Recitation 6 The path of electron flow in photosynthesis from initial
... overall )E0' = +1.14 V, which corresponds to an overall )G0' = -220.1 kJ/mole (using )G = -nF)E) for the overall reaction NADH + ½ O2 + H+ 6 NAD+ + H2O We have also seen that this exergonic reaction can be coupled to the endergonic formation of 3 ATP (2 when FADH2) is the donor, such that the overal ...
... overall )E0' = +1.14 V, which corresponds to an overall )G0' = -220.1 kJ/mole (using )G = -nF)E) for the overall reaction NADH + ½ O2 + H+ 6 NAD+ + H2O We have also seen that this exergonic reaction can be coupled to the endergonic formation of 3 ATP (2 when FADH2) is the donor, such that the overal ...
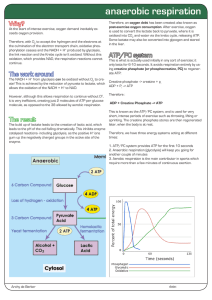
anaerobic respiration
... Therefore, with O2 to accept the hydrogen and the electrons at the culmination of the electron transport chain, oxidative phosphorylation ceases and the NADH + H+ produced by glycolysis, the link reaction and the Krebs cycle isn’t oxidized. Without this oxidation, which provides NAD, the respiration ...
... Therefore, with O2 to accept the hydrogen and the electrons at the culmination of the electron transport chain, oxidative phosphorylation ceases and the NADH + H+ produced by glycolysis, the link reaction and the Krebs cycle isn’t oxidized. Without this oxidation, which provides NAD, the respiration ...
Problem Set# 3
... a. CO2 and ATP are released during the process b. A multienzyme complex removes a carboxyl group, transfers electrons to NAD+, and attaches a coenzyme. c. NAD+ is rejuvenated so glycolysis can continue d. Lactate is produced to rejuvenate free NAD+ ___________________________________________________ ...
... a. CO2 and ATP are released during the process b. A multienzyme complex removes a carboxyl group, transfers electrons to NAD+, and attaches a coenzyme. c. NAD+ is rejuvenated so glycolysis can continue d. Lactate is produced to rejuvenate free NAD+ ___________________________________________________ ...
Midterm - ltcconline.net
... 4) A cell has the following molecules and structures: enzymes, DNA, ribosomes, plasma membrane, and mitochondria. It could be a cell from A) a bacterium. B) an animal, but not a plant. C) a plant, but not an animal. D) a plant or an animal. E) any kind of organism. 5) Which of the following statemen ...
... 4) A cell has the following molecules and structures: enzymes, DNA, ribosomes, plasma membrane, and mitochondria. It could be a cell from A) a bacterium. B) an animal, but not a plant. C) a plant, but not an animal. D) a plant or an animal. E) any kind of organism. 5) Which of the following statemen ...
Chapter 5, part A
... phosphorylate ADP to ATP • Substrate level phosphorylation - a transfer of a phosphate group from one molecule to another – 1,3-diphosphoglyceric acid + ADP ATP + 3phosphoglyceric acid ...
... phosphorylate ADP to ATP • Substrate level phosphorylation - a transfer of a phosphate group from one molecule to another – 1,3-diphosphoglyceric acid + ADP ATP + 3phosphoglyceric acid ...
AP Biology
... c. Where is substrate level phosphorylation happening? step 5 12. What is oxidative phosphorylation? the synthesis of ATP by phosphorylation of ADP for which energy is obtained by electron transport and which takes place in the mitochondria during aerobic respiration 13. What are cytochromes? It is ...
... c. Where is substrate level phosphorylation happening? step 5 12. What is oxidative phosphorylation? the synthesis of ATP by phosphorylation of ADP for which energy is obtained by electron transport and which takes place in the mitochondria during aerobic respiration 13. What are cytochromes? It is ...
Bozeman Science Video: Cellular Respiration Name: Directions
... Directions: Follow along with Mr. Anderson as he explains the process of cellular respiration. Clip can be found at http://www.bozemanscience.com/cellular-respiration 1. Cellular respiration takes organic compounds and converts them to _________, _____________, and ______________ 2. Do plants do cel ...
... Directions: Follow along with Mr. Anderson as he explains the process of cellular respiration. Clip can be found at http://www.bozemanscience.com/cellular-respiration 1. Cellular respiration takes organic compounds and converts them to _________, _____________, and ______________ 2. Do plants do cel ...
Cell Respiration and Fermentation PPT
... The ATP released in the ETC is used to move H+ ions across the membrane from low to high concentrations This stores potential energy (positive side vs negative side) ATP synthase is a protein in the membrane that spins like a mini turbine as H+ ions flow back across the membrane The spinning ATP syn ...
... The ATP released in the ETC is used to move H+ ions across the membrane from low to high concentrations This stores potential energy (positive side vs negative side) ATP synthase is a protein in the membrane that spins like a mini turbine as H+ ions flow back across the membrane The spinning ATP syn ...
