iNTRODUCTiON TO ORGANiC COMPOUNDS
... The last group of compounds we are going to discuss includes some that are of great importance and interest today. Included are the chlorofluorocarbons that are used in refrigeration and air conditioning systems and which are thought to be involved in the depletion of ozone in the upper atmosphere. ...
... The last group of compounds we are going to discuss includes some that are of great importance and interest today. Included are the chlorofluorocarbons that are used in refrigeration and air conditioning systems and which are thought to be involved in the depletion of ozone in the upper atmosphere. ...
chem 102 class notes - Louisiana Tech University
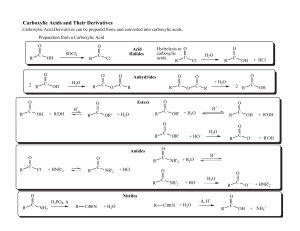
... completion, that is, until one of the reactants runs out. Many reactions do proceed . For such reactions essentially to completion: complete reactions are indicated by it can be assumed that the reactants are quantitatively converted to products and that the amount of limiting reactant that remains ...
... completion, that is, until one of the reactants runs out. Many reactions do proceed . For such reactions essentially to completion: complete reactions are indicated by it can be assumed that the reactants are quantitatively converted to products and that the amount of limiting reactant that remains ...
Preparation of ethers
... ?n in the presence of a catalyst comprising a vanadium pound at reaction conditions, and recovering the resultant ether. 50 containing compound will be effected at reaction condi tions which include an elevated temperature in the range ‘ A speci?c embodiment of this invention is found in a of from a ...
... ?n in the presence of a catalyst comprising a vanadium pound at reaction conditions, and recovering the resultant ether. 50 containing compound will be effected at reaction condi tions which include an elevated temperature in the range ‘ A speci?c embodiment of this invention is found in a of from a ...
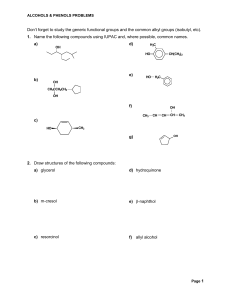
Don`t forget to study the generic functional groups and the common
... 11. Write equations to show how the following transformation can be carried out. More than one step may be necessary. There are marks assigned for each intermediate product (not charged transition states) and there are marks for the reagents used; so list them all. No marks are assigned for mechanis ...
... 11. Write equations to show how the following transformation can be carried out. More than one step may be necessary. There are marks assigned for each intermediate product (not charged transition states) and there are marks for the reagents used; so list them all. No marks are assigned for mechanis ...
File
... 6. How can you tell the difference between a “cis” and “trans” isomer? In what types of hydrocarbons can they be found? 7. What is an aromatic compound, and what is the simplest aromatic structure? 8. Do aromatic hydrocarbons react more than saturated or unsaturated hydrocarbons? Explain why. 9. Wha ...
... 6. How can you tell the difference between a “cis” and “trans” isomer? In what types of hydrocarbons can they be found? 7. What is an aromatic compound, and what is the simplest aromatic structure? 8. Do aromatic hydrocarbons react more than saturated or unsaturated hydrocarbons? Explain why. 9. Wha ...
Excited State Reactions of Carbonyl Compounds
... hand, depending on the nature of the aryl group and the substituent on the aryl ring, the pp* triplet could become the lowest state in aryl and diaryl ketones. In these systems both p and p* orbitals are delocalized into the aryl ring. Because of this, the pp* state of aryl and diaryl ketones are li ...
... hand, depending on the nature of the aryl group and the substituent on the aryl ring, the pp* triplet could become the lowest state in aryl and diaryl ketones. In these systems both p and p* orbitals are delocalized into the aryl ring. Because of this, the pp* state of aryl and diaryl ketones are li ...
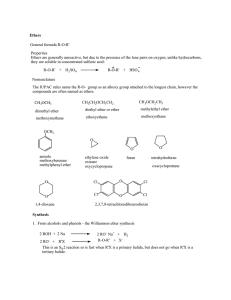
Ethers General formula R-O-R` Properties Ethers are generally
... CH3 Hg(OCOCH 3)2/CH3 CH2OH H2C C CH 3 H2C C CH 3 Hg OCH2 CH3 O C CH 3 ...
... CH3 Hg(OCOCH 3)2/CH3 CH2OH H2C C CH 3 H2C C CH 3 Hg OCH2 CH3 O C CH 3 ...
Amines
... IUPAC nomenclature retains the common name aniline for C6H5NH2, the simplest aromatic amine use numbers to locate substituents or, alternatively, use the prefixes ortho (o), meta (m), and para (p) common names are still widely used NH2 ...
... IUPAC nomenclature retains the common name aniline for C6H5NH2, the simplest aromatic amine use numbers to locate substituents or, alternatively, use the prefixes ortho (o), meta (m), and para (p) common names are still widely used NH2 ...
06_10_13.html
... The more stable the carbocation, the faster it is formed, and the faster the reaction rate. ...
... The more stable the carbocation, the faster it is formed, and the faster the reaction rate. ...
Chem 535 Synthetic Organic Chemistry – Common Reducing
... mild, will not attack ketones without H+ (cat) ...
... mild, will not attack ketones without H+ (cat) ...
KENYATTA UNIVERSITY INSTITUTE OF OPEN LEARNING SCH
... Ketones are not.easily easily oxidised. Oxidation involves breaking the carbon-carbon bonds. They are resistant to mild oxidation with Cr(VI) reagents and acetone can even be used as a solvent for oxidation with such reagents. KMnO4 oxidises ketones and is not a useful reagent for the preparation of ...
... Ketones are not.easily easily oxidised. Oxidation involves breaking the carbon-carbon bonds. They are resistant to mild oxidation with Cr(VI) reagents and acetone can even be used as a solvent for oxidation with such reagents. KMnO4 oxidises ketones and is not a useful reagent for the preparation of ...
A Crash Course In Organic Chemistry
... A functional group is a reactive portion of a molecule that undergoes predictable reactions. All other organic compounds can be considered to be derivatives of hydrocarbons ...
... A functional group is a reactive portion of a molecule that undergoes predictable reactions. All other organic compounds can be considered to be derivatives of hydrocarbons ...
Asymmetric induction

Asymmetric induction (also enantioinduction) in stereochemistry describes the preferential formation in a chemical reaction of one enantiomer or diastereoisomer over the other as a result of the influence of a chiral feature present in the substrate, reagent, catalyst or environment. Asymmetric induction is a key element in asymmetric synthesis.Asymmetric induction was introduced by Hermann Emil Fischer based on his work on carbohydrates. Several types of induction exist.Internal asymmetric induction makes use of a chiral center bound to the reactive center through a covalent bond and remains so during the reaction. The starting material is often derived from chiral pool synthesis. In relayed asymmetric induction the chiral information is introduced in a separate step and removed again in a separate chemical reaction. Special synthons are called chiral auxiliaries. In external asymmetric induction chiral information is introduced in the transition state through a catalyst of chiral ligand. This method of asymmetric synthesis is economically most desirable.