Recap: structure of ATP
... Products of Aerobic respiration Product per molecule of glucose Reduced NAD Reduced FAD CO2 ATP ...
... Products of Aerobic respiration Product per molecule of glucose Reduced NAD Reduced FAD CO2 ATP ...
Cell Respiration
... FAD+ (flavin adenine dinucleotide) • Both collect electrons from the intermediate reactions and shuttle electron (-) and proton (+) ions across membranes. • Oxygen eventually collects all the hydrogen atoms. • FAD is a better carrier, but more energy expensive to make ...
... FAD+ (flavin adenine dinucleotide) • Both collect electrons from the intermediate reactions and shuttle electron (-) and proton (+) ions across membranes. • Oxygen eventually collects all the hydrogen atoms. • FAD is a better carrier, but more energy expensive to make ...
21. Which of the electron carriers in the electron transport
... 21. Which of the electron carriers in the electron transport chain of mitochondria would you find moving electrons through rapid lateral diffusion within the lipid bilayer of the membrane from complex I to complex III? a) a flavoprotein b) cytochrome c c) an iron sulfur protein d) *ubiquinone e) cyt ...
... 21. Which of the electron carriers in the electron transport chain of mitochondria would you find moving electrons through rapid lateral diffusion within the lipid bilayer of the membrane from complex I to complex III? a) a flavoprotein b) cytochrome c c) an iron sulfur protein d) *ubiquinone e) cyt ...
Study Guide and Potential Essay Questions for Chapter 25
... hypothermia, Krebs’ cycle (TCA or citric acid cycle), lactic acid (lactate), metabolic rate, metabolic water, metabolism, minerals, mitochondrial matrix and inner membrane, NAD+/NADH + H+, nutrient, oxidation, oxidative phosphorylation, pyruvate-to-acetate step, reduction, substrate level phosphoryl ...
... hypothermia, Krebs’ cycle (TCA or citric acid cycle), lactic acid (lactate), metabolic rate, metabolic water, metabolism, minerals, mitochondrial matrix and inner membrane, NAD+/NADH + H+, nutrient, oxidation, oxidative phosphorylation, pyruvate-to-acetate step, reduction, substrate level phosphoryl ...
Bacterial Physiology Lec-8 Catabolism: Tricarboxylic acid cycle
... Mitochondrial electron transport chain is composed of a series of electron carriers that operate together to transfer electrons from donors , like NADH and FADH2 to acceptors such as O2. The electrons flow from carriers with more negative reduction potentials to those with more positive potentials a ...
... Mitochondrial electron transport chain is composed of a series of electron carriers that operate together to transfer electrons from donors , like NADH and FADH2 to acceptors such as O2. The electrons flow from carriers with more negative reduction potentials to those with more positive potentials a ...
AP Biology Study Guide
... Krebs cycle/ Citric Acid Cycle oxidative phosphorylation substrate level phosphorylation pyruvate ...
... Krebs cycle/ Citric Acid Cycle oxidative phosphorylation substrate level phosphorylation pyruvate ...
Cellular Metabolism
... – Utilize the protons and electrons that the coenzymes (NAD+ and FAD) “picked up” during glycolysis (NAD+ only) and Kreb's cycle (both NAD+ and FAD). – The electrons “power” the movement of H+ (protons) across the inner membrane space creating a proton motive gradient – This gradient is utilized alo ...
... – Utilize the protons and electrons that the coenzymes (NAD+ and FAD) “picked up” during glycolysis (NAD+ only) and Kreb's cycle (both NAD+ and FAD). – The electrons “power” the movement of H+ (protons) across the inner membrane space creating a proton motive gradient – This gradient is utilized alo ...
Microbial Metabolism
... electrons are higher (more negative) on the tower • To determine which direction the reactions go, see which is “higher” on the electron tower • Note the position of important electron carriers (NAD, FAD, cytochrome a) and external electron donors/acceptors (H2, organic compounds, O2) ...