Cellular Respiration
... a. It produces a net gain of ATP. b. It is an aerobic process. c. It can be performed only by bacteria. d. It produces more energy per glucose molecule than does aerobic respiration. 2. Which kind of metabolic poison would most directly interfere with glycolysis? a. an agent that reacts with oxygen ...
... a. It produces a net gain of ATP. b. It is an aerobic process. c. It can be performed only by bacteria. d. It produces more energy per glucose molecule than does aerobic respiration. 2. Which kind of metabolic poison would most directly interfere with glycolysis? a. an agent that reacts with oxygen ...
Matrix: Citric Acid Cycle and Pyruvate Oxidation Mitochondrion A
... transport of H+ to the intermembrane compartment of the mitochondrion, building a concentration gradient – Chemiosmosis: Diffusion of hydrogen ions (H+) through the differentially permeable inner mitochondrial membrane, resulting in ATP production • H+ can only cross the membrane into the mitochondr ...
... transport of H+ to the intermembrane compartment of the mitochondrion, building a concentration gradient – Chemiosmosis: Diffusion of hydrogen ions (H+) through the differentially permeable inner mitochondrial membrane, resulting in ATP production • H+ can only cross the membrane into the mitochondr ...
Second Sample Exam
... environment to make ATP and NADPH to fix CO2. a) Calculate the ΔG for the transport of protons from the outside to the inside of T. ferrooxidans (pH 6.5; membrane potential is zero) on a hot summer day in Pennsylvania. ...
... environment to make ATP and NADPH to fix CO2. a) Calculate the ΔG for the transport of protons from the outside to the inside of T. ferrooxidans (pH 6.5; membrane potential is zero) on a hot summer day in Pennsylvania. ...
Cell Respiration Flow Chart
... Step 5.T he final process of cellular respiration takes place on the inner membrane of the mitochondria. This inner membrane is much larger than the mitochondria’s outer membrane. In mitochondria that are in the liver, the inner membrane is nearly five times the area of the outer membrane. In mit ...
... Step 5.T he final process of cellular respiration takes place on the inner membrane of the mitochondria. This inner membrane is much larger than the mitochondria’s outer membrane. In mitochondria that are in the liver, the inner membrane is nearly five times the area of the outer membrane. In mit ...
electron transport
... • The number of H+ pass through the ATP synthase to synthesize an ATP, depending on the number of the c-subunit in the F0 • The number of c-subunit in ATP synthase ranges from 8-15, depending on the organism • The ratios of H+/ATP is about 2.7(8/3) to 5(15/3) • Adding one H+ for ATP-ADP translocase ...
... • The number of H+ pass through the ATP synthase to synthesize an ATP, depending on the number of the c-subunit in the F0 • The number of c-subunit in ATP synthase ranges from 8-15, depending on the organism • The ratios of H+/ATP is about 2.7(8/3) to 5(15/3) • Adding one H+ for ATP-ADP translocase ...
cellular respiration
... from another molecule 2. oxidative phosphorylation – use of ATP synthase and energy derived from a proton (H+) gradient to make ATP ...
... from another molecule 2. oxidative phosphorylation – use of ATP synthase and energy derived from a proton (H+) gradient to make ATP ...
Chapter 5: Microbial Metabolism
... 1. ___________ PHOSPHORYLATION- the transfer of a high-energy PO4- to ADP. 2. _________ - energy released from the transfer(loss) of electrons (oxidation) from one compound to another (reduction) is used to generate a proton gradient which is then used to make ATP 3. PHOTOPHOSPHORYLATION – sunlight ...
... 1. ___________ PHOSPHORYLATION- the transfer of a high-energy PO4- to ADP. 2. _________ - energy released from the transfer(loss) of electrons (oxidation) from one compound to another (reduction) is used to generate a proton gradient which is then used to make ATP 3. PHOTOPHOSPHORYLATION – sunlight ...
G:\CLASSES\BI 205\Biol205_S10\exams\Final_S10.wpd
... (6 points) (A) Name two different pathways that each contain a step where a particular molecule gets two phosphate groups attached AND (B) describe the step and/or enzyme within each of these pathways where this step occurs. Finally (C), describe why each of these steps are so critically important t ...
... (6 points) (A) Name two different pathways that each contain a step where a particular molecule gets two phosphate groups attached AND (B) describe the step and/or enzyme within each of these pathways where this step occurs. Finally (C), describe why each of these steps are so critically important t ...
PowerPoint Presentation - Ch. 6 Cellular Respiration
... energy hill. • What happens to the energy of the electrons as it falls down the electron transport chain? • The energy is used to pump H+ against their gradient which then come back through ATP synthase to generate ATP ...
... energy hill. • What happens to the energy of the electrons as it falls down the electron transport chain? • The energy is used to pump H+ against their gradient which then come back through ATP synthase to generate ATP ...
with O 2 - Pedersen Science
... • Ex. fungi, bacteria, human muscle cells • Used to make cheese, yogurt, acetone, methanol • Note: Lactate build-up does NOT causes muscle fatigue and pain (old idea) ...
