Fall 2011 Prelim 1 BioG 1440 Introduction to Comparative
... Cellular respiration consists of three groups of reactions: glycolysis, the citric acid cycle, and oxidative phosphorylation. These three groups of reactions are not independent of each other but are coupled together by several different compounds. As a consequence of this coupling, 8) under anaerob ...
... Cellular respiration consists of three groups of reactions: glycolysis, the citric acid cycle, and oxidative phosphorylation. These three groups of reactions are not independent of each other but are coupled together by several different compounds. As a consequence of this coupling, 8) under anaerob ...
9-1 Chemical Pathways
... Fermentation When oxygen is not present, glycolysis is followed by a different pathway. The combined process of this pathway and glycolysis is called fermentation. Fermentation releases energy from food molecules by producing ATP in the absence of oxygen. There are two main types of fermentation, al ...
... Fermentation When oxygen is not present, glycolysis is followed by a different pathway. The combined process of this pathway and glycolysis is called fermentation. Fermentation releases energy from food molecules by producing ATP in the absence of oxygen. There are two main types of fermentation, al ...
Exam 2 Key
... ___Cytochrome b6f complex 7. (2 pts) In which part of the chloroplast do the 'light reactions' take place? ...
... ___Cytochrome b6f complex 7. (2 pts) In which part of the chloroplast do the 'light reactions' take place? ...
Chemistry –Worksheet: Atomic structure
... water. How many moles of HCl would be required to react with 7.5 moles of lime? How many moles of water would be formed? (15 mol HCl; 7.5 mol water) ______________ For each of the following write balanced chemical equations and then solve the problem. ...
... water. How many moles of HCl would be required to react with 7.5 moles of lime? How many moles of water would be formed? (15 mol HCl; 7.5 mol water) ______________ For each of the following write balanced chemical equations and then solve the problem. ...
Cells
... Hydrophobic fatty acid tails= water fearing and they face the inside towards each other. ...
... Hydrophobic fatty acid tails= water fearing and they face the inside towards each other. ...
Campbell`s Biology, 9e (Reece et al.) Chapter 9 Cellular Respiration
... 17) In addition to ATP, what are the end products of glycolysis? A) CO2 and H2O B) CO2 and pyruvate C) NADH and pyruvate D) CO2 and NADH E) H2O, FADH2, and citrate Answer: C Topic: Concept 9.2 Skill: Knowledge/Comprehension 18) The free energy for the oxidation of glucose to CO2 and water is -686 k ...
... 17) In addition to ATP, what are the end products of glycolysis? A) CO2 and H2O B) CO2 and pyruvate C) NADH and pyruvate D) CO2 and NADH E) H2O, FADH2, and citrate Answer: C Topic: Concept 9.2 Skill: Knowledge/Comprehension 18) The free energy for the oxidation of glucose to CO2 and water is -686 k ...
Chapter Fourteen: Metabolism: Basic Concepts and
... 38. How much ATP is used daily by a typical human? How is it regenerated? Answer: A human uses 40 kg of ATP per day. There is only about 100 g ATP available, thus the ATP is used and regenerated rapidly. ATP is regenerated from ADP and Pi, using the energy from catabolic processes. 39. What is an io ...
... 38. How much ATP is used daily by a typical human? How is it regenerated? Answer: A human uses 40 kg of ATP per day. There is only about 100 g ATP available, thus the ATP is used and regenerated rapidly. ATP is regenerated from ADP and Pi, using the energy from catabolic processes. 39. What is an io ...
How did LUCA make a living?
... Regardless of whether it is produced abiotically or by methanogens deeper within the crust, the methane in alkaline vents gives a clue to the origin of life: life began as a ‘side effect’ of the direct hydrogenation of carbon dioxide, to form methane or acetate. All autotrophs today fix carbon dioxi ...
... Regardless of whether it is produced abiotically or by methanogens deeper within the crust, the methane in alkaline vents gives a clue to the origin of life: life began as a ‘side effect’ of the direct hydrogenation of carbon dioxide, to form methane or acetate. All autotrophs today fix carbon dioxi ...
So, you want to know about siderophore synthesis
... Composed of a linear series of three hydroxamic acids. Composed of two major groups linked by succinic acid. ...
... Composed of a linear series of three hydroxamic acids. Composed of two major groups linked by succinic acid. ...
P3- Biochemical Processes
... For example, in the stomach the enzyme pepsin has a low optimum pH, so the stomach produces acid to maintain this low pH. The enzymes of the pancreas need a higher pH to work. ...
