* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download document 8916804
Neonatal infection wikipedia , lookup
Hygiene hypothesis wikipedia , lookup
DNA vaccination wikipedia , lookup
Globalization and disease wikipedia , lookup
Anti-nuclear antibody wikipedia , lookup
Immunocontraception wikipedia , lookup
Immune system wikipedia , lookup
Duffy antigen system wikipedia , lookup
Molecular mimicry wikipedia , lookup
Adoptive cell transfer wikipedia , lookup
Psychoneuroimmunology wikipedia , lookup
Innate immune system wikipedia , lookup
Adaptive immune system wikipedia , lookup
Monoclonal antibody wikipedia , lookup
Polyclonal B cell response wikipedia , lookup
Cancer immunotherapy wikipedia , lookup
Doctoral thesis from the Department of Immunology,
The Wenner-Gren Institute, Stockholm University, Sweden
Single nucleotide polymorphisms related to
immune responses in Plasmodium
falciparum malaria
Amre Osman Nasr
Stockholm 2008
All previously published papers were reproduced with permission from publishers.
© Amre Osman Nasr, Stockholm, Sweden 2008
ISBN 978-91-7155-726-1
Printed in Sweden by Universitetsservice AB, Stockholm 2008
Distributer: Stockholm University Library
ﺑﺴﻢ اﷲ اﻟﺮﺣﻤﻦ اﻟﺮﺣﻴﻢ
ﻼ
ً وﻣﺎ اوﺗﻴﺘﻢ ﻣﻦ اﻟﻌﻠﻢ اﻹ ﻗﻠﻴ
“No matter how much you know, there is much to know”
To the memory of my mother
To my father
To my brother
To my Sisters
To my family
To my friends
“Do not think of today’s failures, but of the success that may come tomorrow. You have set
yourselves a difficult task, but you will succeed if you persevere; and you will find joy in
overcoming obstacles. Remember, no effort that we make to attain something beautiful is ever
lost’’
Helen Keller
SUMMARY
A comprehension of the genetics of host resistance to malaria is essential to understanding
the complexity of the host immune response and its interaction with the parasite infection. Current
research is directed towards dissection of host genetic factors involved in both the host immune
response and the malaria disease outcome.
First, a possible association between Fc gamma receptor IIa-R/H131 (FcγRIIa-R/H131)
polymorphism and anti-malarial antibody responses was investigated in Sudanese patients in
relation to clinical outcome of Plasmodium falciparum malaria. The frequency of the R/R131
genotype was significantly higher in patients with severe malaria as compared to those with mild
malaria, while the H/H131 genotype showed a significant association with mild malaria. Antimalarial IgG3 antibodies were shown to be associated with reduced risk of clinical malaria in
individuals carrying the H/H131 genotype. Low levels of IgG2 antibodies reactive with the Pf332C231 antigen were also associated with lower risk of severe malaria in individuals carrying the
H131 allele. The levels of anti-malarial IgG1 and IgG3 antibodies were significantly higher in
patients with mild malaria compared with those with severe malaria. Our data suggest the
importance of cytophilic antibodies, since IgG2 can act as such in the combination with FcγRIIaH/H131 genotype. However, the role of IgG2 antibodies in malarial immunity is still a subject of
controversy, with different levels being suggested to underlie protective properties.
Fulani and Masaleit, two sympatric ethnic groups in eastern Sudan, are characterized by
marked differences in susceptibility to P. falciparum malaria. We investigated whether the two
populations differ in the frequency of immunoglobulin GM/KM allotypes, which are known to
influence both humoral and cellular immune responses and to be associated the with susceptibility to
and outcome of several infections. The distribution of GM and KM phenotypes differed
significantly among the two ethnic groups, with Gm 6 being significantly lower among the Fulani,
and the combined frequency of Km 1,3 and Gm 1,17 5,6,13,14 phenotypes was found to be higher
among Masaleit.
Previously, it has been suggested that sickle cell trait carriage (HbAS) may protect against
the most severe forms of malaria. In the present study we investigated whether the two ethnic groups
differ in the frequency of FcγRIIa and HbAS genotypes. The frequency of the FcγRIIa- H/H131
genotype and the H131allele was found to be higher in the Fulani, whereas the R/R131 genotype
was significantly higher in the Masaleit group. On the other hand, the HbAS genotype was less
frequent in the Fulani as compared to Masaleit. Moreover, the Fulani showed higher levels of antimalarial IgG2 and lower IgG1 and IgG3 antibodies when compared to their sympatric non-Fulani
neighbours.
The complement-reactive protein (CRP) shows a differential binding to the FcγRIIa-R/H131
receptors. A tri-allelic SNP (C/T/A) in the CRP gene was investigated for possible ethnic
associations. The A allele, which is associated with higher basal CRP levels, was found to be less
frequent in the Fulani compared with non-Fulani ethnic groups both in Sudan and Mali. Our data
thus indicate that CRP could be a contributing factor to the relative resistance to malaria seen in the
Fulani as compared to other sympatric ethnic groups.
In conclusion, our results suggest possible associations between FcγRIIa-R/H131 and CRP
genotypes, GM/KM allotypes, and anti-malarial antibody responses and the clinical outcome of P.
falciparum malaria.
I
LIST OF PAPERS
This thesis is based on following original papers, which will be referred to by their roman
numerals
I. Nasr, A.*, Iriemenam, N.C.*, Troye-Blomberg, M., Giha, H.A., Balogun, H.A.,
Osman,O.F., Montgomery, S.M., ElGhazali, G., Berzins, K.; 2007. FcγRIIa (CD32)
Polymorphism and Antibody responses to Asexual Blood-stage Antigens of
Plasmodium falciparum Malaria in Sudanese Patients. Scand. J. Immunol. 66, 8796.
* The authors A. Nasr and N. C. Iriemenam contributed equally to this article.
II. Pandey, J.P., Nasr, A., Rocca, K.M., Troye-Blomberg, M., ElGhazali, G.; 2007.
Significant differences in GM allotype frequencies between two sympatric tribes
with markedly differential susceptibility to malaria. Parasite Immunol. 29, 267-269.
III. Nasr, A., ElGhazali, G., Giha, H., Troye-Blomberg, M., Berzins K; 2008. Interethnic
differences in carriage of HbAS and FcγRIIa (CD32) genotypes in children living in
eastern Sudan. Acta Trop. 105, 191-195.
IV. Nasr, A., Iriemenam, N. C., Giha, H. A., Balogun, H.A., Anders, R. F., TroyeBlomberg, M., ElGhazali, G., Berzins, K. FcγRIIa (CD32), polymorphism and antimalarial IgG subclass pattern among Fulani and their sympatric tribes in eastern
Sudan (Manuscript Submitted).
V. Israelsson, E., Ekström, M., Nasr, A., Arambepola, G., Maiga, B., Dolo, A., Doumbo,
O.K., ElGhazali, G., Giha, H.A., Troye-Blomberg, M., Berzins, K., Tornvall, P.
Polymorphism in the CRP gene may contribute to the lower susceptibility to malaria
of the Fulani ethnic group in Africa (Manuscript).
II
TABLE OF CONTENTS
SUMMARY ............................................................................................................................................................ I LIST OF PAPERS................................................................................................................................................ II TABLE OF CONTENTS .................................................................................................................................. III LIST OF ABBREVIATIONS ............................................................................................................................IV INTRODUCTION ................................................................................................................................................. 1 THE IMMUNE SYSTEM ......................................................................................................................................... 1 Innate immunity ............................................................................................................................................. 1 Cells involved in innate immunity ............................................................................................................................. 3 Adaptive Immunity ......................................................................................................................................... 4 MHC ........................................................................................................................................................................... 5 Cytokines .................................................................................................................................................................... 5 T Lymphocytes ........................................................................................................................................................... 6 Th1/ Th2 responses ..................................................................................................................................................... 7 B cells ......................................................................................................................................................................... 7 Immunoglobulins ........................................................................................................................................... 8 Ig classes ..................................................................................................................................................................... 9 Ig allotypes................................................................................................................................................................ 11 Fc receptors ................................................................................................................................................. 11 The FcγR family ....................................................................................................................................................... 11 The Structure of FcγRIIa .......................................................................................................................................... 12
Function of FcγRIIa .................................................................................................................................................. 12
Allelic variants of FcγRIIa ....................................................................................................................................... 13
MALARIA ........................................................................................................................................................... 14 CHARACTERISTICS OF MALARIA ....................................................................................................................... 14 Life cycle and morphology .......................................................................................................................... 14 Pre-patient and incubation periods ........................................................................................................................... 15 Signs and symptoms of malaria................................................................................................................... 16
Severe malaria .......................................................................................................................................................... 16
Diagnosis of malaria ................................................................................................................................... 16
Conventional microscopy ......................................................................................................................................... 16
Alternative diagnostic methods ................................................................................................................................ 17
IMMUNITY TO MALARIA ............................................................................................................................. 19 INNATE RESISTANCE.......................................................................................................................................... 19 Haemoglobin variants ................................................................................................................................. 19 Complement receptors................................................................................................................................. 22 ACQUIRED IMMUNITY ....................................................................................................................................... 22 Immune mechanisms against the pre-erythrocytic stage............................................................................ 23 Immune mechanisms against the erythrocytic stage .................................................................................. 23 RELATED BACKGROUND ............................................................................................................................. 26 THE COMPLEX GENETIC BASIS OF RESISTANCE TO MALARIA ......................................................................... 26 The role of IgG in protection against malarial infection and disease ....................................................... 27 FcγRIIa in Malaria ...................................................................................................................................... 27 Ethnic Differences ....................................................................................................................................... 28 IgG allotypes................................................................................................................................................ 28 C- reactive protein (CRP) ........................................................................................................................... 29 THE PRESENT STUDY .................................................................................................................................... 31 OBJECTIVES OF THE STUDY ............................................................................................................................... 31 MATERIAL AND METHODS USED IN THE STUDY ............................................................................................... 31 RESULTS AND DISCUSSION ............................................................................................................................... 31 FcγRIIa polymorphism in relation to malaria (Papers I, III and IV).......................................................... 32 IgG GM/KM allotypes in relation to malaria (Paper II)............................................................................. 33 IgG subclasses in relation to malaria (papers I and IV) ............................................................................. 34 IgG subclasses in relation to FcγRIIa polymorphism (Papers I and IV).................................................... 36 HbAS in relation to malaria (Paper II) ........................................................................................................ 37 CRP in malaria in relation to ethnicity (Paper V)....................................................................................... 38 CONCLUDING REMARKS AND FUTURE PERSPECTIVES .................................................................. 40 ACKNOWLEDGMENTS .................................................................................................................................. 43 REFERENCES .................................................................................................................................................... 45 III
LIST OF ABBREVIATIONS
ADCI
Antibody-dependent cell mediated inhibition
APC
Antigen presenting cell
BCR
B-cell receptor
CRP
C-reactive protein
DC
Dendritic cell
FcR
Fc receptor
G6PD
Glucose-6-phosphate dehydrogenase
H
Histidine
IFN
Interferon
Ig
Immunoglobulin
IL
Interleukin
iRBC
Infected red-blood cell
ITAM
Immunoreceptor Tyrosin-based Activation Motifs
ITIM
Immunoreceptor Tyrosin-based Inhibiting Motifs
MHC
Major histocompatibility complex
MPL
Mannan-binding lectin
MSP
Merozoite surface protein
NK cell
Natural-killer cell
PCR
Polymerase chain reaction
PRRs
Pattern recognition receptors
R
Arginine
RBC
Red-blood cell
SNP
Single nucleated polymorphism
TCR
T-cell receptor
Th
T helper
TLR
Toll-like receptor
IV
INTRODUCTION
The Immune System
Historically, immunity meant protection from disease, and more specifically, against
infectious diseases. The cells and molecules responsible for immunity constitute the
immune system, and their collective and coordinated response after the introduction of
foreign substance is the immune response. We now know that many of the mechanisms of
resistance to infections also involve the individual’s response to non-infectious foreign
substances 1. However, mechanisms that normally protect individuals from infections and
eliminate foreign substances are themselves capable of causing tissue injury and disease in
some situations. Therefore, a more inclusive and modern definition of immunity is a
reaction to foreign substances, including microbes, as well as macromolecules, such as
proteins and polysaccharides, without implying a physiologic or pathologic consequence of
such a reaction 2. Immunology is the study of immunity in this broader sense, and of the
cellular and molecular events that occur after an organism encounters microbes or other
foreign macromolecules. The immune system is branched into two arms, governing the
innate (natural or nonspecific) and the acquired (adaptive or specific) immune responses 1.
Innate immunity
Most encounters with microorganisms do not result in disease. The few microbes
that manage to cross the barriers of skin, mucus, cilia, and pH are usually eliminated by
innate immune mechanisms, which commence immediately upon pathogen entry 2. If
phagocytosis cannot rapidly eliminate the pathogen, inflammation is induced with the
production of cytokines and acute phase proteins. This early induced response is not
antigen-specific and does not generate immune memory 2. Only if the inflammatory process
is unsuccessful at eliminating the pathogen will the adaptive immune system be activated, a
process which requires several days to produce armed effector cells.
Pathogens infect at specific locations in the host and damage the host by several
mechanisms. Extracellular pathogens and their exotoxins are susceptible to destruction by
phagocytes, either directly or in cooperation with antibodies elicited by the acquired
immune system, so called opsonization. Intracellular pathogens may be eliminated by lysis
of the infected cell by natural-killer (NK) cells
3
or dendritic cells. Pathogens within
macrophages may be killed by the activation of these cells by IFN-γ derived from Th1 cells.
__________________________________________________________________________
Amre Nasr, 2008
1
The immune response can also damage the body by means of inflammation and
cytotoxicity4.
Innate immunity covers many areas of the host defence against pathogenic microbes,
including the recognition of pathogen-associated molecular patterns (PAMPs) present on
the microbes5. In vertebrates, which are the only phylum that possesses an adaptive immune
system, there are also mechanisms that inhibit the activation of the innate immunity. One
example is the inhibition of killing by NK cells, which are known to receive an inhibitory
stimulus from major histocompatibility complex (MHC) class I molecules.
The immune system uses a variety of pattern-recognition receptors (PRRs) that can
be expressed on the cell surface, in intracellular compartments, or secreted into the
bloodstream and tissue fluids2. The principal functions of PRRs include opsonisation,
activation of complement, coagulation cascades, phagocytosis, activation of proinflammatory signalling pathways, and induction of apoptosis.
In brief, the classical complement pathway of activation is initiated by complement
protein 1 (C1) binding to antigen-antibody complexes, while the alternative pathway is
initiated by C3b binding to various activating surfaces, such as microbial cell walls. The
C3b involved in the alternative pathway initiation may be generated in several ways,
including spontaneously, by the classical pathway, or by the alternative pathway itself. Both
pathways converge and lead to the formation of the membrane attack complex.
C-reactive protein (CRP, for further details see section under Related Background),
mannan-binding lectin (MBL), and serum amyloid protein (SAP) are secreted pattern
recognition molecules, produced by the liver during the acute phase response at early stages
of infection 6-8.
Toll-receptors are important components of the innate immunity. They are
evolutionarily conserved and homologous receptors found in plants, insects, worms and
vertebrates. The first member of this family, named Toll, was initially identified in the fruit
fly Drosophila melanogaster 9. This receptor has been shown to play an important role in
protection against fungi in the fruit fly
10
. In humans, 11 Toll-like receptors (TLRs) have
been identified until now. Based on the cellular localization, TLRs are classified into two
groups. TLR 3, 7, 8 and 9 are expressed in endosomes, while the second group, including
TLR1, 2, 4, 5 and 6, are present on the surface of many different cells
11, 12
. Upon
recognition of their cognate ligands, TLRs dimerize and initiate a signalling cascade that
leads to activation of a proinflammatory response
13, 14
. Ligand binding induces two
__________________________________________________________________________
2
signalling pathways, one is MyD88-dependent and the other is MyD88-independent,
inducing the production of proinflammatory cytokines and type I interferons 13, 14.
Cells involved in innate immunity
Monocytes/Macrophages and Neutrophils
Macrophages are generally a population of ubiquitously distributed mononuclear
phagocytes responsible for numerous homeostatic, immunological, and inflammatory
processes. Their wide tissue distribution makes these cells well suited to provide an
immediate defence against foreign elements prior to lymphocyte immigration. Because
macrophages participate in both the specific immunity via antigen presentation, and in the
non-specific immunity against bacterial, viral, fungal, and neoplastic pathogens, it is not
surprising that macrophages display a range of functional and morphological phenotypes 15.
Macrophages have an innate capacity to recognize pathogen-expressed surface structures
and play an important role in immunity to infections. The macrophages are phagocytic
cells, and the phagocytosis is facilitated by Fc receptors for IgG and IgE present on their
cell surface
16
. They process and present antigens to T cells in association with MHC
antigens, initiating the immune response. In addition, macrophages are also involved in the
secretion of many products, some of which are toxic. The macrophage activity is enhanced
by cytokines, notably IFN-γ, secreted by T- helper (Th) cells. Activated macrophages are
more effective than resting ones in eliminating potential pathogens, and they also express
higher levels of MHC class II molecules, allowing them to function more effectively as
antigen-presenting cells (APCs). Activated macrophages are producing a number of reactive
oxygen intermediates and reactive nitrogen intermediates that have potent antimicrobial
activities. When macrophages are activated by LPS together with IFN-γ, they begin to
express high levels of nitric oxide synthase (NOS), an enzyme that oxidizes L-arginine to
yield nitric oxide (NO). Recent evidence suggests that much of the antimicrobial activity of
macrophages is due to NO 17.
Neutrophils can phagocyte foreign substances, and they also use both oxygen
dependent and independent pathways to generate antimicrobial substances.
__________________________________________________________________________
Amre Nasr, 2008
3
NK cells and Dendritic cells (DCs)
NK cells have cytolytic activity against tumours or pathogen-infected cells. They
can also release cytokines, including IFN-γ, TNF, granulocyte-macrophage colony
stimulating factor (GM-CSF) and chemokines 18, 19. The functions of NK cells are regulated
by a complex balance between inhibitory and activating signals, that are generated by an
array of different cell-surface receptors after engagement by their specific cellular ligands18.
Two major subsets of NK cells are distinguished by their low or high expression of
CD56 (CD56bright and CD56dim respectively). CD56dim NK cells constitute the majority of
NK cells among PBMCs and excel in cytotoxicity. CD56bright NK cells are less effective in
cytotoxicity but are very potent cytokine producers 20, 21.
