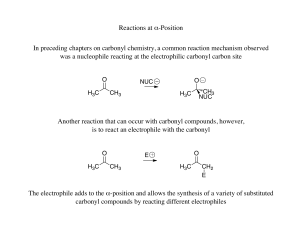
Reactions at α-Position In preceding chapters on carbonyl chemistry
... While reacting two different ketones with alkoxide base is impractical due to the variety of products obtained, the desired product would only be obtained in low yield after a difficult separation, there are methods to react two different carbonyls in an aldol reaction efficiently ...
... While reacting two different ketones with alkoxide base is impractical due to the variety of products obtained, the desired product would only be obtained in low yield after a difficult separation, there are methods to react two different carbonyls in an aldol reaction efficiently ...
Organic Chemistry
... Because tertiary and secondary carbocations are more stable than primary carbocations, Markovnikov addition is observed in the electrophilic addition of HX to alkenes, so the product formed is the one with the halogen substituent upon the more highly substituted carbon. Also, rearrangement (hydride ...
... Because tertiary and secondary carbocations are more stable than primary carbocations, Markovnikov addition is observed in the electrophilic addition of HX to alkenes, so the product formed is the one with the halogen substituent upon the more highly substituted carbon. Also, rearrangement (hydride ...
Ketones and Aldehydes
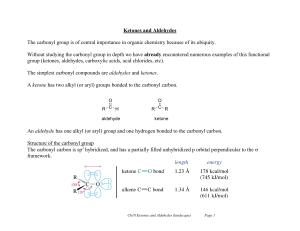
... Ketones and Aldehydes The carbonyl group is of central importance in organic chemistry because of its ubiquity. Without studying the carbonyl group in depth we have already encountered numerous examples of this functional group (ketones, aldehydes, carboxylic acids, acid chlorides, etc). The simples ...
... Ketones and Aldehydes The carbonyl group is of central importance in organic chemistry because of its ubiquity. Without studying the carbonyl group in depth we have already encountered numerous examples of this functional group (ketones, aldehydes, carboxylic acids, acid chlorides, etc). The simples ...
Nucleophilic ring opening of aziridines
... mainly derived from C4 addition trans to the aziridine ring. The reaction proceeded smoothly with alkyl Grignard reagents in high yields, but hindered t-BuMgBr did not add to the aziridine. This was also true for those having sp2 carbon nucleophiles. In addition, other organometallic reagents such a ...
... mainly derived from C4 addition trans to the aziridine ring. The reaction proceeded smoothly with alkyl Grignard reagents in high yields, but hindered t-BuMgBr did not add to the aziridine. This was also true for those having sp2 carbon nucleophiles. In addition, other organometallic reagents such a ...
Detergents The main surfactants used in detergents and personal
... function of the polyoxyethylene (hydration phenomenon). ...
... function of the polyoxyethylene (hydration phenomenon). ...
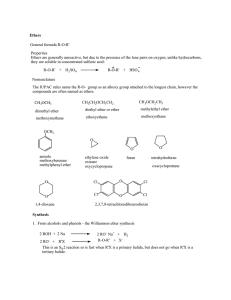
Ethers General formula R-O-R` Properties Ethers are generally
... The IUPAC rules name the R-O- group as an alkoxy group attached to the longest chain, however the compounds are often named as ethers. CH3OCH3 ...
... The IUPAC rules name the R-O- group as an alkoxy group attached to the longest chain, however the compounds are often named as ethers. CH3OCH3 ...
A Crash Course In Organic Chemistry
... system depressants, produce a wide spectrum of effects, from mild ...
... system depressants, produce a wide spectrum of effects, from mild ...
Graphene-Catalyzed Direct Friedel–Crafts Alkylation Reactions
... slightly acidic nature of the GO carbocatalyst, and no changes in the acidity after the reaction.48 (5) EDXS analysis (energydispersive X-ray spectroscopy) of the GO material before the reaction showed C/O atomic ratio of 1.95, which increased to C/O atomic ratio of 3.46 after the reaction, indicati ...
... slightly acidic nature of the GO carbocatalyst, and no changes in the acidity after the reaction.48 (5) EDXS analysis (energydispersive X-ray spectroscopy) of the GO material before the reaction showed C/O atomic ratio of 1.95, which increased to C/O atomic ratio of 3.46 after the reaction, indicati ...
Class Notes
... Super Strong Bases and Nucleophiles • The counterion metal is a spectator • Stability-reactivity principle: very unstable à very reactive • This great reactivity is very useful (as nucleophile) • This great reactivity (as base) has implication for proper technical use (see following) 7. Solvent and ...
... Super Strong Bases and Nucleophiles • The counterion metal is a spectator • Stability-reactivity principle: very unstable à very reactive • This great reactivity is very useful (as nucleophile) • This great reactivity (as base) has implication for proper technical use (see following) 7. Solvent and ...
boehm_rl
... chlorine atoms were assumed to have substituted carbon-bound hydrogen atoms on carbon atoms one and four. The work of Barham, et al. did not indicate that carbon and hydrogen had been determined; these additional data might have given an indication of the oxygen remaining in the chlorostarches. ...