... For example, in the stomach the enzyme pepsin has a low optimum pH, so the stomach produces acid to maintain this low pH. The enzymes of the pancreas need a higher pH to work. ...
respiration revision quiz
... a. Glycolysis: Glucose (….C) is broken down to produce 2 molecules of Pyruvate (….C). b. The link reaction: Pyruvate is dehydrogenated (………………… is removed) and decarboxylated (…………………. is removed) and converted ...
... a. Glycolysis: Glucose (….C) is broken down to produce 2 molecules of Pyruvate (….C). b. The link reaction: Pyruvate is dehydrogenated (………………… is removed) and decarboxylated (…………………. is removed) and converted ...
FMM_Colin_Mitchell - Bioenergetic Failure
... flow of electrons along 5 inner membrane-bound molecular complexes (a process known as the electron transfer chain). These electrons are generated by oxidising the NADH (and FADH2) produced by the citric acid cycle to NAD+ (and FAD). The subsequent transfer of electrons from complex to complex resul ...
... flow of electrons along 5 inner membrane-bound molecular complexes (a process known as the electron transfer chain). These electrons are generated by oxidising the NADH (and FADH2) produced by the citric acid cycle to NAD+ (and FAD). The subsequent transfer of electrons from complex to complex resul ...
1. Energy & Chemical Reactions 2 Basic Forms of Energy 9/29/2015 Chapter 8:
... • light energy* (radiation of photons) • mechanical energy* (structural movement) • chemical energy* (breaking covalent bonds, flow from high to low concentration) ...
... • light energy* (radiation of photons) • mechanical energy* (structural movement) • chemical energy* (breaking covalent bonds, flow from high to low concentration) ...
Chapter 8: An Introduction to Metabolism
... energy for the endergonic synthesis of ATP, whereas ATP hydrolysis releases energy that can be used for other endergonic activities… ...
... energy for the endergonic synthesis of ATP, whereas ATP hydrolysis releases energy that can be used for other endergonic activities… ...
Biosynthesis of Nucleotides 2 - University of Alabama at Birmingham
... The advantages of multifunctional enzymes: The enzymatic activities are catalyzed by single polypeptide chains in mammals. The advantages are: 1. The product of one reaction in a pathway is the substrate for the next, and the product remains bound and are channeled directly to the next active site ...
... The advantages of multifunctional enzymes: The enzymatic activities are catalyzed by single polypeptide chains in mammals. The advantages are: 1. The product of one reaction in a pathway is the substrate for the next, and the product remains bound and are channeled directly to the next active site ...
Exam #3 2 Problem 1. (25 points) You study ligand binding to two
... of CO-hemoglobin are isomorphous with those of oxyhemoglobin, which suggests that COHb has the same conformation as oxyhemoglobin. Each heme in Hb can bind one CO molecule, but O2 and CO cannot simultaneously bind to the same heme. The binding affinity for CO is ~200 times higher than that for O2. E ...
... of CO-hemoglobin are isomorphous with those of oxyhemoglobin, which suggests that COHb has the same conformation as oxyhemoglobin. Each heme in Hb can bind one CO molecule, but O2 and CO cannot simultaneously bind to the same heme. The binding affinity for CO is ~200 times higher than that for O2. E ...
how cells obtain energy from food
... section we trace the major steps in the breakdor.tm,or catabolism, of sugarsand show how they produce ATB NADH, and other activated carrier molecules in animal cells. A very similar pathway also operates in plants, fungi, and many bacteria. As we shall see, the oxidation of fatty acids is equally im ...
... section we trace the major steps in the breakdor.tm,or catabolism, of sugarsand show how they produce ATB NADH, and other activated carrier molecules in animal cells. A very similar pathway also operates in plants, fungi, and many bacteria. As we shall see, the oxidation of fatty acids is equally im ...
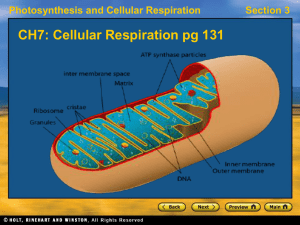
CH7Cellular-Respiration
... • Hydrogen ions diffuse through ATP synthase, providing energy to produce several ATP molecules from ADP. ...
... • Hydrogen ions diffuse through ATP synthase, providing energy to produce several ATP molecules from ADP. ...
amino acid
... -How many amino acids are shown in the polypeptide below? ____6___________ (Hint: count the R groups) -How many peptide bonds are shown in the polypeptide below? ____5________ (Hint: peptide bonds are between which two atoms?) carbon and nitrogen ...
... -How many amino acids are shown in the polypeptide below? ____6___________ (Hint: count the R groups) -How many peptide bonds are shown in the polypeptide below? ____5________ (Hint: peptide bonds are between which two atoms?) carbon and nitrogen ...
ppt
... HEY!!! Here’s the first time O2 shows up!!! It is the final electron acceptor, and water is produced as a waste product! ...