DCs and their precursors are considered to be sentinels of the immune system; they
can circulate through the blood and non-lymphoid peripheral tissues, where they eventually
become resident cells 22. DCs are well known as professional APC, able to activate naïve T
cells, and are thus critical for the development of adaptive immune responses
23
. DCs also
have important effector functions during the innate immune response, such as pathogen
recognition and cytokine production. In fact, various subsets of DCs have been described,
that are associated with specific pathogen recognition mechanisms, locations, phenotypes,
and types of Ag presentation and cytokine production 24. The DCs also up-regulate the costimulatory molecules that are required for an effective interaction with T cells24.
Both NK cells and DCs are specialised cells of the innate immune system, which
play a crucial role during the early phase of the inflammatory reaction. The reciprocal
interaction of these cells results in a potent, activating cross-talk
18, 22
. DCs can prime
resting NK cells, which, in turn after activation, might induce DC maturation 25. However,
NK cells negatively regulate the function of DCs also by killing immature DCs in peripheral
tissues 21. Thus, NK cells have a role in the editing of mature DCs, based on the selective
killing of mature DCs that do not express optimal surface densities of MHC class I
molecules 21.
Adaptive Immunity
The adaptive immune responses are mediated by lymphocytes. These cells recognize
individual pathogens specifically and they possess memory. Lymphocytes fall into two
major categories: T and B lymphocytes. These two types of cells recognize particular
structures via their receptors that are responsible for the enormous diversity seen in the
__________________________________________________________________________
4
adaptive immune system. B cells recognize antigens through the surface immunoglobulins
(Ig), B-cell receptors (BCR), and the T cells through the T-cell receptors (TCR), these
receptors have unique specificities. The recognition of antigen by the TCR is dependent of a
their presentation in the context of MHC molecules. T and B-cells are mainly responsible
for cell-mediated and humoral immunity, respectively. The differentiation of the cells of the
adaptive immune system is dependent on a cross talk with cells of the innate immune
system, and cytokines constitute important mediators in this context.
MHC
The MHC codes for glycoproteins, which are highly specialised molecules with the
ability to form unusually stable complexes with peptide antigens, to be presented on the cell
surface of APCs, required for the activation of T cells system 26. There are two classes of
MHC molecules, class I and class II, which present antigenic peptides that are processed by
the proteasomal pathway and the lysosomal pathway, respectively 26. The MHC alleles are
highly polymorphic, different hosts usually having different MHC genotypes, and they
therefore recognize different spectrums of antigen epitopes 27. MHC plays an important role
in immune regulation. Hosts vary genetically in many of the factors controlling the immune
responses. Many studies have shown associations between MHC polymorphism and disease
susceptibility 28-31.
Cytokines
Interactions between cells in the immune system can be mediated by soluble
proteins, cytokines. All cytokines are produced in small amounts in response to external
stimuli, such as microbes, and generally have a short half-life. Cytokines bind to highaffinity receptors on target cells. Most cytokines act on the cells that produce them (called
autocrine actions) or on adjacent cells (paracrine actions). A minority of cytokines enters
the circulation and acts on distant tissues (endocrine hormonal actions).
Cytokines are involved in the regulation of the innate and adaptive immunity. They
play an important role in activating macrophages to produce large amounts of cytokines
34
, also activating and regulating lymphocytes
35
32-
. Depending on the effect of the immune
response, some cytokines may be referred to Th1 or Th2-associated cytokines.
__________________________________________________________________________
Amre Nasr, 2008
5
T Lymphocytes
Early T cell precursors do not express the CD4 or CD8 co-receptors (doublenegative) and begin the recombination of the TCR gene segments, of which there are 4: α,
β, γ, and δ. Recombination begins at the δ, γ, and β loci, and if expression of the γδ TCR is
successful, commitment to the γδ T cell lineage results
36
. Most γδ T cells remain double-
negative and leave the thymus, populating lymphoid tissues and epithelia. Antigen
recognition by γδ TCR expressing T cells does not require processing and is not MHC
restricted 37. The γδ T cells represent a relatively rare type of T cells in humans, and their
function appears to be distinct from that αβ T cells
38
. Alternatively, a successful β locus
recombination results in β TCR expression, which pairs with the surrogate α receptor (preTα) and forms the pre-TCR. With the immunotyrosine-based activation motif (ITAM) rich
CD3-signaling machinery, the pre-TCR provides ligand-independent signals, enabling
commitment to the αβ T-cell lineage and CD4/CD8 coexpression (double-positive; DP). T
cells are further classified by the type of TCR they express on their surface, having either
the αβ or γδ combination
39
. The αβ+ TCR expressing T cells have evolved primarily to
recognize antigens as peptide fragments in association with MHC antigens present on
antigen-presenting cells (APCs) 40. The αβ+ T cells may differentiate into several subsets, all
with the accessory marker CD3, in the TCR/CD3 complex, combining with MCH class I
(CD8+) or MHC class II (CD4+)41, 42. The CD4+ T cells, designated T helper (Th) cells,
recognize peptides, which are bound to MHC class II molecules. Th cells are polarized
towards either Th1 or Th2 subsets, depending on the nature of the cytokines they produce at
the site of activation 43. CD8+ T cells, referred as CTLs, recognize antigens bound to MHC
class I molecules. CD8+ T cells produce cytotoxic proteins, including perforin and
granzymes, and secrete them at the point of contact with the target cell, the immunologic
synapse, which results in specific killing without bystander-cell damage. Perforin is a
membrane-disrupting protein that facilitates the ability of granzymes to induce apoptosis in
the target cell. In addition to cytolysis, CD8+ effectors produce IFN-γ and TNF 44.
Classically, CD4+ effectors were viewed in the context of the Th1-Th2 paradigm,
but other subsets have emerged, including IL-17–producing T cells (Th17) and T cells with
regulatory function (Treg) 45. The factors that determine the differentiation of these subsets
in vivo are not completely understood, although it seems that the cytokine milieu may be a
major factor.
__________________________________________________________________________
6
Th1/ Th2 responses
Both Th1 and Th2 subsets are generated from a naïve T cells
46
. The Th1/Th2
concept rests largely on a dichotomy in their cytokine profiles, reviewed in recent research
involving human subjects 47.
Th1 cells specialize in macrophage activation by IFN-γ production and contactdependent stimulation by using a variety of cell-surface co-stimulatory ligands and, thus
playing a major role in intracellular pathogen clearance and delayed-type hypersensitivity.
Reactions of Th1 differentiation is directed by IFNs generated by the innate response to
infection, which ultimately leads to up-regulation of T-bet, the master regulator of Th1
differentiation. T-bet directs IFN-γ production and IL-12 receptor expression. The presence
of IL-12 results in STAT4 activation, further enhancing IFN-γ production and Th1-effector
formation.
Th2 cells produce IL-4, IL-5, and IL-13, and specialize in regulating B-cell antibody
responses. They drive B-cell proliferation by IL-4 and contact-dependent CD40 ligand:
CD40 binding, augmenting humoral defences against extracellular pathogens. Furthermore,
IL-4 and IL-5 enable IgE production and eosinophilic inflammation, important for the
clearance of helminthic infections, but also highly relevant in the allergic response. Th2
differentiation is initiated by weak TCR signals coupled with IL-4 receptor signaling and
signal transducer and activator of transcription-6 (STAT-6) activation. This results in upregulation of the transcription factor GATA-3, the master regulator of Th2 differentiation.
GATA-3 enhances Th2-cytokine production and inhibits Th1 pathways.
B cells
B cells provide the humoral immunity against extracellular pathogens through
antibody production. Antibodies neutralize pathogens and toxins, facilitate opsonization,
and activate complement. In addition to binding soluble peptides, the BCRs are capable of
binding conformational epitopes, including protein epitopes discontinuous in the primary
structure, as well as non-peptide antigens, such as polysaccharides and nucleic acids. In
most cases, primary infection or vaccinations result in a prolonged production of highaffinity specific antibodies, the basis of the adaptive humoral immunity. So-called natural
IgM antibodies, produced in the absence of infection, are of lower affinity, often polyreactive and weakly auto-reactive, and bind to many conserved pathogen-associated
polysaccharides. These antibodies play a role in the first-line defence against bacterial
infections and assist in the clearance of endogenous cellular debris.
__________________________________________________________________________
Amre Nasr, 2008
7
Naïve follicular B cells reside in the follicles of secondary lymphoid tissues.
Antigen arrives in lymphoid organs as soluble molecules or as immune complexes, or is
transported there by DCs 48. BCR cross-linking initiates receptor-mediated endocytosis and
antigen processing. Antigen-engaged follicular B cells migrate to the T-B interface, the
border between the T-cell zone and B-cell follicle, increasing the probability of encounter
with primed helper T cells of cognate specificity. On such an encounter, signals from Tcell–derived cytokines and CD40L:CD40 binding sustain B-cell activation and promote
immunoglobulin class switch recombination (CSR).
Immunoglobulins
Immunoglobulins (Ig) generally assume one of two roles: Ig may act as plasma
membrane bound antigen receptors on the surface of B cells or as antibodies free in cellular
fluids functioning to intercept and eliminate antigenic determinants or pathogens. In either
role, antibody function is intimately related to its structure. When bound on the surface, the
Ig functions as a receptor involved in differentiation, activation and apoptosis, while the
secreted form can neutralize foreign antigens and recruit other effector components
49
. Ig
are composed of four polypeptide chains: two "light" chains (lambda or kappa), and two
"heavy" chains (alpha, delta, gamma, epsilon or mu). The type of heavy chain determines
the Ig isotype (IgA, IgD, IgG, IgE, IgM, respectively). Light chains are composed of 220
amino acid residues, while heavy chains are composed of 440-550 amino acids. Each chain
has "constant" and "variable" regions as shown in the (Figure 1).
Figure 1: A simplified structure of the Ig molecule
*Modified from Ollila and Vihinen 50
__________________________________________________________________________
8
Structural differences between Ig are used for their classification. Several different
types of heavy chain exist that define the class or "isotype" of an antibody. These heavy
chain types vary between different animals. More specifically, the isotype is determined by
the primary sequence of amino acids in the constant region of the heavy chain, which in
turn determines the three-dimensional structure of the molecule. Since Igs are proteins, they
can act as antigens, eliciting an immune response that generates anti-Ig antibodies.
However, the structural (three-dimensional) features that define isotypes are not
immunogenic in an animal of the same species, since they are not seen as "foreign". For
example, the five human isotypes, IgM, IgD, IgG, IgE and IgA are normally found in all
humans.
Another means of classifying Ig is defined by the term "allotype". Like isotypes,
allotypes are determined by the amino acid sequence and corresponding three-dimensional
structure of the constant region of the Ig molecule. Unlike isotypes, allotypes reflect genetic
differences between members of the same species. This means that not all members of the
same species will possess any particular allotype. Therefore, injection of any specific
human allotype into another human could possibly generate antibodies directed against the
structural features that define that particular allotypic variation.
A third means of classifying Ig is defined by the term "idiotype". Unlike isotypes
and allotypes, idiotypes are determined by the amino acid sequence and corresponding
three-dimensional structure of the variable region of the Ig molecule. In this regard,
idiotypes reflect the antigen-binding specificity of any particular antibody molecule.
Idiotypes are so unique that an individual person is probably capable of generating
antibodies directed against their own idiotypic determinants.
Ig classes
Upon antigen activation naïve B cells secrete antibodies of the IgM class.
Subsequent to primary IgM production, the B cells may terminally differentiate into IgM
producing plasma cells, or switch to production of other isotypes than IgM. In the complex
process of Ig synthesis, rearrangements in gene segments (VHDJH in heavy chain and VLJL
in light chain) and somatic mutations produce variations in amino acid sequences in the
amino-terminal portions of the chain. The concentrations of the five isotypes in serum vary
widely, reflecting both different numbers of B cells producing each isotype and different
intrinsic half-lives of the Ig classes.
__________________________________________________________________________
Amre Nasr, 2008
9
IgM antibodies are the largest Ig (900 kilodaltons), which can exist as a monomeric
form on the membrane of B cells, but is normally secreted by plasma cells as a pentamer
together with a J-chain
51
. IgM is found in blood and lymph fluid and is the first type of
antibody made in response to an infection. Furthermore, IgM may cause other cells of
immune system to destroy foreign substances
52
. During an immune response, B
lymphocytes can switch expression of Ig class (isotype) from IgM to IgG, IgE, or IgA
51
.
IgM appears to have a role in the defence against parasitic infections 53.
IgD is a monomer and presented on B cells during early stages of differentiation.
Very little is found in the serum. IgD antibodies have a role in B cell signalling, but no other
apparent effector role in the host defence is known 54.
IgG antibodies are monomeric, found in all body fluids, and are the dominating
isotype in humoral immune responses, constituting 75% to 80%) of all antibodies in the
serum. Human IgG is divided into four subclasses, IgG1- IgG4, where IgG1 is the most
common (66%), followed by IgG2 (24%), IgG3 (7%) and IgG4 (3%)
55
. IgG1 and IgG3
antibodies are predominantly produced in response to protein antigens,
stimulation may result in an increased proportion of IgG4
57
56
but chronic
. In contrast, carbohydrate
56
antigens most often induce IgG2 responses . The different subclasses differ in their ability
to activate complement, IgG1 and IgG3 being the most effective ones, and IgG2 is a weak
activator, whereas IgG4 does not activate complement. IgG2 antibodies are very important
in targeting encapsulated bacteria. IgG antibodies are the only isotype that can cross the
placenta in the pregnant woman to help protecting the fetus.
IgA provides mucosal immune protection as a result of its ability to interact with the
polymeric Ig receptor (pIgR), an antibody transporter expressed on the basolateral surface
of epithelial cells
58
. IgA antibodies protect mucosal surfaces that are exposed to outside
foreign substances. In humans there are two heavy chain subclasses of IgA: IgA1 and IgA2.
IgA1 is monomeric primarily found in the serum. IgA2 can polymerize into multimers
linked by a J piece. IgA2 is transported into the secretions by the cooperative effort of
epithelial cells and lymphocytes 59. Dimeric IgA, produced by sub-mucosal B cells, binds to
an epithelial cell-membrane protein called secretary IgA (SIgA) 59.
One important role of IgA is to bind food antigens in the intestinal tract and prevent
the triggering of pro-inflammatory responses. The relative inability of IgA to initiate
__________________________________________________________________________
10
inflammatory responses allows food antigens to sequester without deleterious consequences
60
.
IgE antibodies are found in the lungs, skin, and mucous membranes. Very low
amounts of IgE are present in the serum. Mast cells have high affinity Fc-epsilon receptors
(FcεR) that scavenges IgE so quickly that its serum half-life is only 2 days 61. IgE displayed
on the mast cell surface mediates immediate hypersensitivity or allergic reactions
62
. IgE
appears also to have a role in defence against parasitic infections 63.
Ig allotypes
The Ig GM and KM allotypes are hereditary antigenic determinants of the Ig heavy
chains (γ) and the κ-type light chains, respectively
64
. The striking qualitative and
quantitative differences in the distribution of these determinants among different races, raise
questions concerning the nature of the evolutionary selective mechanism that maintains this
variation. Associations between Ig allotypes and specific antibody responses could be a
selective force for the maintenance of various haplotypes and their frequencies. Significant
associations between certain GM and KM allotypes and immune responsiveness to
particular infectious pathogens and to polysaccharide vaccines were reported
62
. The
importance of GM and KM allotypes will be discussed under the “Related Background”
section.
Fc receptors
Cell surface receptors for the Ig Fc domain of immunoglobulin (Ig) are known to be
expressed on all cells of the immune system. Fc receptors (FcR) play an important role in
immune regulation, as they serve to link antibody-mediated immune responses with cellular
effector functions. Specific FcRs exist for all Ig classes, including IgA (FcαR), IgD (FcδR),
IgE (FcεR), IgG (FcγR), and IgM (FcμR).
As the work in this thesis deals with IgG isotypes, allotypes and Fc receptors
particular Fc gamma RIIa, there are more details on each of them in the following sections.
The FcγR family
The cell-surface receptors for Fc portion of IgG molecules are now known to
consist of three sub-families of related molecules, designated FcγRI, II and III
65
. FcγRI
(CD64), which includes FcγRIa FcγRIb FcγRIc, is derived from a single gene in humans.
The functional receptor consists of a single trans-membrane polypeptide of about 40 KD,
__________________________________________________________________________
Amre Nasr, 2008
11
FcγRI is the only high- affinity receptor for the Fc domain of IgG molecules, and binds
human IgG1 or IgG3 antibodies with a Kd of 10-8 to 10-9 M
66
. The affinity for IgG2 or
IgG4 antibodies is much lower.
In the case of the FcγRII (CD32) gene subfamily, which includes FcγRIIa and
FcγRIIb, it is encoded by at least three separate genes in humans. The affinity of FcγRII for
human IgG1 or IgG3 is relatively weak, for example Kd greater than 10-7 M, and is
essentially nonexistent for IgG2 and IgG4.
Members of the FcγRIII (CD16) subfamily, including FcγRIIIa and FcγRIIIb, are
encoded by two genes in humans. The affinity of FcγRIII for IgG molecules is similar to
that FcγRII65.
The Structure of FcγRIIa
FcγRII identifies a family of type I (cytoplasmic carboxyl terminus) membrane
glycoprotein isoforms of 40 kDa. At least nine human FcγRII isoforms may be produced,
deriving from three genes, FcγRIIa, FcγRIIb and FcγRIIc, located at chromosome 1q23.
The major differences between the isoforms are in the cytoplasmic sequences, although
there are also differences in the extracellular regions, so that some antibodies distinguish
FcyRIIa from FcyRIIb isoforms (Figure 2). In the intracellular region, the FcγRIIb isoforms
have an immunoreceptor tyrosine-based inhibitory motif (ITIM)
67
, while FcγRIIa has an
immune-receptor tyrosine-based activation motif (ITAM) 68.
Figure 2: The family of Fc receptors for human IgG*
*Modified from Falk Nimmerjahn and Jeffrey V. Ravetch, 2008 69.
Function of FcγRIIa
FcγRII is expressed strongly on B cells, monocytes, macrophages, granulocytes
mast cells and platelets. A proportion of T cells express lower levels of CD32 70. There are
characteristic, and functionally significant, differences in isoform expression between
__________________________________________________________________________
12
different cell types. Interestingly, B and T lymphocytes express the ITIM isoforms, while in
the other haematopoietic cells the ITAM form is expressed almost exclusively.
