Learning Guide for Chapter 23: Amines
... VII. Reactions with acid chlorides - p 11 reduction of amides VIII. Reduction of other groups to form amines - p 12 IX. Reactivity of anilines - p 15 aromatic substutition, use of diazonium ions I. Introduction to amines Amines are considered to be organic derivatives of ammonia. They are classified ...
... VII. Reactions with acid chlorides - p 11 reduction of amides VIII. Reduction of other groups to form amines - p 12 IX. Reactivity of anilines - p 15 aromatic substutition, use of diazonium ions I. Introduction to amines Amines are considered to be organic derivatives of ammonia. They are classified ...
Alcohols, Phenols, and Thiols
... very close to that of water, so one might expect a mixture at equilibrium. It turns out that cyclopentanol, a secondary alcohol, is slightly less acidic than water and the equilibrium lies slightly to the left. The pKa of a thiol is about 10 (see Sec. 7.16), so, in part e, the equilibrium clearly wi ...
... very close to that of water, so one might expect a mixture at equilibrium. It turns out that cyclopentanol, a secondary alcohol, is slightly less acidic than water and the equilibrium lies slightly to the left. The pKa of a thiol is about 10 (see Sec. 7.16), so, in part e, the equilibrium clearly wi ...
PowerPoint 演示文稿
... conformations determined from electron diffraction studies by the Norwegian physical chemist Odd Hassel. Thus conformational analysis, which ever after changed the way organic chemists think about structure, synthesis and chemical reactivity, came into being. Barton and Hassel shared the 1969 Nobel ...
... conformations determined from electron diffraction studies by the Norwegian physical chemist Odd Hassel. Thus conformational analysis, which ever after changed the way organic chemists think about structure, synthesis and chemical reactivity, came into being. Barton and Hassel shared the 1969 Nobel ...
Chapter 6
... Classes of Alkyl Halides • Methyl halides: only one C, CH3X • Primary: C to which X is bonded has only one C-C bond. • Secondary: C to which X is bonded has two C-C bonds. • Tertiary: C to which X is bonded has three C-C bonds. ...
... Classes of Alkyl Halides • Methyl halides: only one C, CH3X • Primary: C to which X is bonded has only one C-C bond. • Secondary: C to which X is bonded has two C-C bonds. • Tertiary: C to which X is bonded has three C-C bonds. ...
Ch-6-Alcohols and phenols - Home
... Physical Properties of Phenols • Phenol is a crystalline solid, with a higher boiling point than alcohols, and it is moderately soluble in water. • It is slightly acidic: the phenol molecule has weak tendencies to lose the H+ ion from the hydroxyl group, resulting in the highly water-soluble phenol ...
... Physical Properties of Phenols • Phenol is a crystalline solid, with a higher boiling point than alcohols, and it is moderately soluble in water. • It is slightly acidic: the phenol molecule has weak tendencies to lose the H+ ion from the hydroxyl group, resulting in the highly water-soluble phenol ...
alcohols03
... Simple alcohols, like methanol and ethanol have acidity similar to water. t-butyl alcohol is less acidic because its alkoxide anion is bulky and not easily solvated by water 2,2,2-trifluoroethanol is more acidic than ethanol because the highly electronegative F-atoms inductively withdraw electron de ...
... Simple alcohols, like methanol and ethanol have acidity similar to water. t-butyl alcohol is less acidic because its alkoxide anion is bulky and not easily solvated by water 2,2,2-trifluoroethanol is more acidic than ethanol because the highly electronegative F-atoms inductively withdraw electron de ...
State the main methods used to prepare polymers?
... attached to the carbon with the most hydrogens attached to it already. In this case, the hydrogen becomes attached to the CH2 group, because the CH2 group has more hydrogens than the CH group. Notice that only the hydrogens directly attached to the carbon atoms at either end of the double bond count ...
... attached to the carbon with the most hydrogens attached to it already. In this case, the hydrogen becomes attached to the CH2 group, because the CH2 group has more hydrogens than the CH group. Notice that only the hydrogens directly attached to the carbon atoms at either end of the double bond count ...
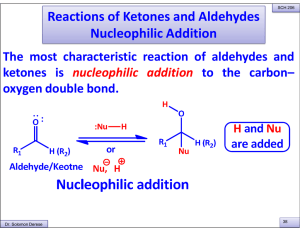
Reactions of Ketones and Aldehydes Nucleophilic Addition
... in the starting alkyne will neither tolerate the strongly basic conditions required for anion formation nor survive in a solution containing carbanions. Acetylide ions add to carbonyl groups . The strategy that is routinely followed is to protect the carbonyl group during the reactions with which it ...