... electrons are higher (more negative) on the tower • To determine which direction the reactions go, see which is “higher” on the electron tower • Note the position of important electron carriers (NAD, FAD, cytochrome a) and external electron donors/acceptors (H2, organic compounds, O2) ...
Aerobic Respiration: steps Coenzyme A
... •Done INSTEAD of fermentation •Pyruvic acid/pyruvate is the starting material •NADH is oxidized to NAD+ (in the end) •Loads of ATP is produced •Oxygen is the terminal electron acceptor, reduced to water ...
... •Done INSTEAD of fermentation •Pyruvic acid/pyruvate is the starting material •NADH is oxidized to NAD+ (in the end) •Loads of ATP is produced •Oxygen is the terminal electron acceptor, reduced to water ...
Chapter 9: Cellular Respiration and Fermentation (Lectures 12 + 13)
... 1.) What are the 3 “stages” of cellular respiration? 2.) Is glycolysis an aerobic or anaerobic pathway? If you oxidize one molecule of glucose, what is the approximate net yield of ATP? 3.) The reactions of glycolysis can all be categorized into one type of chemical reaction, what are these reaction ...
... 1.) What are the 3 “stages” of cellular respiration? 2.) Is glycolysis an aerobic or anaerobic pathway? If you oxidize one molecule of glucose, what is the approximate net yield of ATP? 3.) The reactions of glycolysis can all be categorized into one type of chemical reaction, what are these reaction ...
ETC Inhibitors
... so as to prevent the binding of oxygen. Electron transport is reduced to zero. Breathe all you want - you can't use any of the oxygen you take in. Rotenone, on the other hand, binds competitively, so that a trickle of electron flow is permitted. However, the rate of electron transport is too slow ...
... so as to prevent the binding of oxygen. Electron transport is reduced to zero. Breathe all you want - you can't use any of the oxygen you take in. Rotenone, on the other hand, binds competitively, so that a trickle of electron flow is permitted. However, the rate of electron transport is too slow ...
1 - contentextra
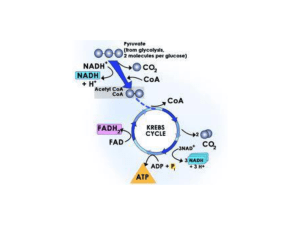
... The Krebs cycle begins and ends with oxaloacetate. Oxidation of the 6-carbon compound produced by the combination of acetyl CoA and oxaloacetate then occurs in the Krebs cycle to produce one ATP, three NADH molecules, one FADH2 molecule and two carbon dioxide molecules. For one molecule of glucose, ...
... The Krebs cycle begins and ends with oxaloacetate. Oxidation of the 6-carbon compound produced by the combination of acetyl CoA and oxaloacetate then occurs in the Krebs cycle to produce one ATP, three NADH molecules, one FADH2 molecule and two carbon dioxide molecules. For one molecule of glucose, ...
Cellular Metabolism
... “picked up” during glycolysis (NAD+ only) and Kreb's cycle (both NAD+ and FAD). – The electrons “power” the movement of H+ (protons) across the inner membrane space creating a proton motive gradient – This gradient is utilized along with oxygen that has entered the mitochondrial matrix to power a ro ...
... “picked up” during glycolysis (NAD+ only) and Kreb's cycle (both NAD+ and FAD). – The electrons “power” the movement of H+ (protons) across the inner membrane space creating a proton motive gradient – This gradient is utilized along with oxygen that has entered the mitochondrial matrix to power a ro ...
BIOL 100 Quiz 2 The four major classes of biological molecules
... 14. What role do hydrogen ions (H+) play in the electron transport chain? A) They add phosphates onto ADP to form ATP B) They are pumped across the mitochondrial inner membrane against their concentration gradient (to where their concentration is high); as the H+ ions flow back to where their concen ...
... 14. What role do hydrogen ions (H+) play in the electron transport chain? A) They add phosphates onto ADP to form ATP B) They are pumped across the mitochondrial inner membrane against their concentration gradient (to where their concentration is high); as the H+ ions flow back to where their concen ...