It is a heterogeneous family of molecules (Figure 2) that plays a critical role in
immunity, by linking the humoral and the cellular responses
65
. Depending on their
cytoplasmic region and/or their associated chains, FcγR display coordinate and opposing
roles in the immune system. The activating FcγR contains an ITAM in their cytoplasmic
region or in their associated signal transducing units 65, which may initiate inflammatory,
cytolytic and phagocytic activities of immune effector cells
contain an ITIM in their cytoplasmic tail
73
71, 72
. The inhibitory FcγR
, which upon cross-linking with ITAM-
containing receptors, down regulate immunological responses.
Allelic variants of FcγRIIa
Functional consequences of FcγRIIa, and implications for the host defence and host
susceptibility to disease, have both acquired and inherited components. FcγRIIa gene has
two single-nucleated polymorphisms (SNPs) at position 27 and 131 on the amino acid
sequence 74. The SNP in the FcγRIIa gene (G494A) (rs1801274) is of particular interest as it
has functional consequences.
FcγRIIa, expressed on mononuclear phagocytes, neutrophils, and platelets, has two
co-dominantly expressed alleles, H131 and R131, which differ at amino acid position 131,
in the extracellular domain (histidine or arginine, respectively), an area which strongly
influences ligand binding. The allelic variants differ substantially in their ability to bind
human IgG2 49, 75. H 131 is a high-binding allele and R131 is low binding. Because IgG2 is
a poor activator of the classical complement pathway, FcγRIIa-H131 is essential for
handling IgG2 immune complexes. The genotype distribution of FcγRIIa in Caucasian and
African American populations is ~ 25% homozygous for H131 ~ 50% heterozygous, and ~
25% homozygous for R131. Among Asians, the frequency of the R131 allele is much
lower, and <10% of the population are homozygous for R131 76.
__________________________________________________________________________
Amre Nasr, 2008
13
MALARIA
Malaria remains a devastating global health problem. Worldwide, an estimated 300–
500 million people contract malaria each year, resulting in 1.5–2.7 million deaths
annually77, 78. Malaria has a broad distribution in both the subtropics and tropics, with many
areas of the tropics endemic for the disease 79. Malaria is estimated to cost Africa more than
$12 billion annually and it accounts for about 25% of all deaths in children under the age of
five on that continent
80
. Because of the increase in global travel to, and immigration of
people from areas endemic for malaria, the incidence of imported cases of malaria in
developed countries has risen. In addition, drug-resistant P. falciparum malaria continues to
spread and at present involves almost all areas of the world. An increasing number of
travellers are exposed to drug-resistant plasmodia.
Malaria is caused by obligate intra-erythrocytic protozoa of the genus Plasmodium.
Humans can be infected with one (or more) of the following five species: Plasmodium
falciparum, P. vivax, P. ovale, P. malariae and P. knowlesi. Plasmodia are primarily
transmitted by the bite of an infected female Anopheles mosquito, but infections can also
occur through exposure to infected blood products (transfusion malaria) and by congenital
transmission. Among the five species, P. falciparum is the predominant one, and
responsible of most of malaria-related morbidity and mortality.
Characteristics of malaria
Life cycle and morphology
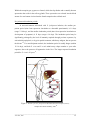
When the infected anopheline mosquito takes a blood meal, sporozoites are
inoculated into the bloodstream (Figure 3). Within 2-30 minutes sporozoites enter
hepatocytes and begin to divide into exo-erythrocytic merozoites (tissue schizogony). For P.
vivax and P. ovale, dormant forms called hypnozoites may typically remain quiescent in the
liver until a later time; P. falciparum does not produce hypnozoites. Once merozoites leave
the liver, they invade erythrocytes and develop into early trophozoites, which are ring
shaped, vacuolated and uninucleated. Once the parasite begins to divide, the trophozoites
are called schizonts, consisting of many daughter merozoites (blood schizogony).
Eventually, the infected erythrocytes are ruptured, releasing merozoites, which subsequently
invade other erythrocytes, starting a new cycle of schizogony. The duration of each cycle in
P. falciparum is about 48 hours. In non-immune humans, the infection is amplified about
20-fold each cycle. After several cycles, some of the merozoites develop into gametocytes,
the sexual stages of malaria, which cause no symptoms, but are infective for mosquitoes 81.
__________________________________________________________________________
14
Within the mosquito gut, zygotes are formed, which develop further and eventually become
sporozoites that reside in the salivary glands. These sporozoites are released into the blood
stream of a new human victim when the female mosquito takes a blood meal.
Pre-patient and incubation periods
In non/semi-immune individuals with P. falciparum infection, the median prepatient period (time from sporozoite inoculation to detectable parasitaemia) is 10 days
(range 5–10 days), and the median incubation period (time from sporozoite inoculation to
development of symptoms) is 11 days (range 6–14 days). The incubation period may be
significantly prolonged by the level of immunity acquired through previous exposures, by
anti-malarial prophylaxis, or by prior partial treatment, which may mitigate, but not prevent
the disease 82. For non-falciparum malaria, the incubation period is usually longer (median
15–16 days), and both P. vivax and P. ovale malaria may relapse months or years after
exposure, due to the presence of hypnozoites in the liver. The longest reported incubation
period for P. vivax is 30 years 83.
Figure 3: The life cycle of P. falciparum in the human (host) and mosquito (vector)*.
*Adopted from Struik and Riley, 2004
89
__________________________________________________________________________
Amre Nasr, 2008
15
Signs and symptoms of malaria
The clinical symptoms of malaria are primarily due to schizont rupture and
destruction of erythrocytes. Malaria can have a gradual or a fulminate course with
nonspecific symptoms. The presentation of malaria often resembles those of common viral
infections; this may lead to a delay in diagnosis 84. The majority of patients experience fever
(>92% of cases), chills (79%), headaches (70%), and diaphoresis (64%)
85
. Additional
common symptoms include dizziness, malaise, myalgia, abdominal pain, nausea, vomiting,
mild diarrhea, and dry cough. Physical signs include fever, tachycardia, jaundice, pallor,
orthostatic hypotension, hepatomegaly, and splenomegaly. Clinical examination in nonimmune persons may be completely unremarkable, even without fever.
Severe malaria
Almost all severe forms and deaths from malaria are caused by P. falciparum.
Rarely, P. vivax or P. ovale produce serious complications, debilitating relapses, and even
death
86
. In 1990, the World Health Organization (WHO) established criteria for severe
malaria in order to assist future clinical and epidemiological studies 87. In 2000, the WHO
revised these criteria to include other clinical manifestations and laboratory values that
portend a poor prognosis based on clinical experience in semi-immune patients (Table 1) 88.
The major complications of severe malaria include cerebral malaria, pulmonary
edema, acute renal failure, severe anaemia, and/or bleeding. Acidosis and hypoglycemia are
the most common metabolic complications. Any of these complications can develop rapidly
and progress to death within hours or days 88. In tropical countries with a high transmission
of malaria (hyperendemic areas), severe malaria is predominantly a disease of young
children (1 month to 5 years of age). Severe malaria accounts for approximately 5% of
imported malaria cases (range 1–38%) 85.
Diagnosis of malaria
Conventional microscopy
Light microscopy of thick and thin stained blood smears remains the standard
method for diagnosing malaria
90
. Thick smears are 20–40 times more sensitive than thin
smears for screening of Plasmodium parasites, with a detection limit of 10–50 trophozoites/
μl. Thin smears allow one to identify malaria species (including the diagnosis of mixed
infections), quantify parasitaemia, and assess for the presence of schizonts, gametocytes,
__________________________________________________________________________
16
and malarial pigment in neutrophils and monocytes. The diagnostic accuracy relies on the
quality of the blood smear and experience of laboratory personnel. The level of parasitaemia
may be expressed either as a percentage of parasitized erythrocytes or as the number of
parasites per micro-liter of blood. In non-falciparum malaria, parasitaemia rarely exceeds
2%, whereas it can be considerably higher (>50%) in falciparum malaria. In non-immune
individuals, hyper-parasitaemia (>5% parasitaemia or >250 000 parasites/μl) is generally
associated with severe disease 91.
Alternative diagnostic methods
Although examination of the thick and thin blood smear is the 'gold standard' for
diagnosing malaria, important advances have been made in diagnostic testing, including
fluorescence microscopy of parasite nuclei stained with acridine orange, rapid dipstick
immunoassay, and polymerase chain reaction assays (PCR). Sensitivity and specificity of
some of these methods approach or even exceed those of the thin and thick smear 92. Rapid
dipstick immunoassays detect species-specific circulating parasite antigens targeting either
the histidine-rich protein-2 of P. falciparum or a parasite-specific lactate dehydrogenase.
Although the dipstick tests may enhance diagnostic speed, microscopic examination
remains mandatory in patients with suspected malaria, because occasionally these dipstick
tests are negative in patients with high parasitaemia, and their sensitivity below 100
parasites/μl is low
90
. Tests based on PCR for species-specific Plasmodium genome are
more sensitive and specific than other tests, detecting as few as 10 parasites/μl blood
93
.
Antibody detection has no value in the diagnosis of acute malaria. It is mainly used for
epidemiologic studies.
__________________________________________________________________________
Amre Nasr, 2008
17
Table 1 Indicators of severe malaria and poor prognosis
Manifestation
Features
Initial World Health Organization criteria from 1990 87
Cerebral malaria
Unrousable coma not attributable to any other cause, with a Glasgow
Coma Scale score ≤ 9. Coma should persist for at least 30 min after a
generalized convulsion
Severe anemia
Hematocrit <15% or hemoglobin < 50 g/l in the presence of parasite
count >10 000/μl
Urine output <400 ml/24 hours in adults (<12 ml/kg/24 hours in
children) and a serum creatinine>265 μmol/l (> 3.0 mg/dl) despite
adequate volume repletion
Renal failure
Pulmonary edema and acute
respiratory distress syndrome
Hypoglycemia
The acute lung injury score is calculated on the basis of radiographic
densities, severity of hypoxemia, and positive end-expiratory
pressure 94
Whole blood glucose concentration <2.2 mmol/l (<40 mg/dl)
Circulatory collapse
(algid malaria)
Abnormal bleeding and/or
disseminated intravascular
coagulation
Systolic blood pressure <70 mmHg in patients > 5 years of age (< 50
mmHg in children aged 1–5 years), with cold clammy skin or a coreskin temperature difference >10°C
Spontaneous bleeding from gums, nose, gastrointestinal tract, or
laboratory evidence of disseminated intravascular coagulation
Repeated generalized convulsions ≥ 3 convulsions observed within 24 hours
Acidemia/acidosis
Macroscopic hemoglobinuria
Arterial pH <7.25 or acidosis (plasma bicarbonate <15 mmol/l)
Hemolysis not secondary to glucose-6-phosphate dehydrogenase
deficiency
Added World Health Organization criteria from 2000 88
Impaired consciousness
Reusable mental condition
Prostration or weakness
Hyper- parasitaemia
Hyperpyrexia
Core body temperature >40°C
Hyperbilirubinemia
*Modified from W.H.O
> 5% parasitized erythrocytes or > 250 000 parasites/μl
(in non-immune individuals)
Total bilirubin >43 μmol/l (> 2.5 mg/dl)
88, 95, 96
__________________________________________________________________________
18
IMMUNITY TO MALARIA
Immunity to malaria infection is complex, mainly due to the complicated life cycle,
that involves different parasitic stages in two different cell types, hepatocytes and
erythrocytes, which result in a large number of antigens. Despite extensive research, the
exact mechanism of immunity related to malaria is not fully understood. It is clear,
however, that it includes both specific and non-specific mechanisms, and that both cellular
and humoral immune responses are involved 97.
Innate resistance
Many intrinsic factors govern the ability of malaria parasites to enter and multiply
within the erythrocytes, some of which are determined genetically. Individuals that lack the
Duffy blood group antigens on their erythrocytes (i.e. Duffy genotype Fya-Fyb-) are resistant
to P .vivax infection, because the receptor for the P. vivax merozoites is associated with
antigens of the Duffy blood group
described in P. knowlesi malaria
98-104
105, 106
. A similar type of resistance has also previously
. Heterozygotes for haemoglobinopathies and other
disorders of the RBCs (thalassemia, sickle cell anaemia and glucose-6-phosphate
dehydrogenase deficiency) promote innate resistance to P. falciparum
107
. Such protection
may be triggered by modification of parasite development within the erythrocytes of these
individuals. However, it has been proposed that these effects may foster processes that lead
to enhancement in the intensity and/or specificity of the adaptive immune response,
enabling individuals carrying these genes to acquire clinical immunity to malaria faster than
others 108.
Haemoglobin variants
Haemoglobin S
Haemoglobin S (HbS) was the first of the structural haemoglobin variants to be
associated with malaria protection, and in the last 60 years it has received more attention
than any since described hamoglobinopathy. HbAS is >90% protective against severe and
lethal malaria 109, 110 and 50% protective against even mild clinical attacks 110. Furthermore,
when children with HbAS do develop clinical malaria, the parasite densities achieved
during such episodes are significantly lower than those in their non-HbAS counterparts
110
.
Innate mechanisms are almost certainly major determinants of this protection, of which
perhaps the most plausible relates to the premature removal of iRBCs through a process
__________________________________________________________________________
Amre Nasr, 2008
19
resembling that seen in glucose-6-phosphate dehydrogenase (G6PD) deficiency
111
.
However, for some years, it has been suggested that the protection might also involve an
immunological component. Recent studies provide strong support for this conclusion. First,
in a cohort study of mild clinical malaria, conducted on the coast of Kenya, the protection
attributable to HbAS was shown to increase with age 112, consistent with the conclusion that
HbAS might accelerate the acquisition of malaria-specific immunity. In a second study
conducted in Gabon, Cabrera et al. used a FACS-based assay to show that, compared to
normal children, HbAS subjects had significantly higher titers of IgG antibodies to the P.
falciparum erythrocytes membrane protein 1
(PfEMP1)
113, 114
. Similarly, HbAS may
contribute to a range of immunological responses to other antigens in the same population
115-118
. The mechanisms, by which HbAS exerts such an important degree of protection,
therefore have still to be fully resolved.
Haemoglobin C
The malaria-protective effect of many of the structural haemoglobin variants is well
supported by population-genetic data, although clinical evidence has been slow to
accumulate. Early studies of haemoglobin C (HbC) were not particularly convincing 119-123,
but more recent observations support a marked effect, particularly against rigorously
defined severe disease 124-126. In contrast to HbS, in which selection for the carrier state has
occurred at the cost of the loss of homozygotes from sickle cell disease, it now appears that
the selective advantage of HbC is greatest in homozygotes (HbCC). In a large study
conducted in Burkina Faso, Modiano et al. found that, although significant, the protective
effect of HbAC was only 30% as compared to >90% in HbCC
126
. A homozygous
advantage for HbC is consistent with observations from two smaller studies that reported no
episodes of severe malaria in HbCC subjects, although both lacked the power to draw
definitive conclusions
124, 125
. These observations square with in vitro studies in which, on
balance, effects have been limited to experiments conducted in HbCC RBCs.
For many years, the favoured hypothesis regarding the protective mechanism of
HbC was that it reduces the ability of P. falciparum parasites to grow and multiply in
variant RBCs
127-130
. However, this mechanism is not strongly supported by in vivo data,
where in most studies, HbC has not been found to exert an effect on parasite densities
126, 131
125,
. A plausible alternative mechanism has now been provided by in vitro studies, which
show that the expression of the major parasite-encoded RBC adhesion protein PfEMP1 is
reduced in HbC-containing RBCs, an effect that is most marked in homozygotes 127, 132. As
__________________________________________________________________________
20
iRBC cytoadherence has been associated with the pathogenesis of severe malaria, this
observation could explain how HbC might influence the incidence of severe disease, yet no
discernible impact on less severe outcomes. Nevertheless, like many other abnormal RBCs,
HbC RBCs are particularly prone to oxidant stress and it is still possible that these
observations might represent some form of in vitro artefact
133
. Further studies are awaited
with considerable interest.
Haemoglobin E
Like most of the other malaria-protective polymorphisms, Haemoglobin E (HbE)
was first identified as a candidate on the basis of its distribution in malaria-endemic
populations. A recent genetic analysis now suggests that in one Thai population, HbE
β26 Glu → Lys, the most frequent variant in Southeast Asia, has reached its current
frequency in less than 5000 years, a rate that is compatible with the conclusion that this
gene expanded under positive selection
134
. Further support for the conclusion, that malaria
was responsible for this selection, has been provided by a hospital-based study conducted in
Thailand, which found that the presence of HbE trait (HbAE) was associated with reduced
disease severity in adults admitted with acute P. falciparum malaria
135
. Like HbC, the
homozygous state of HbE is relatively benign, and it therefore remains possible that
selection might favour both heterozygotes and homozygotes. An intriguing in vitro study
suggests that this is unlikely. Chotivanich et al. 136 cultured P. falciparum parasites in media
containing various mixtures of normal and variant RBCs to show that in subjects with
HbAE, only 25% of RBCs are susceptible to invasion, suggesting that the presence of an
unidentified membrane defect renders the majority of RBCs relatively resistant to infection.
This effect was specific for HbAE and was not seen in cells from subjects with HbEE or
various forms of α-thalassaemia. This raises the possibility that HbAE might protect against
severe malaria by limiting the ability of infections to achieve high parasite densities.
Thalassaemias
Despite compelling population-genetic evidence for malaria-protection by the αthalassaemias (summarised in ref.
137
), all clinical studies conducted to date have proved
confusing. There is now consistent evidence from a number of population studies, showing
that α-thalassaemia is associated with protection from severe and fatal malaria
however, it does not protect against symptom less parasitaemia
140, 142-144
suggest that it even protects against mild episodes of malaria disease
138-141
;
, and few data
140, 142, 144
. In fact,
__________________________________________________________________________
Amre Nasr, 2008
21
paradoxically, in two studies conducted in the Pacifics, the incidence of mild malaria was
higher in young children with α-thalassaemia than in normal children
however, unlike HbAS
110
143, 144
. Interestingly,
, α-thalassaemia has no effect on parasite densities
138-140, 143-145
.