... in the starting alkyne will neither tolerate the strongly basic conditions required for anion formation nor survive in a solution containing carbanions. Acetylide ions add to carbonyl groups . The strategy that is routinely followed is to protect the carbonyl group during the reactions with which it ...
幻灯片 1
... would be given the formula RO. But Williamson, by his ether synthesis, showed that mixed ethers, with two different alkyl groups, could be prepared. Ethers thus has to have the water-type formula ROR', and oxygen had the equivalent weight of 8 but the atomic weight of 16. By this type of argument he ...
... would be given the formula RO. But Williamson, by his ether synthesis, showed that mixed ethers, with two different alkyl groups, could be prepared. Ethers thus has to have the water-type formula ROR', and oxygen had the equivalent weight of 8 but the atomic weight of 16. By this type of argument he ...
Study Guide Chapter 4 Alcohols and Alkyl Halides
... A secondary radical is more stable than a primary one, and so carbon–carbon bond cleavage of 2-methylpropane requires less energy than carbon–carbon bond cleavage of propane. Carbon–carbon bond cleavage of 2,2-dimethylpropane gives a tertiary radical. CH3 ...
... A secondary radical is more stable than a primary one, and so carbon–carbon bond cleavage of 2-methylpropane requires less energy than carbon–carbon bond cleavage of propane. Carbon–carbon bond cleavage of 2,2-dimethylpropane gives a tertiary radical. CH3 ...
Organic synthesis and methodology related to the malaria drug artemisinin
... hindered ketone using either organocerium or acetylide nucleophiles. In addition, two alternative olefin metathesis approaches are described for the final cyclization. Problems associated with the olefination of a key intermediate in our efforts toward dihydroartemisinic acid led us to develop a two ...
... hindered ketone using either organocerium or acetylide nucleophiles. In addition, two alternative olefin metathesis approaches are described for the final cyclization. Problems associated with the olefination of a key intermediate in our efforts toward dihydroartemisinic acid led us to develop a two ...
dr.ebtehal Lec3
... • Drug metabolism can occur in every tissue (e.g. gut, lung and kidney). However, the major drug metabolizing enzymes (DMEs) are expressed at the highest levels in the liver, which thus serves as the major organ of metabolic clearance • Drug metabolism serves to control the exposure of a potentially ...
... • Drug metabolism can occur in every tissue (e.g. gut, lung and kidney). However, the major drug metabolizing enzymes (DMEs) are expressed at the highest levels in the liver, which thus serves as the major organ of metabolic clearance • Drug metabolism serves to control the exposure of a potentially ...
Document
... Markovnikov’s rule (1869) in the addition of HX to an unsymmetrical alkene, the H atom adds to the less substituted carbon atom—that is, the carbon that has the greater number of H atoms to ...
... Markovnikov’s rule (1869) in the addition of HX to an unsymmetrical alkene, the H atom adds to the less substituted carbon atom—that is, the carbon that has the greater number of H atoms to ...
unit 6 alcohols
... Esters: Now the LG is RO-, not usually considered “good,” but the reaction takes place by nucleophilic acyl substitution, not by SN2. In this mechanism, RO- leaving is exothermic and therefore favorable. ...
... Esters: Now the LG is RO-, not usually considered “good,” but the reaction takes place by nucleophilic acyl substitution, not by SN2. In this mechanism, RO- leaving is exothermic and therefore favorable. ...
Chapter 12, Alkenes and Alkynes
... Alkynes are hydrocarbons which have one or more carbon-carbon triple bonds. These have little import to biochemistry and will not be studied further in this course. Fig. 12.UN, p.314 ...
... Alkynes are hydrocarbons which have one or more carbon-carbon triple bonds. These have little import to biochemistry and will not be studied further in this course. Fig. 12.UN, p.314 ...
Dehydration of t-Amyl Alcohol (2-Methyl-2
... If a good nucleophile is present, such as: Cl-, Br-, I- (which would come from using strong acids HCl, HBr and HI respectively) it will form a new bond at the positive carbon. This is a nucleophilic substitution reaction. If the acid's conjugate base is a poor nucleophile such as HSO4- (which would ...
... If a good nucleophile is present, such as: Cl-, Br-, I- (which would come from using strong acids HCl, HBr and HI respectively) it will form a new bond at the positive carbon. This is a nucleophilic substitution reaction. If the acid's conjugate base is a poor nucleophile such as HSO4- (which would ...
phenol
... The ОН group of alcohols can participate in the hydrogen bond network of water. The lower alcohols are completely soluble in water. As the hydrocarbon chain gets larger, the compound begins to look more like an alkane, and more of the hydrogen bonds in water must be broken to make room for the hydro ...