... chlorine atoms were assumed to have substituted carbon-bound hydrogen atoms on carbon atoms one and four. The work of Barham, et al. did not indicate that carbon and hydrogen had been determined; these additional data might have given an indication of the oxygen remaining in the chlorostarches. ...
The Grob Fragmentation
... -Grob fragmentation: Fragmentation substrates are typically 1,3diheterofunctionalized compounds featuring a nucelophilic atom with a negative ...
... -Grob fragmentation: Fragmentation substrates are typically 1,3diheterofunctionalized compounds featuring a nucelophilic atom with a negative ...
Chapter 17 Amines
... as a precision instrument-maker, but became interested in chemistry. Self-educated in chemistry, he carried out chemical experiments in his kitchen. In 1881, he became a lecture assistant to Victor Meyer (1848-1897). He followed Meyer to Göttingen in 1886, but soon returned to Zurich and worked for ...
... as a precision instrument-maker, but became interested in chemistry. Self-educated in chemistry, he carried out chemical experiments in his kitchen. In 1881, he became a lecture assistant to Victor Meyer (1848-1897). He followed Meyer to Göttingen in 1886, but soon returned to Zurich and worked for ...
amines - Gneet`s
... aliphatic amines, whereas there is no stabilization in case of ammonia Basic strength of amines follows ( in gas phase) 3O amine > 2O amine > 1O amine > NH3 Actual order of basic strength in aqueous solution in case of lower members is found to be as 2O amine > 1O amine > 3O amine > NH3 i.e. (CH ...
... aliphatic amines, whereas there is no stabilization in case of ammonia Basic strength of amines follows ( in gas phase) 3O amine > 2O amine > 1O amine > NH3 Actual order of basic strength in aqueous solution in case of lower members is found to be as 2O amine > 1O amine > 3O amine > NH3 i.e. (CH ...
Chem E2b - Organic Chemistry II What is Organic Chemistry?
... and direct the electrophiles to the ortho and para positions • Electron withdrawing substituents deactivate benzene toward EAS, and direct electrophiles to the meta positions • Halogens are deactivating (inductive effect) and ortho/para directing (resonance) • Nucleophiles can add to arenediazonium ...
... and direct the electrophiles to the ortho and para positions • Electron withdrawing substituents deactivate benzene toward EAS, and direct electrophiles to the meta positions • Halogens are deactivating (inductive effect) and ortho/para directing (resonance) • Nucleophiles can add to arenediazonium ...
Alkyl Halides02
... This is also a qualitative test for identifying alcohols, i.e, the Lucas test. HCl with ZnCl 2 catalyst are used. 3 alcohols react quickly, 2 slowly, and 1 don’t react. Note that 1 and 2 alcohols will react with special reagents to produce alkyl halides (i.e., thionyl chloride, SOCl2, or PBr3) ...
... This is also a qualitative test for identifying alcohols, i.e, the Lucas test. HCl with ZnCl 2 catalyst are used. 3 alcohols react quickly, 2 slowly, and 1 don’t react. Note that 1 and 2 alcohols will react with special reagents to produce alkyl halides (i.e., thionyl chloride, SOCl2, or PBr3) ...
Organometallic Methods for Forming and Cleaving Carbon
... The retro benzyl addition reaction was shown by the addition of benzylmagnesium chloride to di-t-butyl ketone followed by exchange of both the benzyl and the ketone moiety with another substrate. Similar experiments were performed with phenylmagnesium bromide and t-butylmagnesium chloride, but in th ...
... The retro benzyl addition reaction was shown by the addition of benzylmagnesium chloride to di-t-butyl ketone followed by exchange of both the benzyl and the ketone moiety with another substrate. Similar experiments were performed with phenylmagnesium bromide and t-butylmagnesium chloride, but in th ...
Amino Acids and Proteins
... The 20 amino acids that occur naturally in proteins differ in the identity of the R group bonded to the α carbon. The R group is called the side chain of the amino acid. The simplest amino acid, called glycine, has R = H. All other amino acids (R ñ H) have a stereogenic center on the ` carbon. As is ...
... The 20 amino acids that occur naturally in proteins differ in the identity of the R group bonded to the α carbon. The R group is called the side chain of the amino acid. The simplest amino acid, called glycine, has R = H. All other amino acids (R ñ H) have a stereogenic center on the ` carbon. As is ...
Slide 1
... HCl (g)) causes ester formation (esterification) along with dehydration. The equilibrium constant is not large (Keq ~ 1) but high yields can be obtained by adding a large excess of one of the reactants and removing the H2O formed. The reaction is reversible. A large excess of H2O favors the reverse ...
... HCl (g)) causes ester formation (esterification) along with dehydration. The equilibrium constant is not large (Keq ~ 1) but high yields can be obtained by adding a large excess of one of the reactants and removing the H2O formed. The reaction is reversible. A large excess of H2O favors the reverse ...
synthetic.applicatio..