Moreover, while HbAS protects against all forms of clinical malaria, the effect of αthalassaemia appears to be relatively specific to severe malaria anaemia
141, 144
. Therefore,
on the basis of the clinical data available to date, it seems unlikely that α-thalassaemia
protects through a similar mechanism as HbAS, but rather protects against the progression
of individual malaria infections to the point at which they succumb to severe anaemia.
Complement receptors
Parasite sequestration is not due only to endothelial binding. Some P. falciparum
isolates show a phenomenon known as rosetting, where a parasitized erythrocyte binds to
other erythrocytes. One mechanism for rosetting is through PfEMP-1 binding to erythrocyte
CR1, encoded by the CR1 gene
146
. Up to 80% of the population in a malarious region of
Papua New Guinea have an erythrocyte CR1 deficiency, which has been associated both
with polymorphisms in the CR1 gene and with α+ thalassaemia, also common in this
population
147
. In the same study, it was found that both CR1 polymorphisms and α
thalassaemia were independently associated with resistance to severe malaria. However,
studies of CR1 polymorphisms in the Gambia found no evidence of association with disease
severity
148, 149
. In Thailand, an restriction fragment length polymorphism (RFLP) of the
CR1 gene, that is associated with reduced expression of CR1 on erythrocytes, has been
associated (in homozygotes) with susceptibility to severe malaria 150.
Acquired immunity
The slow development of acquired immunity to malaria may be explained by poor
immunogenicity of the parasite and/or that immune responses are strain-specific. A large
number of infections would then be required to encounter the whole repertoire of different
antigens. The presence of high proportions of asymptomatic infections in highly endemic
areas may also be due to strain specific immunity.
Early studies of malaria indicated that malaria immunity is specific to the particular
strain inducing the host immune response, enabling an individual to resist infection by that
particular strain only, but not against heterologous strains 151, 152. Strain diversity in malaria
parasites thus apparently delays development of protective immunity in endemic areas. This
__________________________________________________________________________
22
idea is strongly supported by the finding that the malaria parasite species are polymorphic
for a number of specific genetic markers 153, 154.
Immune mechanisms against the pre-erythrocytic stage
Pre-erythrocytic stage immunity was thought to be directed against the sporozoites
in the circulation and mediated by antibodies, which neutralized the sporozoite infectivity
for hepatocytes
155, 156
. However, since the extracellular sporozoites invade the hepatocyte
within 2-30 minutes of inoculation, anti-sporozoite antibodies must be present in the
circulation at high titters and exert their activity within minutes of infection. Hence,
antibody-mediated protective immunity is unlikely to be completely effective
157
. It is now
generally accepted that the P. falciparum parasite developing within the host hepatocyte is
the major target of protective immunity directed against the pre-erythrocytic stage. Both
+
+
CD8 and CD4 T-cells recognize parasite-derived peptides presented by MHC class І or
class ІІ molecules, respectively, on the surface of infected hepatocytes. Nevertheless,
+
protection against the pre-erythrocytic stage is mediated primarily by CD8 T cells
158
and
involves cytokines and other factors, such as nitric oxide. In vitro treatment of Plasmodium
sp, infected hepatocytes with IFN-γ eliminate P. falciparum or P. berghei parasites from the
cultures 159, 160.
In mice, it has been shown that IL-12 and natural killer cells have an additional role
in the protection induced by immunization with either irradiation attenuated sporozoites or
plasmid DNA 161.
+
An interest in effector CD4 T cells in anti-malarial immunity has increased in
+
recent years. Initial adoptive transfer experiments demonstrated that CD4 T cells of the
Th1 phenotype could protect against Plasmodium sp. challenge in vivo, in the absence of
any detectable cytotoxicity
162
. It has been demonstrated that active immunization with
linear synthetic peptides, derived from P. yoelii proteins, confers solid protective immunity,
+
which is mediated by CD4 T-cells and is absolutely dependent on IFN- γ 163, 164.
Immune mechanisms against the erythrocytic stage
Experiments conducted several years ago in animal models identified the importance
of mainly Th2 responses, including both antibody-dependent
mechanisms of immunity to the erythrocytic stages
167, 168
165, 166
and cell mediated
. B cells and antibodies were
found to play an important role in immunity against malaria parasites, and mice lacking B
__________________________________________________________________________
Amre Nasr, 2008
23
cells are unable to clear infections with the P. chabaudi parasite, rather such mice
developed a chronic parasitaemia
169, 170
. However, others have shown that the effector
functions of both Th1 and Th2 cells are necessary for parasite clearance in animals models
171, 172
.
In humans, although several immune mechanisms have been identified in vitro as
potentially parasite-neutralizing, and several antigens have been identified as targets for the
antibodies involved, knowledge about their relative importance in vivo during natural
infections is still limited.
Different mechanisms maybe involved in the protective effects against malaria
parasite of anti-malarial antibodies that have. These antibodies may mediate their effector
functions against the parasite on their own or in collaboration with effector cells.
IgG obtained from the sera of African adults (malaria-immuned), used for passive
immunization of P. falciparum-infected non-immune Thai patients, demonstrated a
reproducible reduction in parasitaemia and clinical symptoms
173
. On their own, antibodies
against merozoite surface-associated proteins may block RBC invasion
174
or block
merozoite release from schizonts, either by binding to surface exposed antigens, or by
entering iRBC through leaky membrane at the time of rupture 175. Moreover, antibodies can
block sequestration of iRBC in internal organs, allowing the parasite to be cleared by the
spleen 176. Anti-malarial antibodies may also inhibit rosetting of RBC to iRBC 177 and may
therefore protect against severe malaria complications (cerebral malaria).
However, in collaboration with other effector immune cells, parasite antigenspecific antibodies may play an important role via a mechanism called antibody-dependent
cellular inhibition (ADCI)
178, 179
. Anti-malarial IgG can inhibit parasite growth efficiently
in co-operation with normal human monocytes. The parasite-neutralizing effect obtained in
the ADCI assay appeared to mainly be mediated by soluble factors released by the
monocytes upon their uptake of opsonized merozoites
179
. These factors, possibly TNF-α
and nitric oxide, block the division of surrounding intra-erythrocytic parasites. Other studies
have shown that monocytes may also kill parasites by phagocytosis of opsonized whole
infected erythrocytes
180
. A study conducted in Senegal, using crude parasite extracts,
reported that high-avidity cytophilic IgG1 antibodies and IgG3 were predominant in
immune subjects 181. A study from Burkina-Faso showed that specific IgG2 antibody levels
increased with the age of the subjects, in parallel with the development of protection against
infection and malaria disease
182
. These antibodies might mediate phagocytosis or lysis of
iRBCs by monocytes. The mechanism responsible for this type of parasite killing was
__________________________________________________________________________
24
suggested to involve capturing of merozoite- or schizont-bound antibodies by FcRs on the
surface of monocytes
173 183
. In general, cytophilic antibodies may allow opsonisation and
clearance of merozoites or iRBCs by monocytes/macrophages and neutrophils 184, 185.
Naturally acquired protection against malaria in adults has been linked to immune
responses to particular malaria parasite antigens
186, 187
. Many targets of humoral immune
response are polymorphic or clonally variant antigens exposed on the surface of merozoites
or iRBCs, respectively
188, 189
. During the acute natural infection, individuals develop
specific antibody responses to antigens of the parasite variant to which they are exposed 190,
191
. These antibody responses are often, but not always, highly specific
192, 193
. Therefore,
clinical immunity to blood-stage malaria is correlated with the acquisition of a repertoire of
antibodies to different variants of parasite antigens, and is related to exposure and age of the
host
194, 195
. Several target antigens for antibody-mediated inhibition of parasite growth or
invasion have been identified
196, 197
. Antibodies to merozoite-surface antigens (especially
MSP-1 and MSP-2), the apical complex organelles of merozoites (EBA-175, Rhop-1–3,
RAP-1–3 and AMA-1) and the dense granule antigen Pf155/RESA, show the capacity to
inhibit merozoite invasion 196. Two other antigens, SERA and ABRA, which are associated
with the merozoite surface at the time of schizont rupture, are found in the immune clusters
formed by antibodies inhibiting merozoite dispersal in parasite cultures
198
. In addition,
antibodies to SERA have been shown to inhibit P. falciparum growth in vitro and
antibodies to ABRA are efficient inhibitors of merozoite invasion
199
. Of the parasite-
derived antigens expressed on the surface of infected erythrocytes, only Pf332 has been
demonstrated to induce parasite-neutralizing antibodies, inhibiting the intra-erythrocytic
growth of the parasite 200.
In addition, lymphocytes and neutrophils have been demonstrated to exert antibodymediated killing of P. falciparum in vitro
polymorphonuclear leukocytes
204, 205
201, 202
, NK cells
. The monocyte-derived macrophages
206
and γδT cells
56
203
,
are able to kill late
stages of the intra-erythrocytic parasite in the absence of antibodies. iRBCs directly interact
with leucocytes such as NK cells, macrophages and DCs. These interactions may depend on
the infecting parasite phenotype and host genetic factor(s) and, together with previous
exposure and age, influence the balance of immune responses. Pro-inflammatory cytokines,
especially TNF-α, IFN-γ and lymphotoxin (LT), have been associated with severe malaria
disease, still are crucial for the initial control of parasitaemia in human. The mechanisms are
not yet well understood, but secretion of IFN-γ by NK cells, and γδT cells may have
cytotoxic effects on parasite growth as well as activate monocytes and macrophages and
__________________________________________________________________________
Amre Nasr, 2008
25
enhance non-opsonic phagocytosis of iRBCs. The concentrations of anti-inflammatory
cytokines, such as IL-10 and transforming growth factors (TGF)-β, concurrent with those of
pro-inflammatory cytokines appear to have a balancing effect. Such cytokines are also
important growth factors for B cells responses.
RELATED BACKGROUND
The Complex Genetic Basis of Resistance to Malaria
The host genetic basis for resistance to malaria is complex at several levels. It is
likely that many different genes are involved, and that they interact with environmental
variables and with parasite genetic factors. A number of further complexities can be
considered when studying genetic resistance to malaria, namely the range of phenotypes
involved, the practical difficulties of studying families, and the remarkable geographic and
ethnic heterogeneity of malaria-resistance factors.
Susceptibility and/or resistance to malaria can be measured in several ways. Usually,
they are studied in regions with high levels of malaria transmission. When everyone is
repeatedly bitten by infected mosquitoes, many children and adults are likely to have
parasites in the blood. Children may have two or three episodes of malarial fever each year,
and a small minority (e.g. 1%) of the malaria-fever episodes lead to severe malaria.
Different genetic factors may determine the risk of an exposed person to developing
parasitaemia, the risk of a parasitaemia person to becoming ill with malaria fever, and the
risk of a person with malaria fever for develop severe malaria. Parasitaemia and fever can
be regarded as quantitative phenotypes that are ascertained by repeated measurements
within the community, whereas severe malaria is a qualitative phenotype that is typically
ascertained in hospital-based studies. In principle, studies of severe malaria would be
expected to detect genetic factors at each stage in the causal chain of disease progression.
Severe malaria is the phenotype that matters most to vaccine developers and to those
interested in evolutionary selection. Severe malaria is composed of a number of subphenotypes that may occur alone or in combination. In African children, these are cerebral
malaria, severe malarial anaemia, and respiratory distress 207. Several factors may associate
with reduced or increased the risk of severe malaria, some such factors will be discussed
below.
__________________________________________________________________________
26
The role of IgG in protection against malarial infection and disease
There is accumulating evidence for a role of IgG antibodies in protection against
malarial infection and susceptibility to disease. Kinyanjui et al.
208
described long term
antibody responses to malaria in Kenyan children. These responses are very brief. The
brevity of the response has been traditionally attributed to a skew in the response towards
IgG3. Human IgG3 antibodies have a half life of 8 days, whereas the half-lives of IgG1,
IgG2, and IgG4 are up to 23 days. That study found that children have variable kinetics of
antibody production and that IgG1 responses increased with age. Although an antibody
response was generated to the infection and peaked after 7 days, the antibody response only
lasted for approximately 6 weeks. This loss of specific antibody production occurred
regardless of the isotype of the antibody measured, and this raised the question as to
whether antibody decay was faster in children, or if there was a failure to produce longlived plasma cells capable of to secreting antibodies for extended periods of time. IgM
responses were minimal in the children, suggesting that the measured antibodies mainly
reflected a secondary antibody response. However, re-infection did not boost antibody
responses. This intriguing observation could have several explanations. It could suggest that
memory-B cells were deleted; alternatively it could be that high levels of serum antibodies
give negative signals to B cells, or that Treg cells block re-activation of memory responses.
Finally, the splenic architecture is disrupted following infection, and this could prevent
activation of memory-B cells.
FcγRIIa in Malaria
As mentioned previously, FcγRIIa exhibits a genetic polymorphism, FcγRIIaR/H131 coded for different capacities of IgG binding and phagocytosis
49, 209
. A defence
mechanism requiring the association of antigen bound IgG antibodies with FcγRs on
monocytes has been proposed to play an important role in protecting immune African adults
from malaria
210
. FcγRIIa is an important receptor mediating the phagocytic function of
monocytes, macrophages, and neutrophils
37
. Thus, different FcγRIIa alleles affect the
ability of phagocytes to internalize IgG-opsonized micro-organisms, indicating that the
clearance of parasites opsonized with different IgG subclasses, may vary according to host’s
genotype. Considerable differences in the distribution of FcγRIIa alleles among different
ethnic groups have been reported
211
. Human IgG2 is the only IgG subclass that binds
efficiently to FcγRIIa-H/H131, but not to FcγRIIa-R/R131, while both alleles bind IgG1 and
IgG3 with similar efficiency 49.
__________________________________________________________________________
Amre Nasr, 2008
27
A study conducted in Kenya, showed that the polymorphism FcγRIIa-R/R131 may
have an influence on protection against the disease 212. Another study showed an association
between the polymorphism FcγRIIa-H/H131 and susceptibility to cerebral malaria
213
.
Follow-up studies in Thailand and the Gambia found that homozygotes for the 131H allele
are susceptible to cerebral malaria
213, 214
. In the Thai study, the FcγRIIa association
involved interaction with an FcγRIIIb gene polymorphism with cerebral malaria 213.
Ethnic Differences
Differences in susceptibility to malaria between ethnic groups suggest host genetic
factors have a role in determining host susceptibility to malaria
215-218
. In that addition, the
patterns of extent ethnic variation in Africa are important for understanding how genetic
variation affects infectious diseases. Studies in West Africa showed that there are ethnic
groups living in sympatry under the same malaria transmission and exposure, but,
surprisingly, one of these groups, the Fulani, is less susceptible to malaria infection
compared to the other ethnic groups
219, 220
. These findings offer an important approach to
studying the immune genes behind the susceptibility/resistance to malaria.
The Fulani people are traditionally nomadic pastoral people, who are found across
West Africa, often settled in close proximity to other ethnic groups. Studies in Burkina Faso
221
, and more recently in Mali
219
, have documented a significantly lower prevalence of
malaria parasitaemia and fewer clinical attacks of malaria among the Fulani than among
other ethnic groups who live in neighbouring villages. The Fulani have a distinctive culture,
but detailed epidemiological investigations indicate that their resistance to malaria arises
primarily from genetic factors
221
. Importantly, it has also been observed that the Fulani
have high levels of anti-malarial antibodies 222, 223 and a low frequency of protective globin
variants and other classic malaria-resistance factors
224
. There is therefore much interest in
discovering the genetic factors that determine the reduced risk to malaria infection
IgG allotypes
Immunoglobulin GM and KM allotypes, are associated with humoral and cellular
immunological properties, and are thus ideal candidate genetic systems for investigations to
identify risk-conferring factors in malaria pathogenesis. GM and KM allotypes are
associated with the susceptibility to and outcome of several infectious agents
allotypes are strongly associated with IgG subclass concentrations
226
55, 225
. GM
, making them relevant
to malarial immunity, as the protective anti-P. falciparum antibodies are predominantly of
__________________________________________________________________________
28
IgG1 and IgG3 subclasses 227. Furthermore, several population genetic properties of the GM
system – the marked differences in the frequencies of GM allotypes among races, strong
linkage disequilibrium within a race, and racially restricted occurrence of GM haplotypes –
suggest that differential selection, caused by major selective forces like malaria, may have
played an important role in the maintenance of polymorphism at these loci 228.
GM/KM markers could directly affect susceptibility to malaria infection and disease
outcomes by influencing the titre and affinity of anti-malarial antibodies. Although GM and
KM markers are located on the constant region, there is a growing body of evidence for the
involvement of these regions in antibody specificity, which usually are associated with the
variable (V) region of the Ig molecule. Possible mechanisms include direct contribution to
the formation of idiotypic determinants, effect on V-region protein conformation,
modulation of antibody-binding affinity, and linkage disequilibrium with alleles coding for
the V-region epitopes
55, 229, 230
. Additionally, GM/KM allotypes may influence antibody-
dependent cellular inhibition and phagocytosis of malarial parasites by their differential
interaction with FcγR. Evidence for such GM allotypic restriction in the binding affinity of
the IgG molecules has been presented for certain virally encoded FcγRs
231
. It has been
suggested that GM and KM allotypes themselves may not directly influence disease
susceptibility, but that the associations may reflect linkage disequilibrium with other
polymorphisms in the constant region genes or with specific variable region genes 12.
C- reactive protein (CRP)
C-reactive protein (CRP) is an acute phase protein that increases rapidly during
infections and/or inflammations. CRP has the ability to activate various immune cells and
has the capacity to bind to certain FcγRs; and both CRP and IgG share a sequence
homology that is important for FcγR binding
232
. The binding of CRP may compete and
interfere with the binding of protective malaria-specific IgG antibodies. The R131 allele of
FcγRIIa was shown to bind CRP with high affinity
233
. CRP can indirectly mediate
activation of the complement and complement receptors, or direct, through receptors for the
FcγRs or even a putative CRP-specific receptor activation of immune cells. Interestingly,
high levels of CRP were shown to be associated with severity of P. falciparum infection 234,
235
, while asymptomatic malaria infections were shown to be associated with low levels of
CRP
236, 237
, these findings suggest that the levels of CRP are not merely an acute phase
response related to the presence of the parasite in the body. Although the role of CRP is not
well defined in P. falciparum malaria, studies showed that it can be associated with
__________________________________________________________________________
Amre Nasr, 2008
29
complement mediated haemolysis of iRBC and subsequent anaemia
defence against pre-erythroctic stage of malaria
239
238
, as well as in
. In addition, CRP can induce the anti-
inflammatory cytokine IL-10 during early stages of malaria infections 240.
