Organic Chemistry
... Addition of HX • in the addition of the second mole of HX, Step 1 is reaction of the electron pair of the remaining pi bond with HBr to form a carbocation • of the two possible carbocations, the favored one is the resonance-stabilized 2° carbocation ...
... Addition of HX • in the addition of the second mole of HX, Step 1 is reaction of the electron pair of the remaining pi bond with HBr to form a carbocation • of the two possible carbocations, the favored one is the resonance-stabilized 2° carbocation ...
Alkenes and Alkynes I
... 6A. How to Favor an E2 Mechanism Use a secondary or tertiary alkyl halide if possible. (Because steric hinderance in the substrate will inhibit substitution) When a synthesis must begin with a primary alkyl halide, use a bulky base. (Because the steric bulk of the base will inhibit substitution) ...
... 6A. How to Favor an E2 Mechanism Use a secondary or tertiary alkyl halide if possible. (Because steric hinderance in the substrate will inhibit substitution) When a synthesis must begin with a primary alkyl halide, use a bulky base. (Because the steric bulk of the base will inhibit substitution) ...
Nucleophilic Acyl Substitution
... In Chapter 10, we saw that the likelihood of a group’s being replaced by another group depends on the relative basicities of the two groups: The weaker the basicity of a group, the better its leaving ability. Recall from Section 10.3 that weak bases are good leaving groups because weak bases do not ...
... In Chapter 10, we saw that the likelihood of a group’s being replaced by another group depends on the relative basicities of the two groups: The weaker the basicity of a group, the better its leaving ability. Recall from Section 10.3 that weak bases are good leaving groups because weak bases do not ...
Naming Organic Compounds I
... N.B.: A complex substituent is alphabetized under the first letter of its name. Branched alkyl groups On page 40, it was shown that straight-chain alkyl (n-alkyl) groups are formed by removal of a terminal (end) hydrogen atom from straight-chain alkanes. It is also possible to generate a large numbe ...
... N.B.: A complex substituent is alphabetized under the first letter of its name. Branched alkyl groups On page 40, it was shown that straight-chain alkyl (n-alkyl) groups are formed by removal of a terminal (end) hydrogen atom from straight-chain alkanes. It is also possible to generate a large numbe ...
amine
... Reductive Amination of Aldehydes and Ketones • Amines can be synthesized in a single step from aldehydes or ketones with ammonia in the presence of a reducing agent ...
... Reductive Amination of Aldehydes and Ketones • Amines can be synthesized in a single step from aldehydes or ketones with ammonia in the presence of a reducing agent ...
CH221 CLASS 13
... Addition of water to alkenes to give alcohols is one of the most important reactions of alkenes. In industry, this is accomplished by the use of strong acid catalysts and high temperatures, but this is not really of much value in the laboratory. However, tertiary alcohols can be produced from highly ...
... Addition of water to alkenes to give alcohols is one of the most important reactions of alkenes. In industry, this is accomplished by the use of strong acid catalysts and high temperatures, but this is not really of much value in the laboratory. However, tertiary alcohols can be produced from highly ...
c8h18 isomers
... ♦ The forces holding together non-polar molecules are van der Waals forces. These intermolecular forces, which operate only over very small distances, result from induced polarization of the electron clouds in molecules. ♦ Within a family: The larger the molecule the stronger the intermolecular forc ...
... ♦ The forces holding together non-polar molecules are van der Waals forces. These intermolecular forces, which operate only over very small distances, result from induced polarization of the electron clouds in molecules. ♦ Within a family: The larger the molecule the stronger the intermolecular forc ...
organic chemistry - Sakshieducation.com
... (iii) The boiling points of ethers are much lower than corresponding alcohols as they show no hydrogen bonding within themselves like alcohols. (iv) Vapours of diethyl ether cause unconsciousness on inhaling and thus used as anaesthetic agent. (v) Ethers having bond angle C-O-C to about 110o and thu ...
... (iii) The boiling points of ethers are much lower than corresponding alcohols as they show no hydrogen bonding within themselves like alcohols. (iv) Vapours of diethyl ether cause unconsciousness on inhaling and thus used as anaesthetic agent. (v) Ethers having bond angle C-O-C to about 110o and thu ...
Chapter 10:Alcohols, Phenols and Ethers
... The ethers have boiling points that are closer to those of alkanes with similar molecular weights. compares the dipole moments of dimethyl ether, diethyl ether, and tetrahydrofuran (THF) with those of alkanes and alcohols of similar molecular weights. An ether such as THF provides a strongly polar s ...
... The ethers have boiling points that are closer to those of alkanes with similar molecular weights. compares the dipole moments of dimethyl ether, diethyl ether, and tetrahydrofuran (THF) with those of alkanes and alcohols of similar molecular weights. An ether such as THF provides a strongly polar s ...