... HEY!!! Here’s the first time O2 shows up!!! It is the final electron acceptor, and water is produced as a waste product! ...
File
... Charges on ions • When atoms form ions they aim to attain electron shells that are either completely full or completely empty. • If we know the electron configuration of an atom we can usually work out how many electrons it must lose or gain to achieve a noble gas configuration. • This will tell us ...
... Charges on ions • When atoms form ions they aim to attain electron shells that are either completely full or completely empty. • If we know the electron configuration of an atom we can usually work out how many electrons it must lose or gain to achieve a noble gas configuration. • This will tell us ...
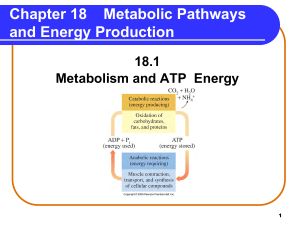
Metabolic Pathways and Energy Production
... Stages of Metabolism Catabolic reactions are organized as • Stage 1: Digestion and hydrolysis break down large molecules to smaller ones that enter the bloodstream. • Stage 2: Degradation break down molecules to two- and three-carbon compounds. • Stage 3: Oxidation of small molecules in the citric ...
... Stages of Metabolism Catabolic reactions are organized as • Stage 1: Digestion and hydrolysis break down large molecules to smaller ones that enter the bloodstream. • Stage 2: Degradation break down molecules to two- and three-carbon compounds. • Stage 3: Oxidation of small molecules in the citric ...
UNIT I - apbiologypathways
... small mass. An atom can be described as having a small, very dense nucleus with a very low density electron cloud surrounding it. Therefore, most of the mass of the universe is made up of protons and neutrons. Strong nuclear forces hold the protons and neutrons together, while the electrons are attr ...
... small mass. An atom can be described as having a small, very dense nucleus with a very low density electron cloud surrounding it. Therefore, most of the mass of the universe is made up of protons and neutrons. Strong nuclear forces hold the protons and neutrons together, while the electrons are attr ...
Biology Notes: Fermentation
... • The Point? Make molecules to restart ________________________________ • Product: _________________________ Cellular Respiration (aerobic respiration) Where? In the mitochondria Steps? Kreb’s cycle and electron transport chain What happens? 2 Pyruvate converted into 34 ATPs (plus the 2 fro ...
... • The Point? Make molecules to restart ________________________________ • Product: _________________________ Cellular Respiration (aerobic respiration) Where? In the mitochondria Steps? Kreb’s cycle and electron transport chain What happens? 2 Pyruvate converted into 34 ATPs (plus the 2 fro ...
Oxidative phosphorylation
Oxidative phosphorylation (or OXPHOS in short) is the metabolic pathway in which the mitochondria in cells use their structure, enzymes, and energy released by the oxidation of nutrients to reform ATP. Although the many forms of life on earth use a range of different nutrients, ATP is the molecule that supplies energy to metabolism. Almost all aerobic organisms carry out oxidative phosphorylation. This pathway is probably so pervasive because it is a highly efficient way of releasing energy, compared to alternative fermentation processes such as anaerobic glycolysis.During oxidative phosphorylation, electrons are transferred from electron donors to electron acceptors such as oxygen, in redox reactions. These redox reactions release energy, which is used to form ATP. In eukaryotes, these redox reactions are carried out by a series of protein complexes within the inner membrane of the cell's mitochondria, whereas, in prokaryotes, these proteins are located in the cells' intermembrane space. These linked sets of proteins are called electron transport chains. In eukaryotes, five main protein complexes are involved, whereas in prokaryotes many different enzymes are present, using a variety of electron donors and acceptors.The energy released by electrons flowing through this electron transport chain is used to transport protons across the inner mitochondrial membrane, in a process called electron transport. This generates potential energy in the form of a pH gradient and an electrical potential across this membrane. This store of energy is tapped by allowing protons to flow back across the membrane and down this gradient, through a large enzyme called ATP synthase; this process is known as chemiosmosis. This enzyme uses this energy to generate ATP from adenosine diphosphate (ADP), in a phosphorylation reaction. This reaction is driven by the proton flow, which forces the rotation of a part of the enzyme; the ATP synthase is a rotary mechanical motor.Although oxidative phosphorylation is a vital part of metabolism, it produces reactive oxygen species such as superoxide and hydrogen peroxide, which lead to propagation of free radicals, damaging cells and contributing to disease and, possibly, aging (senescence). The enzymes carrying out this metabolic pathway are also the target of many drugs and poisons that inhibit their activities.