Current evidence suggests that serum CRP level is a complex trait, influenced by
both clinical and genetic factors
estimated to be 40-60%
242-244
241
. The genetic influence of circulating CRP levels is
. There are several SNPs identified, in the CRP promoter
region, and the -286 C<T<A (rs3091244) has been strongly associated with CRP levels 241.
However, the extent to which clinical and genetic variables together account for the total
variation in serum CRP level is unclear.
__________________________________________________________________________
30
THE PRESENT STUDY
Objectives of the study
This study aims at addressing possible associations of FcγRIIa, IgG allotypes, IgG
subclasses, HbAS and CRP in relation to malaria by investigating:
1. Whether FcγRIIa (CD32) polymorphism, in association with anti-malarial IgG
subclass distribution, is associated with susceptibility/resistance to either mild or
severe P. falciparum malaria among Sudanese patients.
2. The distribution of GM and KM allotypes in two sympatric ethnic groups; the
Fulani, who are relatively resistant to develop malaria, and the Masaleit, who are
relatively susceptible to the malaria infection.
3. Whether FcγRIIa (CD32) and HbAS genotypes differ among the two sympatric ethnic
groups, the Fulani and the Masaleit.
4. The distribution of FcγRIIa (CD32) genotypes, anti-malaria IgG subclass levels and
their associations among sympatric Fulani and non-Fulani groups.
5. Whether a polymorphism in the CRP gene could be associated to the differences in
relative resistance/susceptibility to malaria in sympatric ethnic groups in Mali and
Sudan.
Material and Methods used in the Study
The methodologies, study areas and study populations of the included studies are
described in detail in the corresponding papers.
Results and Discussion
This study was performed in eastern Sudan, an area characterized by seasonal and
unstable malaria transmission. The predominant malaria parasite species in the area is P.
falciparum. The study has two components: Hospital- and village-based investigations. The
hospital study was done in New Halfa teaching hospital and it investigated the possible
association between FcγRIIa polymorphisms and the outcome of clinical malaria i.e. severe
versus mild malaria, while the second part was performed at the Daraweesh village (Fulani
ethnic group) and a neighbouring village (non-Fulani, including the Masaleit), as well as in
a village south of Gedarif (Tabarak Allah), where also the Fulani and the Masaleit live
__________________________________________________________________________
Amre Nasr, 2008
31
together in the same area. The people living in Daraweesh belong to the Fulani ethnic group
245, 246
, which were reported to be less susceptible to develop malaria 219, 220.
FcγRIIa polymorphism in relation to malaria (Papers I, III and IV)
In this study we investigated whether FcγRIIa (CD32) polymorphism, is associated
with P. falciparum malaria outcome in eastern Sudan. Our results (Paper I) provide
evidence for a SNP in the FcγRIIa (CD32) gene being associated with susceptibility to
severe P. falciparum malaria in the Sudanese study population. Our data demonstrated that
the R/R131 genotype and R131 allele were statistically significantly associated with severe
malarial disease as compared to mild malaria. Furthermore, the H/H131 genotype was
statistically significantly associated with mild malaria, suggesting that the H/H131 genotype
might be associated with reduced risk of succumbing to severe malaria. In support for this, a
recent study in Ghana, in an area characterized by seasonal malarial transmission, observed
similar findings, with the R/R131 genotype being more frequent in patients with severe
malarial anaemia and cerebral malaria
247
. Although there are other reports suggesting that
the H/H131 genotype is related to protection against malaria
248
, our data are in contrast
with earlier studies performed in areas with higher malaria endemicity. In these studies, a
correlation between the R/R131 genotype and relative protection from malaria was shown
212-214
. The reasons for these discrepant results are most likely due to genetic differences in
the populations studied, as well as differences in gene-environment interactions and in
patterns of malaria transmission in the different study areas. In addition, the haplotype
structure of the gene might differ, so that a possibly linked causal disease association would
be tagged differently by the SNP typed here. Therefore, an in-depth analysis of
polymorphisms, flanking this gene and the haplotype structure is required in future larger
disease association studies.
When comparing Fulani to non-Fulani individuals in the village-based study (Papers
III, IV) for the distribution of FcγRIIa genotypes and allele frequencies, there was a
significant difference between the two ethnic groups regarding the frequency of
homozygotes (H/H131 and R/R131 genotypes). The H/H131 genotype and H131 allele
were significantly more frequent in the Fulani, who are considered to be less parasitized, as
compared to the non-Fulani ethnic group (including the Masaleit). In contrast, the R/R131
genotype and R131 allele were significantly more frequent in the Masaleit. Thus, the
findings show pronounced interethnic differences in allele and genotype frequencies of this
FcγRIIa SNP, suggesting distinct genetic backgrounds of the sympatric Fulani and non-
__________________________________________________________________________
32
Fulani ethnic groups. Thus, our study population setting is similar to that previously
described in Burkina Faso, where the Fulani were shown to be genetically distant from the
sympatric Mossi and Rimaibé ethnic groups, on the basis of HLA class I markers 249.
While the impact of the FcγRIIa polymorphism on the susceptibility to protection
against malaria remains unclear, this polymorphism has been shown to play an important
role in many other infections. For example, the R/R131 genotype has been found to be a
risk factor for recurrent bacterial respiratory infections, while the H131 allele appears to
play an important role in protection from other infections, in particular those caused by
encapsulated bacteria
250
. However, the pattern of the functional activity of FcγRIIa
genotype in a population may be concurrent with the pattern of the IgG subclass responses
within the respective population. The importance of FcγRIIa in parasite-neutralizing events
mediated by monocytes was indicated by Tebo et al., using THP-1 cells transfected with the
131H or 131R allele of the receptor
251
. While cells expressing the 131R allele of the
receptor showed enhanced phagocytosis of P. falciparum-infected erythrocytes, with sera
dominated by IgG1 antibodies, the phagocytosis by cells expressing the 131H receptor
showed a preference for sera containing high IgG3 antibody levels. Furthermore, in a recent
study on antibody-dependent cell-mediated inhibition of P. falciparum growth in vitro, the
optimal parasite-neutralizing activity by human monocytes was dependent on the
cooperation between FcγRIIa and FcγRIII receptors 252.
Taken together, our study revealed that the FcγRIIa-R/R131 genotype is associated
with increased susceptibility to develop severe malaria in a Sudanese study population, and
that the H/H131 genotype, is more likely to be associated with mild malaria in this
population. In addition, the Fulani, who appear to be less susceptible to malaria as
compared to their sympatric tribes, the H/H131 genotype and H131 allele, are more
common. Thus, it is obvious that genetic polymorphisms in FcγRIIa (CD32) might affect
the outcome of malaria disease differently in different ethnic groups.
IgG GM/KM allotypes in relation to malaria (Paper II)
This part of the study was performed in an area South of Gedarif region (Tabarak
Allah), eastern Sudan, where Fulani and Masaleit ethnic groups live in sympatry. Studying
different malariometric indices (parasite rate, parasite density, and splenomegaly), showed
that, the two ethnic groups differed significantly in various malariometric parameters, the
Fulani being less parasitized than the Masaleit subjects. This study aimed at investigating
__________________________________________________________________________
Amre Nasr, 2008
33
GM/KM allotypes as possible markers of humoral immunity/immunoglobulins that might
reflect differences in the susceptibility to develop malaria between Fulani and Masaleit.
GM allotypes are strongly associated with IgG concentrations
226
, making them
relevant to malaria immunity. These observations led us to hypothesize that the distribution
of GM/KM allotypes in Fulani are significantly different from their sympatric ethnic
neighbours.
Our results showed that the distribution of GM and KM phenotypes is statistically
significantly different between the two ethnic groups. The frequency of GM 6 was
significantly lower in the Fulani than that in the Masaleit subjects while the distribution of
KM phenotypes by itself was not significantly different between the two ethnic groups. In
addition, the prevalence of a particular combination of KM and GM phenotypes, however,
was significantly different: the combined frequency of KM 1,3 and GM 1,17 5,6,13,14
phenotypes was significantly more frequent in the Masaleit ethnic group than in the Fulani.
It is interesting to note that GM 6 is extremely rare or absent in populations other than
Negroes, and this marker is commonly present in the Masaleit ethnic group. Furthermore,
GM 6 has the most variable frequency (8–62%), possibly a result of differential selection
caused by infectious diseases like malaria
228
. GM/KM markers could directly affect the
susceptibility to malaria infection and disease outcomes by influencing the titre and affinity
of anti-malarial antibodies
226
. As mentioned previously, the GM and KM markers may
influence ADCI and phagocytosis of malarial parasites by their differential interaction with
FcγRs especially FcγRIIa, which has been implicated in malaria infection and pathogenesis
212-214
.
Taken together, our results showed differences in frequencies of GM/KM allotypes
in two sympatric ethnic groups, which might contribute to the relative resistance of Fulani
to P. falciparum malaria.
IgG subclasses in relation to malaria (papers I and IV)
In this part of the study the levels of anti-malarial IgG subclasses were compared
among patients with different clinical presentation (mild versus severe malaria) from the
hospital-based study (Paper I), as well as in the Fulani from Daraweesh and their non-Fulani
neighbours (Paper IV).
In the study (Paper IV), antibody responses to four P. falciparum blood-stage
antigens, the AMA1, two alleles of MSP2 (3D7 and FC27) and the Pf332-C231 antigen,
were analysed in the Fulani and non-Fulani ethnic groups. In general, the serum levels of
__________________________________________________________________________
34
IgG1 and IgG3 subclass antibodies were found to be at higher levels than IgG2 and IgG4
antibodies in both ethnic groups. However, the antibody levels varied between the different
antigens. When comparing the antibody levels between the two ethnic groups, the nonFulani showed significantly higher levels of IgG1 antibodies reactive with AMA1. The
responses of IgG2 antibodies reactive with AMA1 were, however, significantly higher in
the Fulani than in the non-Fulani ethnic group. Although antibodies reactive with MSP23D7 showed a similar pattern of interethnic differences in IgG subclass levels as those
reactive with AMA1, IgG1 antibodies reactive with MSP2-FC27 were higher in the Fulani
as compared to the non-Fulani. Regarding anti-Pf332-C231 antibodies, IgG3 and IgG4
subclasses were at significantly higher levels in the non-Fulani as compared to the Fulani.
In paper I, which was a hospital based study, the levels of anti-malarial IgG1 and
IgG3 antibodies were statistically significantly higher in the mild malaria patients when
compared with the severe malaria patients. Our results are in line with previous data 173, 210,
251, 253
regarding the association between cytophilic antibodies and protection in individuals
naturally exposed to P. falciparum malaria. Several studies in Burkina Faso and Mali have
shown that the Fulani have significantly higher levels of anti-malarial antibodies than other
sympatric ethnic groups
221-223
, including all the IgG subclasses, except IgG4
227
.
In
contrast, in the present study, the non-Fulani had significantly higher levels anti-malarial
IgG1 and IgG3 antibodies for all the malarial antigens studied, except for MSP2-FC27, than
the Fulani. The Fulani, however, tended to have significantly higher levels of anti-malarial
IgG2 antibodies than the non Fulani group. These discrepant results can most likely be
explained by differences in patterns of malaria transmission in the different study areas
254
245,
. In Daraweesh an entomological inoculation rate (EIR) of one infective bite/year has
been estimated based on surveys in drought-free years
255
. Almost all malaria cases in
Daraweesh are clinically uncomplicated, probably because of the continuous monitoring by
health team, who visits the village on daily basis and provided free drug treatment 245, 254. It
should be recalled that Sudan is a country characterised by hypo/meso-endemicity, with
seasonal and unstable malarial transmission, unlike Burkina Faso and Mali, which are
hyper/holo endemic 227.
Anti-malarial antibodies against a crude P. falciparum extract (F32) and the
recombinant Pf332-C231 antigens were assessed in different patient categories. The levels
of anti-malarial antibodies of the different Ig isotypes differed between the study groups;
being higher in individuals with mild malaria than in those with severe malaria. Thus, the
levels of IgG1 and IgG3 antibodies against crude F32 were significantly higher in the mild
__________________________________________________________________________
Amre Nasr, 2008
35
malaria patients compared with those seen in the severe malaria patients. High levels of
malaria-specific IgG1 and IgG3 antibodies against crude F32 were associated with reduced
risk of clinical episodes of malaria. Malaria-specific IgG subclasses against the recombinant
Pf332-C231 showed a biased response towards IgG1, IgG2 and IgG3. The levels of antiC231 IgG1 antibodies were found to be significantly higher in patients with mild malaria
than in those with severe malaria. The independent effect before and after adjustment for
sex, age and ethnicity also showed that higher levels of anti-Pf332-C231 IgG1 antibodies
were statistically associated with reduced risk of severe malaria. Low levels of IgG4
antibodies reactive with to the crude malarial antigen (F32) were found to be associated
with lower risk of clinical malaria.
Taken together, our results revealed that natural acquisition of immunity against
clinical malaria appeared to be more associated with IgG1 and IgG3 antibodies, signifying
their roles in parasite-neutralizing immune mechanisms.
IgG subclasses in relation to FcγRIIa polymorphism (Papers I and IV)
In the hospital-based part of the study (Paper I), the allele distribution FcγRIIa
polymorphisms and IgG subclass profiles were analysed and our results showed that antimalarial IgG3 antibodies reactive with the crude malarial antigen, in combination with the
H/H131 genotype, were associated with a reduced risk of severe malaria. In addition, a
correlation between low levels of IgG2 antibodies specific to Pf332-C231 antigen were
found to be associated with lower risk of severe malaria in individuals carrying the H131
allele.
The village-based study (Paper II) showed that the non-Fulani had significantly
higher levels of anti-malarial IgG1 and IgG3 antibodies for all the studied malaria antigens,
except for MSP2-FC27, than the Fulani. The Fulani, however, tended to have significantly
higher levels of anti-malarial IgG2 antibodies than the non-Fulani group. The levels of IgG2
and IgG3 antibodies were associated with the H/H genotype, while higher levels of IgG1
antibodies were associated with the R/R131 genotype. Similarly, the Fulani showed a higher
frequency of the FcγRIIa-H131 allele and higher levels of anti-malarial IgG2 antibodies
than the sympatric non-Fulani ethnic groups. A similar higher frequency of the FcγRIIaH131 allele and a higher proportion of IgG2 among anti-malarial antibodies, were recently
observed in the Fulani in Mali compared to a sympatric non-Fulani ethnic group (Israelsson
et al. in press). There is accumulating evidence for IgG1 and IgG3 antibodies playing an
important role in protection against malarial disease
178
. However, the contribution of
__________________________________________________________________________
36
parasite-reactive IgG2 antibodies in protection against clinical malaria
susceptibility to disease
210, 256
182
and/or in
has been indicated in some epidemiological settings. In
Burkina Faso, the association between anti-malarial IgG2 antibodies and protection against
malaria, was suggested to be due to the relatively high prevalence of the IgG2 binding
H/H131 genotype in that study population
182
. Whether the higher levels of anti-malarial
IgG2 antibodies and the higher frequency of the FcγRIIa-H131 allele in the Fulani may
contribute to the lower susceptibility to malaria in this ethnic group as compared to
sympatric non-Fulani ethnic groups, remains to be investigated in extended epidemiological
studies.
While the FcγRIIa-R131 molecule shows a similar binding of the cytophilic
subclasses IgG1 and IgG3 210, 251, the H131 receptor binds IgG3 more efficiently, and is also
the only FcγR that binds IgG2 efficiently
49, 209, 257-259
. We showed that IgG3 antibodies
specific to crude malarial antigen were more associated with reduced risk of clinical malaria
in individuals with H/H131 than in individuals with R/R131 genotype. In individuals
carrying the H131 receptor, IgG2 antibodies should be considered as cytophilic as it binds
FcγR more efficiently. Others have shown that lower levels of IgG2 subclass antibodies to
several merozoite surface proteins are independently associated with the FcγRIIa-R131
allele and with the carriage of haemoglobin AS in Gabonese children
116
. In addition, the
association between IgG2 antibodies and malaria protection in Burkina Faso may be due to
the relatively high titre of the IgG2 binding H/H131 genotype in that study population 182.
Taken together, our study revealed that the FcγRIIa-R/R131 genotype is associated
with higher levels of anti-malarial IgG1 antibodies and that the FcγRIIa-H/H131 genotype is
associated with higher levels of IgG2 and IgG3. Thus, the higher IgG2 antibody levels seen
in the Fulani may be due to the relatively high prevalence for the H/H131 genotype in this
group. However, further studies are needed to elucidate if the FcγRIIa-R/H131
polymorphism may be a contributing factor to the differential susceptibility to malaria seen
in different ethnic groups.
HbAS in relation to malaria (Paper II)
This study was village-based, involving the Fulani living in the Daraweesh village
and neighbouring sympatric non-Fulani ethnic groups, where the prevalence of HbAS was
analysed. Of the children, 108 carried haemoglobin AA and 32 carried haemoglobin AS.
The frequency of the HbAS genotype was lower in the Fulani compared to the Masaleit. For
__________________________________________________________________________
Amre Nasr, 2008
37
the Hb A/S allele frequencies, there was a trend between the Fulani and Masaleit, the S
allele being more frequent than A allele in the latter ethnic group.
A similar finding was reported from Burkina Faso, where the HbAS genotype was
less frequent in the Fulani, compared to other sympatric ethnic groups, indicating that this
polymorphism is not the reason for the lower susceptibility to malaria seen in the Fulani
224
.
Indeed, the protection provided by HbAS carriage mainly concerns the severe complications
of the malaria disease, such as cerebral malaria and/or severe anaemia, and not, or to a much
lesser extent, the frequency of infection or the incidence of uncomplicated forms
28, 224
,
which are unequivocally reduced among Fulani 220, 221, 224, 249. Thus, our findings are in line
with the previous observation, that the lower susceptibility to malaria seen in the Fulani
does not involve the classical malaria resistance gene HbAS. This suggests that other still
unknown genetic factor(s) or other contributing factor(s) are involved in the differential
susceptibility to malaria seen between the Fulani and other sympatric ethnic groups living in
Sudan.