Cytochromes
... ► Electrons carried by reduced NAD and FAD are transported finally to Oxygen (for oxidative phosphorylation) through a gradual downward movement, from high energy level to low energy level. ► A series of Proteins & enzymes Complexes, cytochromes, present in inner membrane of mitochondria, act as tem ...
... ► Electrons carried by reduced NAD and FAD are transported finally to Oxygen (for oxidative phosphorylation) through a gradual downward movement, from high energy level to low energy level. ► A series of Proteins & enzymes Complexes, cytochromes, present in inner membrane of mitochondria, act as tem ...
Catabolic Pathways and Glycolysis
... Catabolic Pathways and Glycolysis • The ability to do that work depends on catabolic process that harvest the potential energy found in organic molecules. The 2 catabolic processes that occur in organisms are fermentation (breakdown without O2)and cellular respiration (breakdown with O2). ...
... Catabolic Pathways and Glycolysis • The ability to do that work depends on catabolic process that harvest the potential energy found in organic molecules. The 2 catabolic processes that occur in organisms are fermentation (breakdown without O2)and cellular respiration (breakdown with O2). ...
cellrespNed2012 46 KB
... Cristae increase available surface area for ATP synthase (Fo channels protons, F1 synthesizes and releases ATP in matrix) IM= inner membrane; OM= outer membrane; IM space= intermembrane space. Oxidative phosphorylation refers specifically to the simultaneous need for oxygen O2 to drive the maximum a ...
... Cristae increase available surface area for ATP synthase (Fo channels protons, F1 synthesizes and releases ATP in matrix) IM= inner membrane; OM= outer membrane; IM space= intermembrane space. Oxidative phosphorylation refers specifically to the simultaneous need for oxygen O2 to drive the maximum a ...
If you did a 10 minute wall sit, what would your muscles start to feel
... If you did a 10 minute wall sit, what would your muscles start to feel like? Why do they begin to feel like that? ...
... If you did a 10 minute wall sit, what would your muscles start to feel like? Why do they begin to feel like that? ...
AP Biology Ch 9 Cell Respiration J. Dolce Study Questions Identify
... Identify some specific processes the cell does with ATP. Explain why ATP is such a “high energy” molecule. How does ATP “couple reactions”? What is the name of enzymes which phosphorylate molecules? Define each of the following: a. Oxidation 0 b. Reduction What is the role of NAD+ & FAD+2 in respira ...
... Identify some specific processes the cell does with ATP. Explain why ATP is such a “high energy” molecule. How does ATP “couple reactions”? What is the name of enzymes which phosphorylate molecules? Define each of the following: a. Oxidation 0 b. Reduction What is the role of NAD+ & FAD+2 in respira ...
Glycolysis is the first stage of cellular respiration
... reducing agent NAD+ loses energy/electrons while the oxidizing agent glucose gains energy/electrons. The breakdown of a mole of glucose is a nonspontaneous reaction and the cell must invest 2870kj of energy to overcome the EA. During the first few steps of glycolysis the glucose molecule is phosphor ...
... reducing agent NAD+ loses energy/electrons while the oxidizing agent glucose gains energy/electrons. The breakdown of a mole of glucose is a nonspontaneous reaction and the cell must invest 2870kj of energy to overcome the EA. During the first few steps of glycolysis the glucose molecule is phosphor ...
BIO STUDY GUIDE - Biochemistry and Cells
... cc) description of facilitated diffusion dd) description of active transport ee) definition of endo- and exocytosis Cell Energy Overarching Concept: Energy production through chemiosmosis 3. Cellular Respiration a. aerobic respiration b. glycolysis a. cytoplasm c. produce 2 ATP & 2 NADH d. substrate ...