... • Ex. fungi, bacteria, human muscle cells • Used to make cheese, yogurt, acetone, methanol • Note: Lactate build-up does NOT causes muscle fatigue and pain (old idea) ...
CellEnergyReview 2015
... • Ex. fungi, bacteria, human muscle cells • Used to make cheese, yogurt, acetone, methanol • Note: Lactate build-up does NOT causes muscle fatigue and pain (old idea) ...
... • Ex. fungi, bacteria, human muscle cells • Used to make cheese, yogurt, acetone, methanol • Note: Lactate build-up does NOT causes muscle fatigue and pain (old idea) ...
Midterm Final Review
... • Ex. fungi, bacteria, human muscle cells • Used to make cheese, yogurt, acetone, methanol • Note: Lactate build-up does NOT causes muscle fatigue and pain (old idea) ...
... • Ex. fungi, bacteria, human muscle cells • Used to make cheese, yogurt, acetone, methanol • Note: Lactate build-up does NOT causes muscle fatigue and pain (old idea) ...
Questions 6 Metabolism_1
... 4) Which of these is the most widely used form of energy in cell processes? a) glucose b) NADH c) pyruvate d) ATP e) citrate 5) The biochemical reaction, PEP + ADP → Pyruvate + ATP, is an example of which of these? a) An oxidation/reduction reaction. b) Oxidative phosphorylation. c) Substrate-level ...
... 4) Which of these is the most widely used form of energy in cell processes? a) glucose b) NADH c) pyruvate d) ATP e) citrate 5) The biochemical reaction, PEP + ADP → Pyruvate + ATP, is an example of which of these? a) An oxidation/reduction reaction. b) Oxidative phosphorylation. c) Substrate-level ...
File
... • Energy is stored in chemical bonds. • Chemical energy available to do work is termed free energy (G). • Adenosine triphosphate (ATP) is a kind of “energy currency” in cells. • When ATP is hydrolyzed, free energy is released to drive endergonic reactions. ...
... • Energy is stored in chemical bonds. • Chemical energy available to do work is termed free energy (G). • Adenosine triphosphate (ATP) is a kind of “energy currency” in cells. • When ATP is hydrolyzed, free energy is released to drive endergonic reactions. ...
Exam 3
... E. NADH reduces pyruvate to acetyl CoA 7. _____ Which of the following reactions of the citric acid cycle is irreversible? A. succinate + NAD+ fumarate + NADH B. malate + NAD+ oxaloacetate + NADH C. citrate isocitrate D. -ketoglutarate + NAD+ + CoA succinyl CoA + NADH + CO2 E. more than one ...
... E. NADH reduces pyruvate to acetyl CoA 7. _____ Which of the following reactions of the citric acid cycle is irreversible? A. succinate + NAD+ fumarate + NADH B. malate + NAD+ oxaloacetate + NADH C. citrate isocitrate D. -ketoglutarate + NAD+ + CoA succinyl CoA + NADH + CO2 E. more than one ...
Oxidative phosphorylation
Oxidative phosphorylation (or OXPHOS in short) is the metabolic pathway in which the mitochondria in cells use their structure, enzymes, and energy released by the oxidation of nutrients to reform ATP. Although the many forms of life on earth use a range of different nutrients, ATP is the molecule that supplies energy to metabolism. Almost all aerobic organisms carry out oxidative phosphorylation. This pathway is probably so pervasive because it is a highly efficient way of releasing energy, compared to alternative fermentation processes such as anaerobic glycolysis.During oxidative phosphorylation, electrons are transferred from electron donors to electron acceptors such as oxygen, in redox reactions. These redox reactions release energy, which is used to form ATP. In eukaryotes, these redox reactions are carried out by a series of protein complexes within the inner membrane of the cell's mitochondria, whereas, in prokaryotes, these proteins are located in the cells' intermembrane space. These linked sets of proteins are called electron transport chains. In eukaryotes, five main protein complexes are involved, whereas in prokaryotes many different enzymes are present, using a variety of electron donors and acceptors.The energy released by electrons flowing through this electron transport chain is used to transport protons across the inner mitochondrial membrane, in a process called electron transport. This generates potential energy in the form of a pH gradient and an electrical potential across this membrane. This store of energy is tapped by allowing protons to flow back across the membrane and down this gradient, through a large enzyme called ATP synthase; this process is known as chemiosmosis. This enzyme uses this energy to generate ATP from adenosine diphosphate (ADP), in a phosphorylation reaction. This reaction is driven by the proton flow, which forces the rotation of a part of the enzyme; the ATP synthase is a rotary mechanical motor.Although oxidative phosphorylation is a vital part of metabolism, it produces reactive oxygen species such as superoxide and hydrogen peroxide, which lead to propagation of free radicals, damaging cells and contributing to disease and, possibly, aging (senescence). The enzymes carrying out this metabolic pathway are also the target of many drugs and poisons that inhibit their activities.