Study Guide and Solutions Manual
... efore beginning the study of organic chemistry, a few words about “how to do it” are in order. You’ve probably heard that organic chemistry is difficult; there’s no denying that. It need not be overwhelming, though, when approached with the right frame of mind and with sustained effort. First of all ...
... efore beginning the study of organic chemistry, a few words about “how to do it” are in order. You’ve probably heard that organic chemistry is difficult; there’s no denying that. It need not be overwhelming, though, when approached with the right frame of mind and with sustained effort. First of all ...
ORGANIC CHEMISTRY 4th ed Solution Manual
... efore beginning the study of organic chemistry, a few words about “how to do it” are in order. You’ve probably heard that organic chemistry is difficult; there’s no denying that. It need not be overwhelming, though, when approached with the right frame of mind and with sustained effort. First of all ...
... efore beginning the study of organic chemistry, a few words about “how to do it” are in order. You’ve probably heard that organic chemistry is difficult; there’s no denying that. It need not be overwhelming, though, when approached with the right frame of mind and with sustained effort. First of all ...
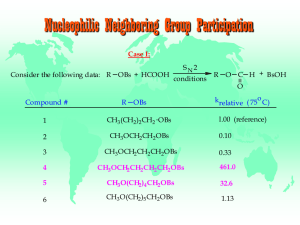
Nucleophilic Neighboring Group Participation Case I
... Nucleophilic Neighboring Group Participation Case I: Rationale Rationale to explain enhanced rates of substitution for compounds 4 & 5: ...
... Nucleophilic Neighboring Group Participation Case I: Rationale Rationale to explain enhanced rates of substitution for compounds 4 & 5: ...
CHAPTER 15 ORGANIC COMPOUNDS AND THE ATOMIC
... bond causes the loss of two hydrogen atoms. c) For hydrocarbons, “saturated” is defined as a compound that cannot add more hydrogen. An unsaturated hydrocarbon contains multiple bonds that react with H2 to form single bonds. The alkanes and cycloalkanes are saturated hydrocarbons since they contain ...
... bond causes the loss of two hydrogen atoms. c) For hydrocarbons, “saturated” is defined as a compound that cannot add more hydrogen. An unsaturated hydrocarbon contains multiple bonds that react with H2 to form single bonds. The alkanes and cycloalkanes are saturated hydrocarbons since they contain ...
Organic Chemistry - Zanichelli online per la scuola
... Introduction to the Student This study guide and solutions book was written to help you learn organic chemistry. The principles and facts of this subject are not easily learned by simply reading them, even repeatedly. Formulas, equations, and molecular structures are best mastered by written pract ...
... Introduction to the Student This study guide and solutions book was written to help you learn organic chemistry. The principles and facts of this subject are not easily learned by simply reading them, even repeatedly. Formulas, equations, and molecular structures are best mastered by written pract ...
Chapter15 odd probs
... double bond causes the loss of two hydrogen atoms. c) For hydrocarbons, “saturated” is defined as a compound that cannot add more hydrogen. An unsaturated hydrocarbon contains multiple bonds that react with H2 to form single bonds. The alkanes and cycloalkanes are saturated hydrocarbons since they c ...
... double bond causes the loss of two hydrogen atoms. c) For hydrocarbons, “saturated” is defined as a compound that cannot add more hydrogen. An unsaturated hydrocarbon contains multiple bonds that react with H2 to form single bonds. The alkanes and cycloalkanes are saturated hydrocarbons since they c ...
organic chemistry - carey - problems solutions
... chlorine. When bonded to carbon, hydrogen and lithium bear a partial positive charge, and carbon bears a partial negative charge. Conversely, when chlorine is bonded to carbon, it bears a partial negative charge, and carbon becomes partially positive. In this group of compounds, lithium is the least ...
... chlorine. When bonded to carbon, hydrogen and lithium bear a partial positive charge, and carbon bears a partial negative charge. Conversely, when chlorine is bonded to carbon, it bears a partial negative charge, and carbon becomes partially positive. In this group of compounds, lithium is the least ...
Chapter 16 Aldehydes and Ketones
... An aldehyde cannot have the molecular formula C5H12O. C5H12 has too many H’s. Since an aldehyde has a double bond, the number of C’s and H’s resembles an alkene, not an alkane. An aldehyde with 5 C’s would have the molecular formula C5H10O. ...