... For the tetrasubstituted aziridine esters 4, use of hexane as solvent with methyl ester gave predominantly the inversion product, while CH2Cl2 gave predominantly the retention product with t-butyl ester. While this behaviour was not fully understood the affinity of nitrogen for the catalyst in the p ...
... For the tetrasubstituted aziridine esters 4, use of hexane as solvent with methyl ester gave predominantly the inversion product, while CH2Cl2 gave predominantly the retention product with t-butyl ester. While this behaviour was not fully understood the affinity of nitrogen for the catalyst in the p ...
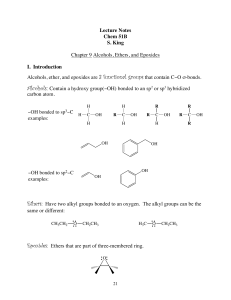
Lecture Notes Chem 51B S. King Chapter 9 Alcohols, Ethers, and
... VII. Other Methods for Converting Alcohols into Alkyl Halides We have seen that alcohols can be converted to alkyl halides by treating them with HCl, HBr, or HI. Better yields can be obtained , and carbocation rearrangements can be avoided if a phosphorus trihalide (PCl3, PBr3, or PI3), or thionyl ...
... VII. Other Methods for Converting Alcohols into Alkyl Halides We have seen that alcohols can be converted to alkyl halides by treating them with HCl, HBr, or HI. Better yields can be obtained , and carbocation rearrangements can be avoided if a phosphorus trihalide (PCl3, PBr3, or PI3), or thionyl ...
Reactions of Alkyl Halides (SN1, SN2, E1, and E2 reactions)
... elimination. In 3° substrates, only SN1 is possible. In Me° and 1° substrates, SN2 is faster. For 2° substrates, the mechanism of substitution depends upon the solvent. 2. Strong bases, like OH- and OR-, are also good nucleophiles. Substitution and elimination compete. In 3° and 2° alkyl halides, E2 ...
... elimination. In 3° substrates, only SN1 is possible. In Me° and 1° substrates, SN2 is faster. For 2° substrates, the mechanism of substitution depends upon the solvent. 2. Strong bases, like OH- and OR-, are also good nucleophiles. Substitution and elimination compete. In 3° and 2° alkyl halides, E2 ...
Reactions of Alkyl Halides (SN1, SN2, E1, and E2 reactions)
... elimination. In 3° substrates, only SN1 is possible. In Me° and 1° substrates, SN2 is faster. For 2° substrates, the mechanism of substitution depends upon the solvent. 2. Strong bases, like OH- and OR-, are also good nucleophiles. Substitution and elimination compete. In 3° and 2° alkyl halides, E2 ...
... elimination. In 3° substrates, only SN1 is possible. In Me° and 1° substrates, SN2 is faster. For 2° substrates, the mechanism of substitution depends upon the solvent. 2. Strong bases, like OH- and OR-, are also good nucleophiles. Substitution and elimination compete. In 3° and 2° alkyl halides, E2 ...
Alkyl Halides SN and E reactions
... elimination. In 3° substrates, only SN1 is possible. In Me° and 1° substrates, SN2 is faster. For 2° substrates, the mechanism of substitution depends upon the solvent. 2. Strong bases, like OH- and OR-, are also good nucleophiles. Substitution and elimination compete. In 3° and 2° alkyl halides, E2 ...
... elimination. In 3° substrates, only SN1 is possible. In Me° and 1° substrates, SN2 is faster. For 2° substrates, the mechanism of substitution depends upon the solvent. 2. Strong bases, like OH- and OR-, are also good nucleophiles. Substitution and elimination compete. In 3° and 2° alkyl halides, E2 ...
Chemical Reactivity and Biological Activity of Diketene
... pesticides, chemicals, pharmaceuticals, and dyestuffs, was investigated kinetically. The nucleophile 4-(pnitrobenzyl)pyridine (NBP), a trap for alkylating agents with nucleophilic characteristics similar to DNA bases, was used as an alkylation substrate. The alkylation reactions were performed in wa ...
... pesticides, chemicals, pharmaceuticals, and dyestuffs, was investigated kinetically. The nucleophile 4-(pnitrobenzyl)pyridine (NBP), a trap for alkylating agents with nucleophilic characteristics similar to DNA bases, was used as an alkylation substrate. The alkylation reactions were performed in wa ...
Tiffeneau–Demjanov rearrangement

The Tiffeneau–Demjanov rearrangement (TDR) is the chemical reaction of a 1-aminomethyl-cycloalkanol with nitrous acid to form an enlarged cycloketone.The Tiffeneau–Demjanov ring expansion, Tiffeneau–Demjanov rearrangement, or TDR, provides an easy way to increase amino-substituted cycloalkanes and cycloalkanols in size by one carbon. Ring sizes from cyclopropane through cyclooctane are able to undergo Tiffeneau–Demjanov ring expansion with some degree of success. Yields decrease as initial ring size increases, and the ideal use of TDR is for synthesis of five, six, and seven membered rings. A principal synthetic application of Tiffeneau–Demjanov ring expansion is to bicyclic or polycyclic systems. Several reviews on this reaction have been published.