... cc) description of facilitated diffusion dd) description of active transport ee) definition of endo- and exocytosis Cell Energy Overarching Concept: Energy production through chemiosmosis 3. Cellular Respiration a. aerobic respiration b. glycolysis a. cytoplasm c. produce 2 ATP & 2 NADH d. substrate ...
cyt c - mustafaaltinisik.org.uk
... aka succinate dehydrogenase (from TCA cycle!) four subunits Two largest subunits contain 2 Fe-S proteins Other subunits involved in binding succinate dehydrogenase to membrane and passing e- to Ubiquinone • FAD accepts 2 e- and then passes 1 e- at a time to Fe-S protein • No protons pumped from this ...
... aka succinate dehydrogenase (from TCA cycle!) four subunits Two largest subunits contain 2 Fe-S proteins Other subunits involved in binding succinate dehydrogenase to membrane and passing e- to Ubiquinone • FAD accepts 2 e- and then passes 1 e- at a time to Fe-S protein • No protons pumped from this ...
SI Session 10-13-14 The molecule that functions as the reducing
... Why does the oxidation of organic compounds by molecular oxygen to produce CO2 and water release free energy? A) The covalent bonds in organic molecules are higher energy bonds than those in water and carbon dioxide. B) Electrons are being moved from atoms that have a lower affinity for electrons (s ...
... Why does the oxidation of organic compounds by molecular oxygen to produce CO2 and water release free energy? A) The covalent bonds in organic molecules are higher energy bonds than those in water and carbon dioxide. B) Electrons are being moved from atoms that have a lower affinity for electrons (s ...
Cellular Respiration Stations Worksheet Station 1: Overview Why is
... Step 3: Dr. Cao does not expect you to remember the rest of the cycle…just know that oxaloacetate is formed again in the cycle, this keeps the cycle going (hence….citric acid cycle…) 6. But do list what products are ultimately produced and how many (i.e., how many ATP, NADH, ...
... Step 3: Dr. Cao does not expect you to remember the rest of the cycle…just know that oxaloacetate is formed again in the cycle, this keeps the cycle going (hence….citric acid cycle…) 6. But do list what products are ultimately produced and how many (i.e., how many ATP, NADH, ...
Oxidative phosphorylation
Oxidative phosphorylation (or OXPHOS in short) is the metabolic pathway in which the mitochondria in cells use their structure, enzymes, and energy released by the oxidation of nutrients to reform ATP. Although the many forms of life on earth use a range of different nutrients, ATP is the molecule that supplies energy to metabolism. Almost all aerobic organisms carry out oxidative phosphorylation. This pathway is probably so pervasive because it is a highly efficient way of releasing energy, compared to alternative fermentation processes such as anaerobic glycolysis.During oxidative phosphorylation, electrons are transferred from electron donors to electron acceptors such as oxygen, in redox reactions. These redox reactions release energy, which is used to form ATP. In eukaryotes, these redox reactions are carried out by a series of protein complexes within the inner membrane of the cell's mitochondria, whereas, in prokaryotes, these proteins are located in the cells' intermembrane space. These linked sets of proteins are called electron transport chains. In eukaryotes, five main protein complexes are involved, whereas in prokaryotes many different enzymes are present, using a variety of electron donors and acceptors.The energy released by electrons flowing through this electron transport chain is used to transport protons across the inner mitochondrial membrane, in a process called electron transport. This generates potential energy in the form of a pH gradient and an electrical potential across this membrane. This store of energy is tapped by allowing protons to flow back across the membrane and down this gradient, through a large enzyme called ATP synthase; this process is known as chemiosmosis. This enzyme uses this energy to generate ATP from adenosine diphosphate (ADP), in a phosphorylation reaction. This reaction is driven by the proton flow, which forces the rotation of a part of the enzyme; the ATP synthase is a rotary mechanical motor.Although oxidative phosphorylation is a vital part of metabolism, it produces reactive oxygen species such as superoxide and hydrogen peroxide, which lead to propagation of free radicals, damaging cells and contributing to disease and, possibly, aging (senescence). The enzymes carrying out this metabolic pathway are also the target of many drugs and poisons that inhibit their activities.























