Nomenclature Chapter
... allows us to retrieve it when searching through the literature. With the explosion of information that is occurring in our time, this becomes more important with each passing day. As a foreign language, organic is relatively simple. But as a scientific language, organic is probably more complicated ...
... allows us to retrieve it when searching through the literature. With the explosion of information that is occurring in our time, this becomes more important with each passing day. As a foreign language, organic is relatively simple. But as a scientific language, organic is probably more complicated ...
Reactions of Alkenes
... transition state for attack of water on bromonium ion has carbocation character; more stable transition state (left) has positive charge on more highly substituted carbon ...
... transition state for attack of water on bromonium ion has carbocation character; more stable transition state (left) has positive charge on more highly substituted carbon ...
CHAPTER 5
... PROBLEM 5.5 Female houseflies attract males by sending a chemical signal known as a pheromone. The substance emitted by the female housefly that attracts the male has been identified as cis-9-tricosene, C23H46. Write a structural formula, including stereochemistry, for this compound. ...
... PROBLEM 5.5 Female houseflies attract males by sending a chemical signal known as a pheromone. The substance emitted by the female housefly that attracts the male has been identified as cis-9-tricosene, C23H46. Write a structural formula, including stereochemistry, for this compound. ...
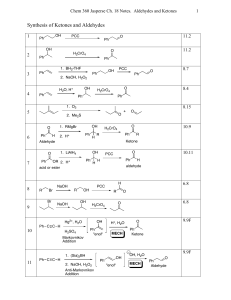
Synthesis of Ketones and Aldehydes
... • There may seem to be a dizzying number of mechanisms this chapter. But all of them simplify into some combination of acid- or base-catalyzed addition reaction, elimination reaction and/or substitution reaction. • To predict what product forms that can be isolated, you will need to know when an add ...
... • There may seem to be a dizzying number of mechanisms this chapter. But all of them simplify into some combination of acid- or base-catalyzed addition reaction, elimination reaction and/or substitution reaction. • To predict what product forms that can be isolated, you will need to know when an add ...
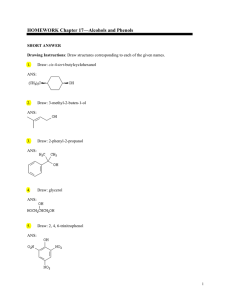
HOMEWORK Chapter 17—Alcohols and Phenols
... Acid-catalyzed dehydration of 2,2-dimethylcyclohexanol yields 1,2-dimethylcyclohexene as one of the major products. Write the complete stepwise mechanism for this reaction. Show all electron flow with arrows and show all intermediate structures. ...
... Acid-catalyzed dehydration of 2,2-dimethylcyclohexanol yields 1,2-dimethylcyclohexene as one of the major products. Write the complete stepwise mechanism for this reaction. Show all electron flow with arrows and show all intermediate structures. ...
Synthesis of Ketones and Aldehydes
... • There may seem to be a dizzying number of mechanisms this chapter. But all of them simplify into some combination of acid- or base-catalyzed addition reaction, elimination reaction and/or substitution reaction. • To predict what product forms that can be isolated, you will need to know when an add ...
... • There may seem to be a dizzying number of mechanisms this chapter. But all of them simplify into some combination of acid- or base-catalyzed addition reaction, elimination reaction and/or substitution reaction. • To predict what product forms that can be isolated, you will need to know when an add ...
+ H 2 O(l) - Knockhardy
... • Recall the definition of a covalent bond • Recall the difference types of physical bonding • Be able to balance simple equations • Be able to write out structures for simple organic molecules • Understand the IUPAC nomenclature rules for simple organic compounds • Recall the chemical properties of ...
... • Recall the definition of a covalent bond • Recall the difference types of physical bonding • Be able to balance simple equations • Be able to write out structures for simple organic molecules • Understand the IUPAC nomenclature rules for simple organic compounds • Recall the chemical properties of ...
No Slide Title
... • Recall the definition of a covalent bond • Recall the difference types of physical bonding • Be able to balance simple equations • Be able to write out structures for simple organic molecules • Understand the IUPAC nomenclature rules for simple organic compounds • Recall the chemical properties of ...
... • Recall the definition of a covalent bond • Recall the difference types of physical bonding • Be able to balance simple equations • Be able to write out structures for simple organic molecules • Understand the IUPAC nomenclature rules for simple organic compounds • Recall the chemical properties of ...