... An aldehyde cannot have the molecular formula C5H12O. C5H12 has too many H’s. Since an aldehyde has a double bond, the number of C’s and H’s resembles an alkene, not an alkane. An aldehyde with 5 C’s would have the molecular formula C5H10O. ...
irm_ch17
... possible between amine molecules but not between alkane molecules. 17.28 Hydrogen bonds involving oxygen (the alcohol) are stronger than hydrogen bonds involving nitrogen (the amine). 17.29 a. CH3–CH2–NH2 is more soluble in water because it has a shorter carbon chain (less nonpolar character than th ...
... possible between amine molecules but not between alkane molecules. 17.28 Hydrogen bonds involving oxygen (the alcohol) are stronger than hydrogen bonds involving nitrogen (the amine). 17.29 a. CH3–CH2–NH2 is more soluble in water because it has a shorter carbon chain (less nonpolar character than th ...
Haloalkanes-haloarenes
... CH3–CH=CH2 to CH3–CH2–CH2–OH A. CH3–CH2=CH2 + HBr org. peroxide CH3–CH2–CH2Br ii) Ethanol to butyne–1 CH3–CH2OH A. CH3CH2OH HBr ...
... CH3–CH=CH2 to CH3–CH2–CH2–OH A. CH3–CH2=CH2 + HBr org. peroxide CH3–CH2–CH2Br ii) Ethanol to butyne–1 CH3–CH2OH A. CH3CH2OH HBr ...
Course Notes
... electromagnetic radiation: energy that is transmitted through space in the form of waves wavelength: (λ): the distance from the crest of one wave to the crest of the next wave frequency: (ν): the number of complete cycles per second ...
... electromagnetic radiation: energy that is transmitted through space in the form of waves wavelength: (λ): the distance from the crest of one wave to the crest of the next wave frequency: (ν): the number of complete cycles per second ...
Organic – Nomenclature – III
... In the first case, the C=C is excluded from the longest chain and the parent is octanoic acid and the branch is methylene which is attached to C-5. Since methylene is a longer term than methyl, it is placed after methyl. In the second example, the longest chain is C-6 and hence the parent is hexenoi ...
... In the first case, the C=C is excluded from the longest chain and the parent is octanoic acid and the branch is methylene which is attached to C-5. Since methylene is a longer term than methyl, it is placed after methyl. In the second example, the longest chain is C-6 and hence the parent is hexenoi ...
Problems for Chapter 2
... at about 3600 cm–1. What is the reduced mass (µ) for O–H? What happens to the reduced mass when you double the mass of each atom in turn, i.e. what is µ for O–D and what is µ for S–H? In fact, both O–D and S–H stretches come at about 2,500 cm –1. Why? ...
... at about 3600 cm–1. What is the reduced mass (µ) for O–H? What happens to the reduced mass when you double the mass of each atom in turn, i.e. what is µ for O–D and what is µ for S–H? In fact, both O–D and S–H stretches come at about 2,500 cm –1. Why? ...
Don`t forget to study the generic functional groups and the common
... 1 Hg(CH3COO-)2, THF, H2O or 2 NaBH4 Any of the standard reagents used for hydration of alkenes ...
... 1 Hg(CH3COO-)2, THF, H2O or 2 NaBH4 Any of the standard reagents used for hydration of alkenes ...
Sample
... MARKOVNIKOV'S RULE : The addition of a proton acid to the double bond of an alkene result in a product with the acid proton bonded to the carbon atom that already holds the greater number of hydrogen atoms. This is the original statement of Markovnikov's rule. Reactions that follow this rule are sai ...
... MARKOVNIKOV'S RULE : The addition of a proton acid to the double bond of an alkene result in a product with the acid proton bonded to the carbon atom that already holds the greater number of hydrogen atoms. This is the original statement of Markovnikov's rule. Reactions that follow this rule are sai ...
Alkenes 4 - ChemWeb (UCC)
... Long before anything was known about the mechanism of this reaction it was recognised that 'Addition of HX to an alkene will proceed in such a way as to attach hydrogen to the least substituted carbon and X to the most substituted carbon'. This is known as Markovnikov's Rule after the Russian chemis ...
... Long before anything was known about the mechanism of this reaction it was recognised that 'Addition of HX to an alkene will proceed in such a way as to attach hydrogen to the least substituted carbon and X to the most substituted carbon'. This is known as Markovnikov's Rule after the Russian chemis ...
OC 2/e Ch 11
... 11 Ethers - Protecting Grps • the new C-C bond can be formed by alkylation of the acetylide anion • the OH group, however, is more acidic (pKa 16-18) than the terminal alkyne (pKa 25) • treating the compound with one mole of NaNH2 will give the alkoxide anion rather than the acetylide HC CCH2 CH2 C ...