In our study of the FcγRIIa complex in the individuals of the two ethnic groups
carrying the HbAS genotype (sickle cell trait patients), the frequency of the FcγRIIaH/H131 genotype was higher, as compared to that of the R/R131 genotype. In a study from
USA, an association between FcγRIIa and HbAS has previously been observed, with the
H/H131 genotype being overrepresented in black children with sickle cell disease and a
history of H. influenzae type b infection
260
. These findings indicate that the FcγRIIa-
H/H131 genotype is more likely to be associated with the HbAS genotype, suggesting a
selection for the AS and H/H131 genotypes as a result of protection against severe malaria.
CRP in malaria in relation to ethnicity (Paper V)
In an attempt to define the role of CRP in susceptibility to malaria, as well as to
further define potential immunological factors contributing to the ethnic difference in
malaria susceptibility, we analyzed the -286 C>T>A CRP genotypes and the circulating
CRP levels in relation to malariometric data in sympatric individuals from two countries,
Sudan and Mali, with marked different malaria endemicity.
For the Fulani groups in both countries, the AA, AT and TT genotypes were the
least common and the AC, CT and CC genotypes were the most common. The Malian nonFulani had the highest frequency of AA, AT and AC, while the Sudanese non-Fulani had
the highest frequency for AA, CC and AC genotypes. Post-hoc analyses showed that the
AA, AT and CC frequencies in Mali and the AA, AT, CT and TT frequencies in Sudan,
__________________________________________________________________________
38
differed significantly between the two ethnic groups. As mentioned previously, the
influence of genetic variations on the levels of circulating CRP is estimated to be 40-60% of
the concentrations of CRP 242, and a functional polymorphism in the promoter region of the
CRP gene (-286 C>T>A) has been strongly associated with the circulating concentrations of
CRP 261. Previous studies on this CRP polymorphism has revealed that the A allele is more
common in African American than in Caucasian populations, and this difference in
genotypes is associated with higher CRP levels in the former population
241
. This suggests
that CRP could have been selected for in the African ancestors as a factor beneficial for the
survival from infectious diseases.
Our study showed that the allele frequencies were different between the two ethnic
groups in both Mali and Sudan. The A allele frequency (high producer allele for CRP
levels) was higher in the non-Fulani groups from both countries. In both Mali and Sudan,
the, the frequency of non-A allele carriers dominated (low producer alleles for CRP levels)
in the Fulani groups, while the A-allele carriers were pre-dominantly found in the nonFulani groups. The non-Fulani group presented a high frequency of A allele carriers, and in
line with this, there was a tendency for higher CRP levels in the non-Fulani group than in
the Fulani of Mali. All individuals in the Sudanese study populations had undetectable CRP
levels, most likely because all blood samples were collected before the rainy season and all
individuals were negative for malaria parasites.
In conclusion, we suggest that the levels of CRP seen in asymptomatic individuals
could influence the malaria infection. Also, our data suggest that the ethnic differences seen
in the functional -286 CRP genotypes could be a contributing factor for the lower
susceptibility to malaria seen in the Fulani from both Sudan and Mali.
__________________________________________________________________________
Amre Nasr, 2008
39
CONCLUDING REMARKS AND FUTURE PERSPECTIVES
Considerable efforts have been made to identify effector and host genetic factors
that contribute to protection and/or pathogenesis of malaria. The very wide range of such
factors involved and their diversity present an arduous task in malaria research.
Nevertheless, some factors have been identified as “host genetic factors” and therefore
worth pursuing to investigate.
The data presented in this thesis lend support to earlier suggestions that human
monocytes are important effector cells in the innate and adaptive immune responses. In the
case of malaria, we have identified that the FcγRIIa-R/H131 polymorphisms may play a
role in regulating the clinical outcome of the disease. In the case of severe malaria, the
R/R131 genotype was significantly associated with susceptibility to severe, while the
presence of H131 allele and H/H131 genotype appeared to be an important factor protecting
from severe malarial disease. Binding of antibodies to the merozoites or infected red blood
cells results in their opsonization, usually involving the cytophilic IgG1 and IgG3
subclasses. However, in individuals carrying the FcγRIIa-H131 allele, high levels of IgG2
antibodies have been positively associated with protection from malarial infection and
disease. The FcγRIIa-H/H131 and high level for IgG2 might be involved in better parasite
clearance and also reduce the risk of severe malaria outcome. These results suggest that a
protective role of IgG3 and IgG2, which may activate effector cells through FcγRIIa in
malarial infection and disease.
The distribution of GM phenotypes as a whole, as well as a particular combination
of KM and GM phenotypes, differed significantly between the two ethnic groups. The data
suggest that GM allotypes may contribute to the genetic aetiology of malaria. The results
presented here provide an impetus for large-scale studies to explore the role of GM and KM
allotypes with IgG subclass concentrations in the pathogenesis of malaria with differential
prevalence in the Fulani as compared with sympatric ethnic groups. It would be interesting
to investigate any possible associations between GM and KM allotypes and FcγRIIa in the
Fulani as compared with their sympatric non-Fulani neighbours.
Our data showed that the Fulani had lower parasite rates, lower malaria
susceptibility, lower frequency of the sickle cell trait (HbAS) and higher frequency of the
FcγRIIa-H/H131 genotype and H131allele carriers. The results also indicate that the relative
resistance to malaria infection seen in the Fulani does not rely mainly on the HbAS trait.
Rather, other factor(s), such as FcγRIIa-H/H131 genotype, may play an important role in
__________________________________________________________________________
40
the immune responses, which can control the infection and consequently avoid development
of clinical malaria.
Our data showed that the Fulani ethnic group had lower levels of anti-malarial
antibodies (both IgG1 and IgG3) and higher IgG2 antibody levels compared to their nonFulani neighbours. The higher IgG2 antibody levels seen in the Fulani were shown to be
associated with relatively a high prevalence of the H/H131 genotype in this ethnic group.
Unexpectedly, the results of the present study suggest that the magnitude and the quality of
the anti-malarial humoral response is not the key element for relative resistance to malaria
infection observed in the Fulani. Rather, the results suggest that IgG2 when acting as a
cytophilic isotype may contribute to parasite clearance.
The influence of genetic variations on the levels of circulating CRP is expected to be
associated with mutations in the promoter region of the CRP gene. Our data showed that, in
the Fulani ethnic group, from both Sudan and Mali, a significantly higher frequency of the
low producer alleles (non-A alleles) was observed, while the high producer allele carriers
(A allele) were predominantly found in the non-Fulani. Based on these results, we suggest
that the lower susceptibility to malaria seen in the Fulani group could be influenced by this
polymorphism. A previous study indicated that CRP can induce an anti-inflammatory IL-10
response
240
. However, further studies are needed to investigate the role of IL-10 and other
cytokines in relation to protection against malaria in different endemic settings in Africa.
Finally, our data provide evidence that the Fulani and the Masaleit/other sympatric
non-Fulani ethnic groups are genetically different. Furthermore, the FcγRIIa polymorphism,
the GM/KM allotypes and CRP genotype may be involved or contribute as host genetic
factor(s), which could be the real determinant of resistance/protection against malaria
infection in the Fulani ethnic groups.
To address the actual association of FcγRs polymorphism, especially with malaria
out-come, analysis of haplotype studies are required. Sequencing of FcγRs genes in
individuals who differ significantly in susceptibility/resistance to malaria would improve
our knowledge. It will be of interest to study the associations and interactions of FcγRs
polymorphisms, GM/KM allotypes and anti-malarial IgG subclasses in the Fulani as
compared to the non-Fulani and also among Fulanis with differential susceptibility to
develop malaria infection (intra-Fulani differences). In addition, the impact of IL-4, IL-10
and TGF-β polymorphisms on anti-malarial IgG subclasses and susceptibility to develop
malaria infection is of importance to investigate.
__________________________________________________________________________
Amre Nasr, 2008
41
__________________________________________________________________________
42
ACKNOWLEDGMENTS
I wish to express my warm gratitude to everyone who has contributed in any way in making
this thesis a reality and give me support during these intense and fascinating years, especially:
My supervisors at Stockholm University: Klavs Berzins, for your patience, careful
comments, advice and good sense of humor that has been really helpful, thanks for always having
the time and ready to answer my questions with such a great patience and knowledge which really
added a lot to this thesis. Marita Troye-Blomberg, for introducing me into the fascinating world of
immunology, for her continuous support, patience and encouragement even when life gets tough and
Gehad ElGhazali, currently at King Fahad Medical City in Saudi Arabia, my warmest gratitude for
always finding solutions, for believing on me and giving me the strength and opportunity to carry on
and finish my studies. Thank you all for your great help and concern, really thanks for the support
that I got over the years till I developed as a scientist/researcher, and for making me believe in
myself. Thanks for always being willing to answer questions and to give hands in all what I request.
Former and present seniors at the Department of Immunology: Carmen Fernández, Eva
Severinson, Eva Sverremark-Ekström and Manucherhr Abedi Valugerdi. Thank you for
sharing your vast knowledge in the field of immunology and for your support and help with my
work.
Hedvig Perlmann for your inspiration, kindness, friendly character and your motivation in
science. I would like to recognize late Peter Perlmann whose generous and dedicated personality
has been, and continues to be, an inspiration for researchers in the field of immunology.
Ann Sjölund and Margareta Hagstedt “Maggan”; for your excellent technical support
and for your responding to any call for help. I have enjoyed working with you and miss our coffee
times.
Gelana Yadeta and Anna-Leena Jarva, for making the department run smoothly and for
excellent administration, always having a smile, and for your readiness to assist.
Past and present colleagues at the department of Immunology - Nnaemeka C. Iriemenam,
Manijeh Vafa, Salah Eldin Farouk, Elisabeth Israelsson (Lisa),
Nancy Awah, Halima
Balogun, John Arko-Mensah, Ahmed Bolad, Pablo Giusti, Anna-Karin Larsson, Petra
Amoudruz, Jacob Minang, Alice Nyakeriga, Jubayer Rahman, Camilla Rydström, Valentina
Screpanti, Ylva Sjögren, Anna Tjärnlund, Khaleda Qazi Rahman, Nina-Maria Vasconcelos,
Stefania Varani, Nora Bachmayer, Shanie Saghafian-Hedengren, Ebba, Olga, Irene,
Fernando, Maria, Charles, Olivia Simone, Yvonne Sundström and Magdi M. Ali, for making
the department a nice and friendly place to work, co-authors, for your contributions, friends, in and
out of the university, for the very beautiful time.
Additional co-authors, for the great collaboration that made this thesis a reality: Hayder A.
Giha, Scot M. Montgomery, Omran F. Osman, Mattias Ekström, Gishanthi Arambepola,
Amagana Dolo, Ogobara K. Doumbo, Per Tornvall, Robin F. Anders.
__________________________________________________________________________
Amre Nasr, 2008
43
We thank our collaborators at Uppsala University for providing a friendly atmosphere and
stimulating discussions that paved the way to yield our novel and interesting finding.
We also thank the collaborators at Khartoum University, Faculty of Medicine especially
Prof. Mustafa I. Basher, all staff at the MalRC-Biochemistry department for assistance with
storage and hosting in Khartoum.
We gratefully acknowledge the continued support and collaboration of the people in the
study areas. Dr. Ishag Adam, Dr. Azam Ebid, Dr. Aisha Abdo, Abdallh Hifzalla, Ehasan
Osman, Mustafa Hamid, Faiz Omer, Adil Ameen, New Halfa Teaching Hospital staff, Halfa
Suger Factory staff, are thanked for excellent assistance in the field and laboratory.
The villagers of Daraweesh are thanked for kind are generous way they have housed the
study. I also would like to thank the donors, their families, New Halfa, for their participation in this
study.
I would like to greet my teachers, colleagues and friends in Sudan, for great help and
support. To the memory of Shadin Abd-Ghafar, for the immense inspiration and tremendous
knowledge.
We gratefully thank my home meds Amir and Kamal for the healthy home environment
and great help and support during the last four years.
We also thanks my friends, in Sweden for cheerful times, special thanks to the Sudanese
Association in Stockholm & Uppsala, for creating a friendly and supportive community that
makes all of us feel home. Special thanks to my friends Noman, Amani, Raja Mirghani, Ahmed,
Seham, Babikir Elebaid, Moawia, Samar, Susan, Hana, Nada, Noon, Mazin, Tota, Hadia, Ali,
Hind, Abdallh, Amani, Ghada, Zafir, Azza, Nagat, Misara, Amir Al-bodani, Nagat, Mohamed,
Hussam, Yassir, Seif Eyazal, Barsham, Randa, Anika, Elain, Liana, Hani, Anna and my
friends at KTH and Cassi restraint at Karlplan.
Past and present diplomatic staff at Sudanese Embassy Stockholm/ Oslo, special thanks to
the ambassadors and their families: Omar Gorani, Dr. Omar Abdalmaged, Zeinab Mahmoud,
Dr. Mohamed A. Altom for your encouragement and support.
Finally, and most of all, I would like to express my deepest and warmest gratitude to my
beloved family, my father, Osman Nasr, my brother Ayman, my sisters Iman, Wisal and Abeer, to
all my nephews and my nieces. Words are not enough to express my love and gratitude to you.
Whatever I have, whatever I am, whatever I get, it is all because of you. This thesis is in fact yours.
The studies were financially supported by the by grants from the Swedish Agency for
Research Development with Developing Countries (SIDA, SAREC), the Swedish Medical Research
Council (VR), as well as a grant within the BioMalPar European Network of Excellence (LSHP-CT2004-503578).
I received a fellowship from the Swedish Institute (SI) during the period of
September 2006 to June 2007.
__________________________________________________________________________
44
REFERENCES
1.
2.
3.
4.
5.
6.
7.
8.
9.
10.
11.
12.
13.
14.
15.
16.
17.
18.
19.
20.
Delves, P. J. & Roitt, I. M. The immune system. First of two parts. N Engl J Med
343, 37-49 (2000).
Medzhitov, R. & Janeway, C. A., Jr. Innate immunity: impact on the adaptive
immune response. Curr Opin Immunol 9, 4-9 (1997).
Biron, C. A., Nguyen, K. B., Pien, G. C., Cousens, L. P. & Salazar-Mather, T. P.
Natural killer cells in antiviral defense: function and regulation by innate cytokines.
Annu Rev Immunol 17, 189-220 (1999).
Moretta, A. et al. Major histocompatibility complex class I-specific receptors on
human natural killer and T lymphocytes. Immunol Rev 155, 105-17 (1997).
Janeway, C. A., Jr. Approaching the asymptote? Evolution and revolution in
immunology. Cold Spring Harb Symp Quant Biol 54 Pt 1, 1-13 (1989).
Fraser, I. P., Koziel, H. & Ezekowitz, R. A. The serum mannose-binding protein and
the macrophage mannose receptor are pattern recognition molecules that link innate
and adaptive immunity. Semin Immunol 10, 363-72 (1998).
Gewurz, H., Mold, C., Siegel, J. & Fiedel, B. C-reactive protein and the acute phase
response. Adv Intern Med 27, 345-72 (1982).
Schwalbe, R. A., Dahlback, B., Coe, J. E. & Nelsestuen, G. L. Pentraxin family of
proteins
interact
specifically
with
phosphorylcholine
and/or
phosphorylethanolamine. Biochemistry 31, 4907-15 (1992).
Anderson, K. V., Jurgens, G. & Nusslein-Volhard, C. Establishment of dorsalventral polarity in the Drosophila embryo: genetic studies on the role of the Toll
gene product. Cell 42, 779-89 (1985).
Lemaitre, B., Nicolas, E., Michaut, L., Reichhart, J. M. & Hoffmann, J. A. The
dorsoventral regulatory gene cassette spatzle/Toll/cactus controls the potent
antifungal response in Drosophila adults. Cell 86, 973-83 (1996).
Coban, C., Ishii, K. J., Horii, T. & Akira, S. Manipulation of host innate immune
responses by the malaria parasite. Trends Microbiol 15, 271-8 (2007).
Propert, D. Immunoglobulin allotypes and RFLPs in disease association. Exp Clin
Immunogenet 12, 198-205 (1995).
Akira, S. TLR signaling. Curr Top Microbiol Immunol 311, 1-16 (2006).
Kawai, T. & Akira, S. TLR signaling. Cell Death Differ 13, 816-25 (2006).
Taylor, P. R. et al. Macrophage receptors and immune recognition. Annu Rev
Immunol 23, 901-44 (2005).
Unkeless, J. C., Fleit, H. & Mellman, I. S. Structural Aspects and Heterogeneity of
Immunoglobulin Fc Receptors. Adv Immunol 31, 247-70 (1981).
Lefkowitz, D. L. & Lefkowitz, S. S. Macrophage-neutrophil interaction: a paradigm
for chronic inflammation revisited. Immunol Cell Biol 79, 502-6 (2001).
Cooper, M. A., Fehniger, T. A. & Caligiuri, M. A. The biology of human natural
killer-cell subsets. Trends Immunol 22, 633-40 (2001).
Cooper, L. J., Robertson, D., Granzow, R. & Greenspan, N. S. Variable domainidentical antibodies exhibit IgG subclass-related differences in affinity and kinetic
constants as determined by surface plasmon resonance. Mol Immunol 31, 577-84
(1994).
Dulphy, N. et al. An unusual CD56(bright) CD16(low) NK cell subset dominates the
early posttransplant period following HLA-matched hematopoietic stem cell
transplantation. J Immunol 181, 2227-37 (2008).
__________________________________________________________________________
Amre Nasr, 2008
45
21.
22.
23.
24.
25.
26.
27.
28.
29.
30.
31.
32.
33.
34.
35.
36.
37.
38.
39.
40.
Romagnani, C. et al. CD56brightCD16- killer Ig-like receptor- NK cells display
longer telomeres and acquire features of CD56dim NK cells upon activation. J
Immunol 178, 4947-55 (2007).
Banchereau, J. & Steinman, R. M. Dendritic cells and the control of immunity.
Nature 392, 245-52 (1998).
Banchereau, J. et al. Immunobiology of dendritic cells. Annu Rev Immunol 18, 767811 (2000).
Shortman, K. & Liu, Y. J. Mouse and human dendritic cell subtypes. Nat Rev
Immunol 2, 151-61 (2002).
Gerosa, F. et al. Reciprocal activating interaction between natural killer cells and
dendritic cells. J Exp Med 195, 327-33 (2002).
Engelhard, V. H. Structure of peptides associated with class I and class II MHC
molecules. Annu Rev Immunol 12, 181-207 (1994).
Madden, D. R., Garboczi, D. N. & Wiley, D. C. The antigenic identity of peptideMHC complexes: a comparison of the conformations of five viral peptides presented
by HLA-A2. Cell 75, 693-708 (1993).