Slides from Chapter 4 - FIU Faculty Websites
... 2. Number the parent to give the hydroxyl the lowest possible number" 3. The other substituents take their locations accordingly" ...
... 2. Number the parent to give the hydroxyl the lowest possible number" 3. The other substituents take their locations accordingly" ...
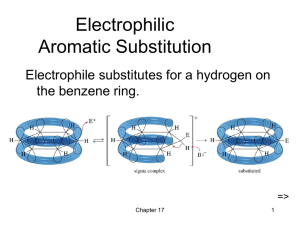
Electrophilic Aromatic Substit
... • All steps are reversible, so sulfonic acid group can be removed by heating in dilute sulfuric acid. • This process is used to place deuterium in place of hydrogen on benzene ring. D ...
... • All steps are reversible, so sulfonic acid group can be removed by heating in dilute sulfuric acid. • This process is used to place deuterium in place of hydrogen on benzene ring. D ...
ch221 class 5
... The alkanes contain carbon and hydrogen only, bonded via single bonds only. They are thus called saturated hydrocarbons because of their elemental content and because their molecules possess the maximum number of hydrogen atoms per carbon atom. The general formula is CnH2n+2. Another general name fo ...
... The alkanes contain carbon and hydrogen only, bonded via single bonds only. They are thus called saturated hydrocarbons because of their elemental content and because their molecules possess the maximum number of hydrogen atoms per carbon atom. The general formula is CnH2n+2. Another general name fo ...
OChem1 Course Pack
... 10. (8 pts) The following hydroboration-oxidation sequence results in the alkylborane and alcohol shown below. Explain how the regiochemical and stereochemical outcomes help determine the mechanism in each step. (Klein Chapter 9) ...
... 10. (8 pts) The following hydroboration-oxidation sequence results in the alkylborane and alcohol shown below. Explain how the regiochemical and stereochemical outcomes help determine the mechanism in each step. (Klein Chapter 9) ...
Alcools
... The OH group has a higher priority than a multiple CC bond, a halogen, and an alkyl group in determining the carbon chain numbering. ...
... The OH group has a higher priority than a multiple CC bond, a halogen, and an alkyl group in determining the carbon chain numbering. ...
Alcohols phenols
... alcohols as the latter are easily available. It is a nucleophilic substitution reaction in which hydroxide ion substitutes halide ions. Among alkyl halides, alkyl Iodides undergo nucleophilic substitution at the fastest rate. The mode of mechanism SN1 and SN2 depends on the nature of alkyl group. Te ...
... alcohols as the latter are easily available. It is a nucleophilic substitution reaction in which hydroxide ion substitutes halide ions. Among alkyl halides, alkyl Iodides undergo nucleophilic substitution at the fastest rate. The mode of mechanism SN1 and SN2 depends on the nature of alkyl group. Te ...
Chemistry of alcohols (powerpoint)
... • Recall the definition of a covalent bond • Recall the difference types of physical bonding • Be able to balance simple equations • Be able to write out structures for simple organic molecules • Understand the IUPAC nomenclature rules for simple organic compounds • Recall the chemical properties of ...
... • Recall the definition of a covalent bond • Recall the difference types of physical bonding • Be able to balance simple equations • Be able to write out structures for simple organic molecules • Understand the IUPAC nomenclature rules for simple organic compounds • Recall the chemical properties of ...
Carbonyl Condensation Reactions
... Regular alcohols dehydrate only in the presence of acid but not base, because hydroxide is a poor leaving group. When the hydroxy group is β to a carbonyl group, however, loss of H and OH from the α and β carbons forms a conjugated double bond, and the stability of the conjugated system makes up for ...
... Regular alcohols dehydrate only in the presence of acid but not base, because hydroxide is a poor leaving group. When the hydroxy group is β to a carbonyl group, however, loss of H and OH from the α and β carbons forms a conjugated double bond, and the stability of the conjugated system makes up for ...
Melt Modification of Poly(styrene-co-maleic anhydride)
... cyclic anhydrides involves the formation of a bicyclic species, which undergoes a rearrangement in order to give the more stable ester imide as shown in Figure 2 (pathway b).19 For the polymer analogous reaction of SMA with 1,3-oxazolines, a mechanism involving bicylic species was first considered b ...
... cyclic anhydrides involves the formation of a bicyclic species, which undergoes a rearrangement in order to give the more stable ester imide as shown in Figure 2 (pathway b).19 For the polymer analogous reaction of SMA with 1,3-oxazolines, a mechanism involving bicylic species was first considered b ...