... The ОН group of alcohols can participate in the hydrogen bond network of water. The lower alcohols are completely soluble in water. As the hydrocarbon chain gets larger, the compound begins to look more like an alkane, and more of the hydrogen bonds in water must be broken to make room for the hydro ...
Carbonyl Compounds I. Aldehydes and Ketones
... debye units (D). A pair of ions, C and 0 , as point charges at the C=O distance of 1.22 A, would have a dipole moment of 5.9 D. Thus, if the dipole moment of a carbonyl compound is 2.7 D , we can estimate the "% ionic character" of the bond to be (2.715.9) x 100 = 46%. The analysis is oversimplified ...
... debye units (D). A pair of ions, C and 0 , as point charges at the C=O distance of 1.22 A, would have a dipole moment of 5.9 D. Thus, if the dipole moment of a carbonyl compound is 2.7 D , we can estimate the "% ionic character" of the bond to be (2.715.9) x 100 = 46%. The analysis is oversimplified ...
Alkenes
... • When elimination can occur in more than one direction, the principal alkene is the one formed by loss of H from the carbon having the fewest hydrogens. ...
... • When elimination can occur in more than one direction, the principal alkene is the one formed by loss of H from the carbon having the fewest hydrogens. ...
PowerPoint ******
... “Wagner-Meerwein Rearrangements” must form the final products that are thermodynamically more stable than the starting materials. Some processes proceeding do appear to require uphill steps (formation of less stable carbocation). ...
... “Wagner-Meerwein Rearrangements” must form the final products that are thermodynamically more stable than the starting materials. Some processes proceeding do appear to require uphill steps (formation of less stable carbocation). ...
ALCOHOLS, PHENOLS AND ETHERS
... The formation of ethers by dehydration is a substitution type of reaction and gives only symmetrical ethers. You will study a better method of synthesis of ethers later under the section of ethers in this lesson. 6. Oxidation Alcohols can be oxidised to carbonyl compounds. Primary alcohols give alde ...
... The formation of ethers by dehydration is a substitution type of reaction and gives only symmetrical ethers. You will study a better method of synthesis of ethers later under the section of ethers in this lesson. 6. Oxidation Alcohols can be oxidised to carbonyl compounds. Primary alcohols give alde ...
Chapter 16
... Due to electronegativity of oxygen, however, this double bond is more polarized than an alkene with a greater electron density on the oxygen than carbon ...
... Due to electronegativity of oxygen, however, this double bond is more polarized than an alkene with a greater electron density on the oxygen than carbon ...
Ch. 10 Notes with Answers
... 6. View as carbanions: RMgBr = R Super Strong Bases and Nucleophiles • The counterion metal is a spectator • Stability-reactivity principle: very unstable ! very reactive • This great reactivity is very useful (as nucleophile) • This great reactivity (as base) has implication for proper technical us ...
... 6. View as carbanions: RMgBr = R Super Strong Bases and Nucleophiles • The counterion metal is a spectator • Stability-reactivity principle: very unstable ! very reactive • This great reactivity is very useful (as nucleophile) • This great reactivity (as base) has implication for proper technical us ...
Organic Chemistry II with Dr Roche
... Grignard and organolithium reagents also attack epoxides at the least hindered carbon to generate alcohols (after acidic workup). (Organometallics are typically strong bases – therefore basic conditions ring opening). ...
... Grignard and organolithium reagents also attack epoxides at the least hindered carbon to generate alcohols (after acidic workup). (Organometallics are typically strong bases – therefore basic conditions ring opening). ...
Tiffeneau–Demjanov rearrangement

The Tiffeneau–Demjanov rearrangement (TDR) is the chemical reaction of a 1-aminomethyl-cycloalkanol with nitrous acid to form an enlarged cycloketone.The Tiffeneau–Demjanov ring expansion, Tiffeneau–Demjanov rearrangement, or TDR, provides an easy way to increase amino-substituted cycloalkanes and cycloalkanols in size by one carbon. Ring sizes from cyclopropane through cyclooctane are able to undergo Tiffeneau–Demjanov ring expansion with some degree of success. Yields decrease as initial ring size increases, and the ideal use of TDR is for synthesis of five, six, and seven membered rings. A principal synthetic application of Tiffeneau–Demjanov ring expansion is to bicyclic or polycyclic systems. Several reviews on this reaction have been published.