Hill, A. V. et al. Common west African HLA antigens are associated with protection
from severe malaria. Nature 352, 595-600 (1991).
Hill, A. V. et al. Molecular analysis of the association of HLA-B53 and resistance to
severe malaria. Nature 360, 434-9 (1992).
Hill, G. R. et al. Interleukin-11 promotes T cell polarization and prevents acute
graft-versus-host disease after allogeneic bone marrow transplantation. J Clin Invest
102, 115-23 (1998).
Wright, K. L. et al. CIITA stimulation of transcription factor binding to major
histocompatibility complex class II and associated promoters in vivo. Proc Natl
Acad Sci U S A 95, 6267-72 (1998).
Belosevic, M., Davis, C. E., Meltzer, M. S. & Nacy, C. A. Lymphokine-induced
macrophage resistance to infection with Leishmania major. Adv Exp Med Biol 239,
239-44 (1988).
Belosevic, M., Finbloom, D. S., Van Der Meide, P. H., Slayter, M. V. & Nacy, C. A.
Administration of monoclonal anti-IFN-gamma antibodies in vivo abrogates natural
resistance of C3H/HeN mice to infection with Leishmania major. J Immunol 143,
266-74 (1989).
Nacy, C. A. et al. Lymphokine regulation of macrophage effector activities. Adv
Exp Med Biol 239, 1-11 (1988).
Hopkins, S. J. The pathophysiological role of cytokines. Leg Med (Tokyo) 5 Suppl
1, S45-57 (2003).
Lauritsen, J. P., Haks, M. C., Lefebvre, J. M., Kappes, D. J. & Wiest, D. L. Recent
insights into the signals that control alphabeta/gammadelta-lineage fate. Immunol
Rev 209, 176-90 (2006).
Indik, Z., Kelly, C., Chien, P., Levinson, A. I. & Schreiber, A. D. Human Fc gamma
RII, in the absence of other Fc gamma receptors, mediates a phagocytic signal. J
Clin Invest 88, 1766-71 (1991).
O'Brien, R. L., Lahn, M., Born, W. K. & Huber, S. A. T cell receptor and function
cosegregate in gamma-delta T cell subsets. Chem Immunol 79, 1-28 (2001).
Miescher, G. C., Howe, R. C., Lees, R. K. & MacDonald, H. R. CD3-associated
alpha/beta and gamma/delta heterodimeric receptors are expressed by distinct
populations of CD4- CD8- thymocytes. J Immunol 140, 1779-82 (1988).
Braciale, T. J. et al. Antigen presentation pathways to class I and class II MHCrestricted T lymphocytes. Immunol Rev 98, 95-114 (1987).
__________________________________________________________________________
46
41.
42.
43.
44.
45.
46.
47.
48.
49.
50.
51.
52.
53.
54.
55.
56.
57.
58.
59.
60.
de Gast, G. C. et al. Recovery of T cell subsets after autologous bone marrow
transplantation is mainly due to proliferation of mature T cells in the graft. Blood 66,
428-31 (1985).
Thibault, G. & Bardos, P. Compared TCR and CD3 epsilon expression on alpha beta
and gamma delta T cells. Evidence for the association of two TCR heterodimers
with three CD3 epsilon chains in the TCR/CD3 complex. J Immunol 154, 3814-20
(1995).
Mosmann, T. R. Properties and functions of interleukin-10. Adv Immunol 56, 1-26
(1994).
Tschopp, J. & Nabholz, M. Perforin-mediated target cell lysis by cytolytic T
lymphocytes. Annu Rev Immunol 8, 279-302 (1990).
Weaver, C. T., Harrington, L. E., Mangan, P. R., Gavrieli, M. & Murphy, K. M.
Th17: an effector CD4 T cell lineage with regulatory T cell ties. Immunity 24, 67788 (2006).
Lafaille, J. J. The role of helper T cell subsets in autoimmune diseases. Cytokine
Growth Factor Rev 9, 139-51 (1998).
Dent, L. A. For better or worse: common determinants influencing health and
disease in parasitic infections, asthma and reproductive biology. J Reprod Immunol
57, 255-72 (2002).
Bergtold, A., Desai, D. D., Gavhane, A. & Clynes, R. Cell surface recycling of
internalized antigen permits dendritic cell priming of B cells. Immunity 23, 503-14
(2005).
Warmerdam, P. A., van de Winkel, J. G., Vlug, A., Westerdaal, N. A. & Capel, P. J.
A single amino acid in the second Ig-like domain of the human Fc gamma receptor
II is critical for human IgG2 binding. J Immunol 147, 1338-1343 (1991).
Ollila, J. & Vihinen, M. B cells. Int J Biochem Cell Biol 37, 518-23 (2005).
Kracker, S. & Radbruch, A. Immunoglobulin class switching: in vitro induction and
analysis. Methods Mol Biol 271, 149-59 (2004).
Perlmann, H. et al. Dissection of the human antibody response to the malaria antigen
Pf155/RESA into epitope specific components. Immunol Rev 112, 115-32 (1989).
Couper, K. N., Phillips, R. S., Brombacher, F. & Alexander, J. Parasite-specific IgM
plays a significant role in the protective immune response to asexual erythrocytic
stage Plasmodium chabaudi AS infection. Parasite Immunol 27, 171-80 (2005).
Vladutiu, A. O. Immunoglobulin D: properties, measurement, and clinical relevance.
Clin Diagn Lab Immunol 7, 131-40 (2000).
Pandey, J. P. Immunoglobulin GM and KM allotypes and vaccine immunity.
Vaccine 19, 613-7 (2000).
Troye-Blomberg, M. et al. Human gamma delta T cells that inhibit the in vitro
growth of the asexual blood stages of the Plasmodium falciparum parasite express
cytolytic and proinflammatory molecules. Scand J Immunol 50, 642-50 (1999).
Jefferis, R. & Kumararatne, D. S. Selective IgG subclass deficiency: quantification
and clinical relevance. Clin Exp Immunol 81, 357-67 (1990).
Mostov, K. E. & Deitcher, D. L. Polymeric immunoglobulin receptor expressed in
MDCK cells transcytoses IgA. Cell 46, 613-21 (1986).
Macpherson, A. J. & Slack, E. The functional interactions of commensal bacteria
with intestinal secretory IgA. Curr Opin Gastroenterol 23, 673-8 (2007).
Stokes, C. R., Soothill, J. F. & Turner, M. W. Immune exclusion is a function of
IgA. Nature 255, 745-6 (1975).
__________________________________________________________________________
Amre Nasr, 2008
47
61.
62.
63.
64.
65.
66.
67.
68.
69.
70.
71.
72.
73.
74.
75.
76.
77.
78.
79.
80.
81.
Wagner, B. et al. Occurrence of IgE in foals: evidence for transfer of maternal IgE
by the colostrum and late onset of endogenous IgE production in the horse. Vet
Immunol Immunopathol 110, 269-78 (2006).
Ng, O. T. et al. Naturally acquired human Plasmodium knowlesi infection,
Singapore. Emerg Infect Dis 14, 814-6 (2008).
Farouk, S. E. et al. Different antibody- and cytokine-mediated responses to
Plasmodium falciparum parasite in two sympatric ethnic tribes living in Mali.
Microbes Infect 7, 110-7 (2005).
Grubb, R. Advances in human immunoglobulin allotypes. Exp Clin Immunogenet
12, 191-7 (1995).
Ravetch, J. V. & Bolland, S. IgG Fc receptors. Annu Rev Immunol 19, 275-90
(2001).
Ravetch, J. V. & Kinet, J. P. Fc receptors. Annu Rev Immunol 9, 457-92 (1991).
Daeron, M. et al. Regulation of tyrosine-containing activation motif-dependent cell
signalling by Fc gamma RII. Immunol Lett 44, 119-23 (1995).
van de Winkel, J. G. & Capel, P. J. Human IgG Fc receptor heterogeneity: molecular
aspects and clinical implications. Immunol Today 14, 215-21 (1993).
Nimmerjahn, F. & Ravetch, J. V. Fcgamma receptors as regulators of immune
responses. Nat Rev Immunol 8, 34-47 (2008).
Mantzioris, B. X., Berger, M. F., Sewell, W. & Zola, H. Expression of the Fc
receptor for IgG (Fc gamma RII/CDw32) by human circulating T and B
lymphocytes. J Immunol 150, 5175-84 (1993).
Hulett, M. D. & Hogarth, P. M. Molecular basis of Fc receptor function. Adv
Immunol 57, 1-127 (1994).
Hulett, M. D., Witort, E., Brinkworth, R. I., McKenzie, I. F. & Hogarth, P. M.
Multiple regions of human Fc gamma RII (CD32) contribute to the binding of IgG. J
Biol Chem 270, 21188-94 (1995).
Amigorena, S. et al. Cytoplasmic domain heterogeneity and functions of IgG Fc
receptors in B lymphocytes. Science 256, 1808-12 (1992).
Junghans, R. P. Next-generation Fc chimeric proteins: avoiding immune-system
interactions. Trends Biotechnol 15, 155 (1997).
Clark, M. R., Stuart, S. G., Kimberly, R. P., Ory, P. A. & Goldstein, I. M. A single
amino acid distinguishes the high-responder from the low-responder form of Fc
receptor II on human monocytes. Eur J Immunol 21, 1911-6 (1991).
Lehrnbecher, T. et al. Variant genotypes of the low-affinity Fc gamma receptors in
two control populations and a review of low-affinity Fc gamma receptor
polymorphisms in control and disease populations. Blood 94, 4220-4232 (1999).
Muentener, P., Schlagenhauf, P. & Steffen, R. Imported malaria (1985-95): trends
and perspectives. Bull World Health Organ 77, 560-6 (1999).
Sachs, J. & Malaney, P. The economic and social burden of malaria. Nature 415,
680-5 (2002).
Breman, J. G., Egan, A. & Keusch, G. T. The intolerable burden of malaria: a new
look at the numbers. Am J Trop Med Hyg 64, iv-vii (2001).
Snow, R. W., Korenromp, E. L. & Gouws, E. Pediatric mortality in Africa:
Plasmodium falciparum malaria as a cause or risk? Am J Trop Med Hyg 71, 16-24
(2004).
Garcia, L. S. Malaria and babesiosis. In: Garcia LS. , editor. Diagnostic Medical
Parasitology. Washington, DC: American Society for Microbiology, pp. 159–204
(2001).
__________________________________________________________________________
48
82.
83.
84.
85.
86.
87.
88.
89.
90.
91.
92.
93.
94.
95.
96.
97.
98.
99.
100.
101.
102.
Taylor, T. E. & Strickland, G. T. Malaria. In: Hunter GW, Strickland GT, Magill AJ,
Kersey R. , editor. Hunter's Tropical Medicine and Emerging Infectious Diseases.
Philadelphia: WB Saunders, pp. 614– 43 (2000).
White, N. J. Malaria. In: Cook GC, Zumla AI, Weir J. , editor. Manson's Tropical
Diseases. Philadelphia, PA: WB Saunders, pp. 1205– 95 (2003).
Murphy, G. S. & Oldfield, E. C., 3rd. Falciparum malaria. Infect Dis Clin North Am
10, 747-75 (1996).
Genton, B. & D'Acremont, V. Clinical features of malaria in returning travelers and
migrants. In: Schlagenhauf P. , editor. Travelers' malaria. Hamilton: BC Decker, pp.
371– 92 (2001).
Svenson, J. E., MacLean, J. D., Gyorkos, T. W. & Keystone, J. Imported malaria.
Clinical presentation and examination of symptomatic travelers. Arch Intern Med
155, 861-8 (1995).
Organization, W. H. Severe and complicated malaria. World Health Organization,
Communicable Diseases Cluster. Trans R Soc Trop Med Hyg 84 Suppl 2, S1- 65
(1990).
WHO. Severe falciparum malaria. World Health Organization, Communicable
Diseases Cluster. Trans R Soc Trop Med Hyg 94 Suppl 1, S1-90 (2000).
Struik, S. S. & Riley, E. M. Does malaria suffer from lack of memory? Immunol
Rev 201, 268-90 (2004).
Moody, A. H. & Chiodini, P. L. Methods for the detection of blood parasites. Clin
Lab Haematol 22, 189-201 (2000).
Torres, J. R. Malaria and babesiosis. In:Badour LM, Grobach SL, editor. Therapy of
Infectionus Diseases Philadelphia, PA: Saunders; pp. 597-613 (2003).
Lee, S. H. et al. New strategies for the diagnosis and screening of malaria. Int J
Hematol 76 Suppl 1, 291-3 (2002).
Hanscheid, T. & Grobusch, M. P. How useful is PCR in the diagnosis of malaria?
Trends Parasitol 18, 395-8 (2002).
Gachot, B., Wolff, M., Nissack, G., Veber, B. & Vachon, F. Acute lung injury
complicating imported Plasmodium falciparum malaria. Chest 108, 746-9 (1995).
WHO. Severe and complicated malaria. World Health Organization, Communicable
Diseases Cluster. Trans R Soc Trop Med Hyg 84 Suppl 2, S1- 65 (1990).
WHO. Tropical Disease Research Progress (1975- 1994). Twelfth Programme
Report of the UNDP/World Bank/WHO Tropical Diseases TDR. (1995).
Reilly, A. F. et al. Genetic diversity in human Fc receptor II for immunoglobulin G:
Fc gamma receptor IIA ligand-binding polymorphism. Clin Diagn Lab Immunol 1,
640-644 (1994).
Barnwell, J. W., Nichols, M. E. & Rubinstein, P. In vitro evaluation of the role of
the Duffy blood group in erythrocyte invasion by Plasmodium vivax. J Exp Med
169, 1795-802 (1989).
Barnwell, J. W. & Wertheimer, S. P. Plasmodium vivax: merozoite antigens, the
Duffy blood group, and erythrocyte invasion. Prog Clin Biol Res 313, 1-11 (1989).
Miller, L. H. & Carter, R. A review. Innate resistance in malaria. Exp Parasitol 40,
132-46 (1976).
Miller, L. H. & Dvorak, J. A. Visualization of red cell membranes of lysed malariainfected cells by differential interference microscopy. J Parasitol 59, 202-3 (1973).
Miller, L. H., Dvorak, J. A., Shiroishi, T. & Durocher, J. R. Influence of erythrocyte
membrane components on malaria merozoite invasion. J Exp Med 138, 1597-601
(1973).
__________________________________________________________________________
Amre Nasr, 2008
49
103.
104.
105.
106.
107.
108.
109.
110.
111.
112.
113.
114.
115.
116.
117.
118.
119.
120.
Miller, L. H., Mason, S. J., Clyde, D. F. & McGinniss, M. H. The resistance factor
to Plasmodium vivax in blacks. The Duffy-blood-group genotype, FyFy. N Engl J
Med 295, 302-4 (1976).
Miller, L. H., McGinniss, M. H., Holland, P. V. & Sigmon, P. The Duffy blood
group phenotype in American blacks infected with Plasmodium vivax in Vietnam.
Am J Trop Med Hyg 27, 1069-72 (1978).
Miller, L. H., Aikawa, M. & Dvorak, J. A. Malaria (Plasmodium knowlesi)
merozoites: immunity and the surface coat. J Immunol 114, 1237-42 (1975).
Miller, L. H., Mason, S. J., Dvorak, J. A., McGinniss, M. H. & Rothman, I. K.
Erythrocyte receptors for (Plasmodium knowlesi) malaria: Duffy blood group
determinants. Science 189, 561-3 (1975).
Weatherall, D. J. Common genetic disorders of the red cell and the 'malaria
hypothesis'. Ann Trop Med Parasitol 81, 539-48 (1987).
Bayoumi, R. A. Does the mechanism of protection from falciparum malaria by red
cell genetic disorders involve a switch to a balanced TH1/TH2 cytokine production
mode? Med Hypotheses 48, 11-7 (1997).
Aidoo, M. et al. Protective effects of the sickle cell gene against malaria morbidity
and mortality. Lancet 359, 1311-2 (2002).
Williams, T. N. et al. Sickle cell trait and the risk of Plasmodium falciparum malaria
and other childhood diseases. J Infect Dis 192, 178-86 (2005).
Ayi, K., Turrini, F., Piga, A. & Arese, P. Enhanced phagocytosis of ring-parasitized
mutant erythrocytes: a common mechanism that may explain protection against
falciparum malaria in sickle trait and beta-thalassemia trait. Blood 104, 3364-71
(2004).
Williams, T. N. et al. An immune basis for malaria protection by the sickle cell trait.
PLoS Med 2, e128 (2005).
Cabrera, G. et al. The sickle cell trait is associated with enhanced immunoglobulin G
antibody responses to Plasmodium falciparum variant surface antigens. J Infect Dis
191, 1631-8 (2005).
Marsh, K., Otoo, L., Hayes, R. J., Carson, D. C. & Greenwood, B. M. Antibodies to
blood stage antigens of Plasmodium falciparum in rural Gambians and their relation
to protection against infection. Trans R Soc Trop Med Hyg 83, 293-303 (1989).
Ntoumi, F. et al. Sickle cell trait carriage: imbalanced distribution of IgG subclass
antibodies reactive to Plasmodium falciparum family-specific MSP2 peptides in
serum samples from Gabonese children. Immunol Lett 84, 9-16 (2002).
Ntoumi, F. et al. Influence of carriage of hemoglobin AS and the Fc gamma receptor
IIa-R131 allele on levels of immunoglobulin G2 antibodies to Plasmodium
falciparum merozoite antigens in Gabonese children. J Infect Dis 192, 1975-80
(2005).
Ntoumi, F. et al. Plasmodium falciparum: sickle-cell trait is associated with higher
prevalence of multiple infections in Gabonese children with asymptomatic
infections. Exp Parasitol 87, 39-46 (1997).
Ntoumi, F. et al. Imbalanced distribution of Plasmodium falciparum MSP-1
genotypes related to sickle-cell trait. Mol Med 3, 581-92 (1997).
Gilles, H. M. et al. Glucose-6-phosphate-dehydrogenase deficiency, sickling, and
malaria in African children in South Western Nigeria. Lancet 1, 138-40 (1967).
Guinet, F. et al. A comparison of the incidence of severe malaria in Malian children
with normal and C-trait hemoglobin profiles. Acta Trop 68, 175-82 (1997).
__________________________________________________________________________
50
121.
122.
123.
124.
125.
126.
127.
128.
129.
130.
131.