Amidations of Rosin with Isocyanates
... Color is an important indication of modified rosin products, which somewhat reflects oxidation of rosin (Hind et aZ. 1954; Nakamura, 1970). In this study, we used color number according to JIS K5902 to monitor color changes of the reaction products of rosin with phenyl isocyanate as shown in Table 3 ...
... Color is an important indication of modified rosin products, which somewhat reflects oxidation of rosin (Hind et aZ. 1954; Nakamura, 1970). In this study, we used color number according to JIS K5902 to monitor color changes of the reaction products of rosin with phenyl isocyanate as shown in Table 3 ...
1 THE BARTON-McCOMBIE REACTION STUART W. McCOMBIE 28
... and limitations of these processes with respect to both the thioacylation and reduction steps, and exemplify some applications to both total synthesis and the modification of natural products. Comparisons are made with alternative free radical methods, with hydrogenolytic methods, and with deoxygena ...
... and limitations of these processes with respect to both the thioacylation and reduction steps, and exemplify some applications to both total synthesis and the modification of natural products. Comparisons are made with alternative free radical methods, with hydrogenolytic methods, and with deoxygena ...
C1 polymerization and related C-C bond forming - UvA-DARE
... and dilute solutions to handle their reactivity.6 Although they are very reactive, flammable and explosive,7 they make excellent precursors for carbenes and have been applied in polymerization reactions. Although the thermal decomposition of diazomethane (reported already in 1898) is the first known ...
... and dilute solutions to handle their reactivity.6 Although they are very reactive, flammable and explosive,7 they make excellent precursors for carbenes and have been applied in polymerization reactions. Although the thermal decomposition of diazomethane (reported already in 1898) is the first known ...
Forward
... 18-crown-6 is present. The reason for this has to do with the electron distribution of 18crown-6 as shown in Figure 16.2a. The electrostatic potential surface consists of essentially two regions: an electron-rich interior associated with the oxygens and a hydrocarbonlike exterior associated with the ...
... 18-crown-6 is present. The reason for this has to do with the electron distribution of 18crown-6 as shown in Figure 16.2a. The electrostatic potential surface consists of essentially two regions: an electron-rich interior associated with the oxygens and a hydrocarbonlike exterior associated with the ...
General and Selective Synthesis of (Z)-3
... oxidation reaction of Pd(0) with CuX2 to start a new catalytic cycle. In summary, PdX2 catalyzed carbonylation of terminal alkynes with CO and various alcohols including the bulky tertiary alcohol in the presence of CuX2 to form (Z)-3haloacrylates (halo ) Cl and Br) in moderate to good yields with e ...
... oxidation reaction of Pd(0) with CuX2 to start a new catalytic cycle. In summary, PdX2 catalyzed carbonylation of terminal alkynes with CO and various alcohols including the bulky tertiary alcohol in the presence of CuX2 to form (Z)-3haloacrylates (halo ) Cl and Br) in moderate to good yields with e ...
Oxygen containing organic compound
... Are they soluble? aldehydes and ketones • electronegative O atom of carbonyl group •aldehydes and ketones •hydrogen bonds with water •not between each other, thus •higher boiling points •alkanes and ethers of similar mass. •lower boiling points •alcohols of similar mass. ...
... Are they soluble? aldehydes and ketones • electronegative O atom of carbonyl group •aldehydes and ketones •hydrogen bonds with water •not between each other, thus •higher boiling points •alkanes and ethers of similar mass. •lower boiling points •alcohols of similar mass. ...
Tiffeneau–Demjanov rearrangement

The Tiffeneau–Demjanov rearrangement (TDR) is the chemical reaction of a 1-aminomethyl-cycloalkanol with nitrous acid to form an enlarged cycloketone.The Tiffeneau–Demjanov ring expansion, Tiffeneau–Demjanov rearrangement, or TDR, provides an easy way to increase amino-substituted cycloalkanes and cycloalkanols in size by one carbon. Ring sizes from cyclopropane through cyclooctane are able to undergo Tiffeneau–Demjanov ring expansion with some degree of success. Yields decrease as initial ring size increases, and the ideal use of TDR is for synthesis of five, six, and seven membered rings. A principal synthetic application of Tiffeneau–Demjanov ring expansion is to bicyclic or polycyclic systems. Several reviews on this reaction have been published.