Alcohols, Phenols, and Ethers
... Lower molecular weight alcohols are soluble in water. This is due to hydrogen bonding between hydroxy group and water. ...
... Lower molecular weight alcohols are soluble in water. This is due to hydrogen bonding between hydroxy group and water. ...
Hydrocarbons Note
... Another problem arises as far as nomenclature is concerned. Hydrogens can be replaced or substituted by other elements or groups. For example a hydrogen of methane (CH4) may be replaced by a chlorine atom to form chloromethane (CH3Cl). Branched alkanes contain substitutions derived from smaller alka ...
... Another problem arises as far as nomenclature is concerned. Hydrogens can be replaced or substituted by other elements or groups. For example a hydrogen of methane (CH4) may be replaced by a chlorine atom to form chloromethane (CH3Cl). Branched alkanes contain substitutions derived from smaller alka ...
Project Overview
... Professor William Tam received his B.Sc. at the University of Hong Kong in 1990 and his Ph.D. at the University of Toronto (Canada) in 1995. He was an NSERC postdoctoral fellow at the Imperial College (UK) and at Harvard University (USA). He joined the Department of Chemistry at the University of Gu ...
... Professor William Tam received his B.Sc. at the University of Hong Kong in 1990 and his Ph.D. at the University of Toronto (Canada) in 1995. He was an NSERC postdoctoral fellow at the Imperial College (UK) and at Harvard University (USA). He joined the Department of Chemistry at the University of Gu ...
AMIDES AND AMINES: ORGANIC NITROGEN COMPOUNDS
... Unlike esters, unsubstituted amides can hydrogen bond to each other. Thus, they have a higher melting point than esters of similar molar mass. ...
... Unlike esters, unsubstituted amides can hydrogen bond to each other. Thus, they have a higher melting point than esters of similar molar mass. ...
Chapter 4 Alcohols and Alkyl Halides
... Alcohols and alkyl halides are very important functional groups. A functional group is an atom or group of atoms that undergoes certain reactions that are typical of that functional group. It is important to recognize functional groups since it makes the organization and learning of organic chemistr ...
... Alcohols and alkyl halides are very important functional groups. A functional group is an atom or group of atoms that undergoes certain reactions that are typical of that functional group. It is important to recognize functional groups since it makes the organization and learning of organic chemistr ...
Efficient and Convenient Procedure for Protection of Hydroxyl
... of the hydroxyl groups, due to the reasonable stability to the nonacidic media widely utilized in organic synthesis [1 – 3]. On the other hand, oxidation of these ethers to their aldehydes or ketones is an important transformation in organic chemistry [4, 5]. A variety of reagents have been develope ...
... of the hydroxyl groups, due to the reasonable stability to the nonacidic media widely utilized in organic synthesis [1 – 3]. On the other hand, oxidation of these ethers to their aldehydes or ketones is an important transformation in organic chemistry [4, 5]. A variety of reagents have been develope ...
Title Several Reactions of Isocyanide and Related Compounds
... The catalytic activities· of IB and lIB metal compounds, especially of (:opper compounds, for the carbonylation of primary and secondary amines to produce formamide derivatives have been investigated in Chapter 1. ...
... The catalytic activities· of IB and lIB metal compounds, especially of (:opper compounds, for the carbonylation of primary and secondary amines to produce formamide derivatives have been investigated in Chapter 1. ...
Novel Transition Metal-Catalysed Syntheses of Carboxylic Acid
... Consecutive reactions differ from tandem reactions in that another reagent, mediator or catalyst is added after the first transformation without isolation of the first formed product; the subsequent reaction steps then lead to the final product. Whilst some operational simplicity is lost, such react ...
... Consecutive reactions differ from tandem reactions in that another reagent, mediator or catalyst is added after the first transformation without isolation of the first formed product; the subsequent reaction steps then lead to the final product. Whilst some operational simplicity is lost, such react ...
IUPAC System of Nomenclature
... The rules for naming alkenes are basically the same as those of alkanes (see previous section), but with two differences. The parent chain must include the double bond even if it makes it shorter than the others. And the parent alkene chain must be numbered from whichever end gives the first carbon ...
... The rules for naming alkenes are basically the same as those of alkanes (see previous section), but with two differences. The parent chain must include the double bond even if it makes it shorter than the others. And the parent alkene chain must be numbered from whichever end gives the first carbon ...
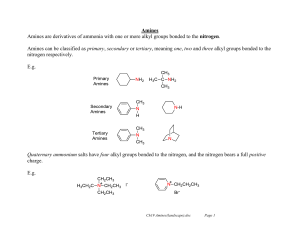
Amines Amines are derivatives of ammonia with one or more alkyl
... The reaction of amines with nitrous acid (HNO2) is a very useful synthetic reaction. Nitrous acid is unstable and needs to be generated in situ by reaction of sodium nitrite and hydrochloric acid. Na+ -O-N=O + HCl H-O-N=O + Na+ClIn very acidic media, nitrous acid can become protonated and lose wat ...