... 11 Ethers - Protecting Grps • the new C-C bond can be formed by alkylation of the acetylide anion • the OH group, however, is more acidic (pKa 16-18) than the terminal alkyne (pKa 25) • treating the compound with one mole of NaNH2 will give the alkoxide anion rather than the acetylide HC CCH2 CH2 C ...
Chiral Enolate Equivalents
... confines the range of usable electrophiles to aldehydes, some primary or activated alkyl halides, unsaturated carbonyls, electrophilic halogens, oxaziridines, aza compounds, and a handful of other reactive electrophiles.3 Intramolecular reactions may tolerate slightly less reactive electrophiles. Wi ...
... confines the range of usable electrophiles to aldehydes, some primary or activated alkyl halides, unsaturated carbonyls, electrophilic halogens, oxaziridines, aza compounds, and a handful of other reactive electrophiles.3 Intramolecular reactions may tolerate slightly less reactive electrophiles. Wi ...
an introduction to organic reactions
... talk to your TA, visit Professor Ziegler and don't stop until you get it! attitude that a certain concept is needed for 1 exam. ...
... talk to your TA, visit Professor Ziegler and don't stop until you get it! attitude that a certain concept is needed for 1 exam. ...
barker_rg
... The tendency of an unsymmetrical anhydride to disproportionate into the twocomponent simple anhydrides often makes isolation impossible or at least difficult. This behavior greatly complicates the purification and determination of physical constants and frequently requires immediate use after prepar ...
... The tendency of an unsymmetrical anhydride to disproportionate into the twocomponent simple anhydrides often makes isolation impossible or at least difficult. This behavior greatly complicates the purification and determination of physical constants and frequently requires immediate use after prepar ...
C07C - Cooperative Patent Classification
... – a compound is considered to be saturated if it does not contain carbon atoms bound to each other by multiple bonds; – a compound is considered to be unsaturated if it contains carbon atoms bound to each other by multiple bonds, which includes six-membered aromatic ring, unless otherwise specified ...
... – a compound is considered to be saturated if it does not contain carbon atoms bound to each other by multiple bonds; – a compound is considered to be unsaturated if it contains carbon atoms bound to each other by multiple bonds, which includes six-membered aromatic ring, unless otherwise specified ...
Organic Chemistry - hrsbstaff.ednet.ns.ca
... Sources of hydrocarbons include wood, the products that result from the fermentation of plants, and fossil fuels. However, one fossil fuel— petroleum—is the main source of the hydrocarbons that are used for fuels and many other products, such as plastics and synthetic fabrics. Petroleum, sometimes r ...
... Sources of hydrocarbons include wood, the products that result from the fermentation of plants, and fossil fuels. However, one fossil fuel— petroleum—is the main source of the hydrocarbons that are used for fuels and many other products, such as plastics and synthetic fabrics. Petroleum, sometimes r ...
Alcohols, Ethers, and Epoxides
... In Chapter 9, we take the principles learned in Chapters 7 and 8 about leaving groups, nucleophiles, and bases, and apply them to alcohols, ethers, and epoxides, three new functional groups that contain polar C – O bonds. In the process, you will discover that all of the reactions in Chapter 9 follo ...
... In Chapter 9, we take the principles learned in Chapters 7 and 8 about leaving groups, nucleophiles, and bases, and apply them to alcohols, ethers, and epoxides, three new functional groups that contain polar C – O bonds. In the process, you will discover that all of the reactions in Chapter 9 follo ...
presentation source
... Each carbon forms a maximum of four single bonds, OR two single and one double bond, OR one single and triple bond. The arrangement of carbon atoms determines the skeleton, so a straight chain and a bent chain represent the same skeleton. ...
... Each carbon forms a maximum of four single bonds, OR two single and one double bond, OR one single and triple bond. The arrangement of carbon atoms determines the skeleton, so a straight chain and a bent chain represent the same skeleton. ...
Tiffeneau–Demjanov rearrangement

The Tiffeneau–Demjanov rearrangement (TDR) is the chemical reaction of a 1-aminomethyl-cycloalkanol with nitrous acid to form an enlarged cycloketone.The Tiffeneau–Demjanov ring expansion, Tiffeneau–Demjanov rearrangement, or TDR, provides an easy way to increase amino-substituted cycloalkanes and cycloalkanols in size by one carbon. Ring sizes from cyclopropane through cyclooctane are able to undergo Tiffeneau–Demjanov ring expansion with some degree of success. Yields decrease as initial ring size increases, and the ideal use of TDR is for synthesis of five, six, and seven membered rings. A principal synthetic application of Tiffeneau–Demjanov ring expansion is to bicyclic or polycyclic systems. Several reviews on this reaction have been published.