generally, strong nucleophiles have a formal negative charge) under neutral, generally anhydrous conditions, or with weak nucleophiles (those with lone pairs, but no charge) under mild acid catalysis ...
Esters from Carboxylic Acid Anhydrides
... Acid Chlorides Synthesis of Acid Chlorides Acid chlorides are made from carboxylic acids by reaction with thionyl chloride, phosphorus trichloride or phosphorus ...
... Acid Chlorides Synthesis of Acid Chlorides Acid chlorides are made from carboxylic acids by reaction with thionyl chloride, phosphorus trichloride or phosphorus ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.