* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Chemistry of Opioids
Metalloprotein wikipedia , lookup
Lipid signaling wikipedia , lookup
Paracrine signalling wikipedia , lookup
Biosynthesis wikipedia , lookup
Metabolomics wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Biochemistry wikipedia , lookup
Signal transduction wikipedia , lookup
Endocannabinoid system wikipedia , lookup
NMDA receptor wikipedia , lookup
Neurotransmitter wikipedia , lookup
G protein–coupled receptor wikipedia , lookup
Pharmacometabolomics wikipedia , lookup
Clinical neurochemistry wikipedia , lookup
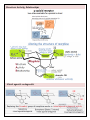
Chemistry of Opioids Opium alkaloids Alkaloid: naturally occurring compound contain basic nitrogen Opium: morphine + codeine + thebaine + papaverine + noscapine Morphine o Structure: 5 rings, 2 at right angles with each other o Chirality is key only the (–) isomer has active analgesic properties Codeine = methylated morphine = prodrug Heroin = diacetylmorphine = morphine + acetic acid o ↑Lipophilicity ↑BBB penetration ↑BA Endogenous opioid peptides Endorphin = endogenous + morphine o Different structure but similar action as morphine o Tyr as 1st amino acid residue is essential for activity Enkephalins o Opioid receptor affinity: δ > μ o Met-enkephalin: Tyr-Gly-Gly-Phe-Met o Leu-enkephalin: Tyr-Gly-Gly-Phe-Leu β-endorphin: contains Met-enkephalin sequence at amino terminal, μ opioid receptor Dynorphins: contains Leu-enkephalin sequence at amino terminal Others: endomorphins, neoendorphin, nociceptin Opioid receptors G-protein coupled receptors Receptor subtypes o μ: β-endorphin > enkephalins > dynorphins o δ: enkephalins > endorphins & dynorphins o κ: dynorphins >> endorphins & enkephalins Metabolism Morphine metabolism o Major metabolism pathway: conjugation with glucuronic acid active metabolite (M-6-G) & inactive metabolite (M-3-G) Codeine metabolism o Codeine (prodrug) → morphine o Codeine → inactive metabolite norcodeine Prodrugs: have “cod” in them o Codeine, hydrocodone, oxycodone metabolized to more potent opioids Structure Activity Relationships Mixed agonist-antagonists Pure antagonists Atypical morphine analogues Buprenorphine o Partial agonist activity @ μ receptor due to metabolite nor-buprenorphine o Partial antagonist activity@ κ receptor o Antihyperalgesic effect: κ receptor antagonistic activity o ↓Adverse effects: ↓respiratory depression, not altered by age or renal dysfunction Meperidine o Binds to P-site: phenolic ring of meperidine resembles phenylalanine of P-site o High abuse potential: penetrates BBB quickly, rapid onset o Metabolism to nor-meperidine via N-demethylation CNS excitation (e.g. tremors, seizures) o Inhibits 5-HT reuptake: potential for serotonin syndrome Absolute contraindication: meperidine + MAOIs Fentanyl o Structural features: Extra nitrogen between ring and phenyl group ↓CNS toxicities compared to meperidine Phenethyl group on piperidine nitrogen Binds both T- and P-sites 80x more potent than morphine, 940x meperidine o ↑Lipophilicity quick onset of action, quick metabolism, quick duration of action Methadone o Treats heroin withdrawal o R enantiomer: responsible for main opioid activity o MOA: μ-receptor agonist, NMDA antagonist, inhibits 5-HT & NE reuptake o Metabolism: demethylation results in many active metabolites Tramadol o Main MOA: inhibits NE & 5-HT reuptake NE reuptake inhibition: via (–) enantiomer 5-HT reuptake inhibition: via (+) enantiomer o Other MOA: weak μ agonist o Metabolism: O-demethylation by CYP2D6 more potent μ-receptor agonist (200x affinity) Tapentadol o Indication: moderate to severe pain o MOA: dual action: (+)μ-receptor & (–)NE reuptake o Formulation: IR & PO o SE: ↑tolerability but ↑risk for respiratory depression o Potency: tramadol < tapentadol < morphine