... The reaction of amines with nitrous acid (HNO2) is a very useful synthetic reaction. Nitrous acid is unstable and needs to be generated in situ by reaction of sodium nitrite and hydrochloric acid. Na+ -O-N=O + HCl H-O-N=O + Na+ClIn very acidic media, nitrous acid can become protonated and lose wat ...
NAME Chem 204 Spring, 1990 Final Exam FINAL EXAM: Chaps. 1
... one of one reaction and one of two reactions) for converting a chiral alcohol into its inverted alkyl chloride, there is NO one reaction that will convert a chiral alcohol into its alkyl chloride with "retained" configuration. Thus, it is necessary to use several reaction steps, each of 100% inversi ...
... one of one reaction and one of two reactions) for converting a chiral alcohol into its inverted alkyl chloride, there is NO one reaction that will convert a chiral alcohol into its alkyl chloride with "retained" configuration. Thus, it is necessary to use several reaction steps, each of 100% inversi ...
Chemistry 360 - Athabasca University
... What are the hazards of working with concentrated acids like glacial acetic acid and sulfuric acid? a) They are both extremely flammable b) They are only mildly corrosive and no significant precautions are needed c) Boiling these highly corrosive acids increases the danger to the experimenter, espec ...
... What are the hazards of working with concentrated acids like glacial acetic acid and sulfuric acid? a) They are both extremely flammable b) They are only mildly corrosive and no significant precautions are needed c) Boiling these highly corrosive acids increases the danger to the experimenter, espec ...
06. Alcohols. Phenols. Ethers
... are called monohydric alcohols. These are further classified as primary (1'), secondary (2'), and tertiary (3') according as the ОН group is attached to primary, secondary and tertiary carbon atoms respectively. For example: ...
... are called monohydric alcohols. These are further classified as primary (1'), secondary (2'), and tertiary (3') according as the ОН group is attached to primary, secondary and tertiary carbon atoms respectively. For example: ...
Chapter 7. Alcohols, Thiols, Phenols, Ethers
... There are many examples of the use of Grignard reactions. The example above could be expanded into many variations just by changing the halide and the carbonyl compound. Variations with other types of organometallic compound are also available. The reaction is given in more detail in chapter 8 on al ...
... There are many examples of the use of Grignard reactions. The example above could be expanded into many variations just by changing the halide and the carbonyl compound. Variations with other types of organometallic compound are also available. The reaction is given in more detail in chapter 8 on al ...
Chapter - FIU Faculty Websites
... • Like gem-diol formation, the synthesis of acetals is reversible, and often, the equilibrium favors the reactants. • In acetal synthesis, since water is formed as a by-product, the equilibrium can be driven to the right by removing H2O as it is formed using distillation or other techniques. Please ...
... • Like gem-diol formation, the synthesis of acetals is reversible, and often, the equilibrium favors the reactants. • In acetal synthesis, since water is formed as a by-product, the equilibrium can be driven to the right by removing H2O as it is formed using distillation or other techniques. Please ...
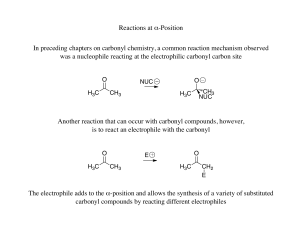
Reactions at α-Position In preceding chapters on carbonyl chemistry
... While reacting two different ketones with alkoxide base is impractical due to the variety of products obtained, the desired product would only be obtained in low yield after a difficult separation, there are methods to react two different carbonyls in an aldol reaction efficiently ...
... While reacting two different ketones with alkoxide base is impractical due to the variety of products obtained, the desired product would only be obtained in low yield after a difficult separation, there are methods to react two different carbonyls in an aldol reaction efficiently ...
Tiffeneau–Demjanov rearrangement

The Tiffeneau–Demjanov rearrangement (TDR) is the chemical reaction of a 1-aminomethyl-cycloalkanol with nitrous acid to form an enlarged cycloketone.The Tiffeneau–Demjanov ring expansion, Tiffeneau–Demjanov rearrangement, or TDR, provides an easy way to increase amino-substituted cycloalkanes and cycloalkanols in size by one carbon. Ring sizes from cyclopropane through cyclooctane are able to undergo Tiffeneau–Demjanov ring expansion with some degree of success. Yields decrease as initial ring size increases, and the ideal use of TDR is for synthesis of five, six, and seven membered rings. A principal synthetic application of Tiffeneau–Demjanov ring expansion is to bicyclic or polycyclic systems. Several reviews on this reaction have been published.