132.
133.
134.
135.
136.
137.
138.
139.
140.
141.
Ringelhann, B., Hathorn, M. K., Jilly, P., Grant, F. & Parniczky, G. A new look at
the protection of hemoglobin AS and AC genotypes against Plasmodium falciparum
infection: a census tract approach. Am J Hum Genet 28, 270-9 (1976).
Thompson, G. R. Significance of haemoglobins S and C in Ghana. Br Med J 1, 6825 (1962).
Thompson, G. R. Malaria and Stress in Relation to Haemoglobins S and C. Br Med
J 2, 976-8 (1963).
Agarwal, A. et al. Hemoglobin C associated with protection from severe malaria in
the Dogon of Mali, a West African population with a low prevalence of hemoglobin
S. Blood 96, 2358-63 (2000).
Mockenhaupt, F. P. et al. Hemoglobin C and resistance to severe malaria in
Ghanaian children. J Infect Dis 190, 1006-9 (2004).
Modiano, D. et al. Haemoglobin C protects against clinical Plasmodium falciparum
malaria. Nature 414, 305-8 (2001).
Fairhurst, R. M., Fujioka, H., Hayton, K., Collins, K. F. & Wellems, T. E. Aberrant
development of Plasmodium falciparum in hemoglobin CC red cells: implications
for the malaria protective effect of the homozygous state. Blood 101, 3309-15
(2003).
Friedman, M. J., Roth, E. F., Nagel, R. L. & Trager, W. The role of hemoglobins C,
S, and Nbalt in the inhibition of malaria parasite development in vitro. Am J Trop
Med Hyg 28, 777-80 (1979).
Olson, J. A. & Nagel, R. L. Synchronized cultures of P. falciparum in abnormal red
cells: the mechanism of the inhibition of growth in HbCC cells. Blood 67, 997-1001
(1986).
Pasvol, G. & Wilson, R. J. The interaction of malaria parasites with red blood cells.
Br Med Bull 38, 133-40 (1982).
Rihet, P., Flori, L., Tall, F., Traore, A. S. & Fumoux, F. Hemoglobin C is associated
with reduced Plasmodium falciparum parasitemia and low risk of mild malaria
attack. Hum Mol Genet 13, 1-6 (2004).
Fairhurst, R. M. et al. Abnormal display of PfEMP-1 on erythrocytes carrying
haemoglobin C may protect against malaria. Nature 435, 1117-21 (2005).
Duffy, P. E. & Fried, M. Red blood cells that do and red blood cells that don't: how
to resist a persistent parasite. Trends Parasitol 22, 99-101 (2006).
Ohashi, J. et al. Extended linkage disequilibrium surrounding the hemoglobin E
variant due to malarial selection. Am J Hum Genet 74, 1198-208 (2004).
Hutagalung, R. et al. Influence of hemoglobin E trait on the severity of Falciparum
malaria. J Infect Dis 179, 283-6 (1999).
Chotivanich, K. et al. Hemoglobin E: a balanced polymorphism protective against
high parasitemias and thus severe P. falciparum malaria. Blood 100, 1172-6 (2002).
Flint, J., Harding, R. M., Boyce, A. J. & Clegg, J. B. The population genetics of the
haemoglobinopathies. Baillieres Clin Haematol 11, 1-51 (1998).
Allen, S. J. et al. alpha+-Thalassemia protects children against disease caused by
other infections as well as malaria. Proc Natl Acad Sci U S A 94, 14736-41 (1997).
Mockenhaupt, F. P. et al. Alpha(+)-thalassemia protects African children from
severe malaria. Blood 104, 2003-6 (2004).
Wambua, S. et al. The effect of alpha+-thalassaemia on the incidence of malaria and
other diseases in children living on the coast of Kenya. PLoS Med 3, e158 (2006).
Williams, T. N. et al. Both heterozygous and homozygous alpha+ thalassemias
protect against severe and fatal Plasmodium falciparum malaria on the coast of
Kenya. Blood 106, 368-71 (2005).
__________________________________________________________________________
Amre Nasr, 2008
51
142.
143.
144.
145.
146.
147.
148.
149.
150.
151.
152.
153.
154.
155.
156.
157.
158.
159.
160.
Mockenhaupt, F. P. et al. The contribution of alpha+-thalassaemia to anaemia in a
Nigerian population exposed to intense malaria transmission. Trop Med Int Health
4, 302-7 (1999).
Oppenheimer, S. J. et al. The interaction of alpha thalassaemia with malaria. Trans R
Soc Trop Med Hyg 81, 322-6 (1987).
Williams, T. N. et al. High incidence of malaria in alpha-thalassaemic children.
Nature 383, 522-5 (1996).
Williams, T. N. et al. Negative epistasis between the malaria-protective effects of
alpha+-thalassemia and the sickle cell trait. Nat Genet 37, 1253-7 (2005).
Rowe, J. A., Moulds, J. M., Newbold, C. I. & Miller, L. H. P. falciparum rosetting
mediated by a parasite-variant erythrocyte membrane protein and complementreceptor 1. Nature 388, 292-5 (1997).
Cockburn, I. A. et al. A human complement receptor 1 polymorphism that reduces
Plasmodium falciparum rosetting confers protection against severe malaria. Proc
Natl Acad Sci U S A 101, 272-7 (2004).
Bellamy, R., Kwiatkowski, D. & Hill, A. V. Absence of an association between
intercellular adhesion molecule 1, complement receptor 1 and interleukin 1 receptor
antagonist gene polymorphisms and severe malaria in a West African population.
Trans R Soc Trop Med Hyg 92, 312-6 (1998).
Zimmerman, P. A. et al. CR1 Knops blood group alleles are not associated with
severe malaria in the Gambia. Genes Immun 4, 368-73 (2003).
Nagayasu, E. et al. CR1 density polymorphism on erythrocytes of falciparum
malaria patients in Thailand. Am J Trop Med Hyg 64, 1-5 (2001).
Collins, W. E. & Jeffery, G. M. A retrospective examination of sporozoite- and
trophozoite-induced infections with Plasmodium falciparum in patients previously
infected with heterologous species of Plasmodium: effect on development of
parasitologic and clinical immunity. Am J Trop Med Hyg 61, 36-43 (1999).
Jeffery, G. M. Epidemiological significance of repeated infections with homologous
and heterologous strains and species of Plasmodium. Bull World Health Organ 35,
873-82 (1966).
Anders, R. F. Antigenic diversity in Plasmodium falciparum. Acta Leiden 60, 57-67
(1991).
Smythe, J. A. et al. Structural diversity in the Plasmodium falciparum merozoite
surface antigen 2. Proc Natl Acad Sci U S A 88, 1751-5 (1991).
Nussenzweig, V. & Nussenzweig, R. S. Rationale for the development of an
engineered sporozoite malaria vaccine. Adv Immunol 45, 283-334 (1989).
Nussenzweig, V. & Nussenzweig, R. S. Circumsporozoite proteins of malaria
parasites. Bull Mem Acad R Med Belg 144, 493-504 (1989).
Saul, A. Kinetic constraints on the development of a malaria vaccine. Parasite
Immunol 9, 1-9 (1987).
Good, M. F. et al. Human T-cell recognition of the circumsporozoite protein of
Plasmodium falciparum: immunodominant T-cell domains map to the polymorphic
regions of the molecule. Proc Natl Acad Sci U S A 85, 1199-203 (1988).
White, J. H. & Kilbey, B. J. DNA replication in the malaria parasite. Parasitol Today
12, 151-5 (1996).
Zhan, Z., Wang, X. & Yu, J. [Erythrocyte immunity and regulating factors in mice
infected with Plasmodium berghei ANKA strain]. Zhongguo Ji Sheng Chong Xue
Yu Ji Sheng Chong Bing Za Zhi 13, 260-3 (1995).
__________________________________________________________________________
52
161.
162.
163.
164.
165.
166.
167.
168.
169.
170.
171.
172.
173.
174.
175.
176.
177.
Sedegah, M., Hedstrom, R., Hobart, P. & Hoffman, S. L. Protection against malaria
by immunization with plasmid DNA encoding circumsporozoite protein. Proc Natl
Acad Sci U S A 91, 9866-70 (1994).
Renia, L. et al. Effector functions of circumsporozoite peptide-primed CD4+ T cell
clones against Plasmodium yoelii liver stages. J Immunol 150, 1471-8 (1993).
Charoenvit, Y. et al. Plasmodium yoelii: 17-kDa hepatic and erythrocytic stage
protein is the target of an inhibitory monoclonal antibody. Exp Parasitol 80, 419-29
(1995).
Wang, R. et al. Protection against malaria by Plasmodium yoelii sporozoite surface
protein 2 linear peptide induction of CD4+ T cell- and IFN-gamma-dependent
elimination of infected hepatocytes. J Immunol 157, 4061-7 (1996).
Freeman, R. R. & Parish, C. R. Plasmodium yoelii: antibody and the maintenance of
immunity in BALB/c mice. Exp Parasitol 52, 18-24 (1981).
Grun, J. L. & Weidanz, W. P. Antibody-independent immunity to reinfection
malaria in B-cell-deficient mice. Infect Immun 41, 1197-204 (1983).
Amante, F. H., Crewther, P. E., Anders, R. F. & Good, M. F. A cryptic T cell
epitope on the apical membrane antigen 1 of Plasmodium chabaudi adami can prime
for an anamnestic antibody response: implications for malaria vaccine design. J
Immunol 159, 5535-44 (1997).
Amante, F. H. & Good, M. F. Prolonged Th1-like response generated by a
Plasmodium yoelii-specific T cell clone allows complete clearance of infection in
reconstituted mice. Parasite Immunol 19, 111-26 (1997).
Linke, A. et al. Plasmodium chabaudi chabaudi: differential susceptibility of genetargeted mice deficient in IL-10 to an erythrocytic-stage infection. Exp Parasitol 84,
253-63 (1996).
von der Weid, T., Honarvar, N. & Langhorne, J. Gene-targeted mice lacking B cells
are unable to eliminate a blood stage malaria infection. J Immunol 156, 2510-6
(1996).
Helmby, H., Kullberg, M. & Troye-Blomberg, M. Expansion of IL-3-responsive IL4-producing non-B non-T cells correlates with anemia and IL-3 production in mice
infected with blood-stage Plasmodium chabaudi malaria. Eur J Immunol 28, 255970 (1998).
Langhorne, J. The immune response to the blood stages of Plasmodium in animal
models. Immunol Lett 41, 99-102 (1994).
Bouharoun-Tayoun, H., Attanath, P., Sabchareon, A., Chongsuphajaisiddhi, T. &
Druilhe, P. Antibodies that protect humans against Plasmodium falciparum blood
stages do not on their own inhibit parasite growth and invasion in vitro, but act in
cooperation with monocytes. J Exp Med 172, 1633-41 (1990).
Wahlin, B. et al. Human antibodies to a Mr 155,000 Plasmodium falciparum antigen
efficiently inhibit merozoite invasion. Proc Natl Acad Sci U S A 81, 7912-6 (1984).
Green, T. J., Morhardt, M., Brackett, R. G. & Jacobs, R. L. Serum inhibition of
merozoite dispersal from Plasmodium falciparum schizonts: indicator of immune
status. Infect Immun 31, 1203-8 (1981).
David, P. H., Hommel, M., Miller, L. H., Udeinya, I. J. & Oligino, L. D. Parasite
sequestration in Plasmodium falciparum malaria: spleen and antibody modulation of
cytoadherence of infected erythrocytes. Proc Natl Acad Sci U S A 80, 5075-9
(1983).
Carlson, J., Holmquist, G., Taylor, D. W., Perlmann, P. & Wahlgren, M. Antibodies
to a histidine-rich protein (PfHRP1) disrupt spontaneously formed Plasmodium
falciparum erythrocyte rosettes. Proc Natl Acad Sci U S A 87, 2511-5 (1990).
__________________________________________________________________________
Amre Nasr, 2008
53
178.
179.
180.
181.
182.
183.
184.
185.
186.
187.
188.
189.
190.
191.
192.
193.
Bouharoun-Tayoun, H. & Druilhe, P. Plasmodium falciparum malaria: evidence for
an isotype imbalance which may be responsible for delayed acquisition of protective
immunity. Infect Immun 60, 1473-1481 (1992).
Bouharoun-Tayoun, H., Oeuvray, C., Lunel, F. & Druilhe, P. Mechanisms
underlying the monocyte-mediated antibody-dependent killing of Plasmodium
falciparum asexual blood stages. J Exp Med 182, 409-418 (1995).
Celada, A., Cruchaud, A. & Perrin, L. H. Assessment of immune phagocytosis of
Plasmodium falciparum infected red blood cells by human monocytes and
polymorphonuclear leukocytes. A method for visualizing infected red blood cells
ingested by phagocytes. J Immunol Methods 63, 263-71 (1983).
Ferreira, M. U., Kimura, E. A., De Souza, J. M. & Katzin, A. M. The isotype
composition and avidity of naturally acquired anti-Plasmodium falciparum
antibodies: differential patterns in clinically immune Africans and Amazonian
patients. Am J Trop Med Hyg 55, 315-23 (1996).
Aucan, C. et al. High immunoglobulin G2 (IgG2) and low IgG4 levels are associated
with human resistance to Plasmodium falciparum malaria. Infect Immun 68, 12521258 (2000).
Gysin, J., Dubois, P. & Pereira da Silva, L. Protective antibodies against
erythrocytic stages of Plasmodium falciparum in experimental infection of the
squirrel monkey, Saimiri sciureus. Parasite Immunol 4, 421-430 (1982).
Ndungu, F. M. et al. Naturally acquired immunoglobulin (Ig)G subclass antibodies
to crude asexual Plasmodium falciparum lysates: evidence for association with
protection for IgG1 and disease for IgG2. Parasite Immunol 24, 77-82 (2002).
Taylor, R. R., Allen, S. J., Greenwood, B. M. & Riley, E. M. IgG3 antibodies to
Plasmodium falciparum merozoite surface protein 2 (MSP2): increasing prevalence
with age and association with clinical immunity to malaria. Am J Trop Med Hyg 58,
406-13 (1998).
Hviid, L. Clinical disease, immunity and protection against Plasmodium falciparum
malaria in populations living in endemic areas. Expert Rev Mol Med 1998, 1-10
(1998).
Hviid, L. Naturally acquired immunity to Plasmodium falciparum malaria in Africa.
Acta Trop 95, 270-5 (2005).
Kyes, S., Horrocks, P. & Newbold, C. Antigenic variation at the infected red cell
surface in malaria. Annu Rev Microbiol 55, 673-707 (2001).
Rich, S. M. & Ayala, F. J. Population structure and recent evolution of Plasmodium
falciparum. Proc Natl Acad Sci U S A 97, 6994-7001 (2000).
Ahlborg, N., Ling, I. T., Howard, W., Holder, A. A. & Riley, E. M. Protective
immune responses to the 42-kilodalton (kDa) region of Plasmodium yoelii merozoite
surface protein 1 are induced by the C-terminal 19-kDa region but not by the
adjacent 33-kDa region. Infect Immun 70, 820-5 (2002).
Bull, P. C., Lowe, B. S., Kortok, M. & Marsh, K. Antibody recognition of
Plasmodium falciparum erythrocyte surface antigens in Kenya: evidence for rare
and prevalent variants. Infect Immun 67, 733-9 (1999).
Chattopadhyay, R. et al. Plasmodium falciparum infection elicits both variantspecific and cross-reactive antibodies against variant surface antigens. Infect Immun
71, 597-604 (2003).
Franks, S. et al. Genetic diversity and antigenic polymorphism in Plasmodium
falciparum: extensive serological cross-reactivity between allelic variants of
merozoite surface protein 2. Infect Immun 71, 3485-95 (2003).
__________________________________________________________________________
54
194.
195.
196.
197.
198.
199.
200.
201.
202.
203.
204.
205.
206.
207.
208.
209.
210.
Bull, P. C. et al. Plasmodium falciparum-infected erythrocytes: agglutination by
diverse Kenyan plasma is associated with severe disease and young host age. J
Infect Dis 182, 252-9 (2000).
Dodoo, D. et al. Levels of antibody to conserved parts of Plasmodium falciparum
merozoite surface protein 1 in Ghanaian children are not associated with protection
from clinical malaria. Infect Immun 67, 2131-7 (1999).
Berzins, K. & Anders , R. F. The malaria antigens. In: Wahlgren M, Perlmann P,
eds. Malaria Molecular and Clinical Aspects. Chur, Switzerland: Harwood
Academic Publishers. 181- 216 (1999).
Perkins, M. E. Approaches to study merozoite invasion of erythrocytes. Res
Immunol 142, 662-5 (1991).
Lyon, J. A., Thomas, A. W., Hall, T. & Chulay, J. D. Specificities of antibodies that
inhibit merozoite dispersal from malaria-infected erythrocytes. Mol Biochem
Parasitol 36, 77-85 (1989).
Sharma, P. et al. Characterization of protective epitopes in a highly conserved
Plasmodium falciparum antigenic protein containing repeats of acidic and basic
residues. Infect Immun 66, 2895-904 (1998).
Ahlborg, N. et al. Plasmodium falciparum: differential parasite growth inhibition
mediated by antibodies to the antigens Pf332 and Pf155/RESA. Exp Parasitol 82,
155-63 (1996).
Brown, J. & Smalley, M. E. Specific antibody-dependent cellular cytotoxicity in
human malaria. Clin Exp Immunol 41, 423-9 (1980).
Brown, J. & Smalley, M. E. Inhibition of the in vitro growth of Plasmodium
falciparum by human polymorphonuclear neutrophil leucocytes. Clin Exp Immunol
46, 106-9 (1981).
Jones, K. R., Cottrell, B. J., Targett, G. A. & Playfair, J. H. Killing of Plasmodium
falciparum by human monocyte-derived macrophages. Parasite Immunol 11, 585-92
(1989).
Kharazmi, A. & Jepsen, S. Enhanced inhibition of in vitro multiplication of
Plasmodium falciparum by stimulated human polymorphonuclear leucocytes. Clin
Exp Immunol 57, 287-92 (1984).
Nnalue, N. A. & Friedman, M. J. Evidence for a neutrophil-mediated protective
response in malaria. Parasite Immunol 10, 47-58 (1988).
Orago, A. S. & Facer, C. A. Cytotoxicity of human natural killer (NK) cell subsets
for Plasmodium falciparum erythrocytic schizonts: stimulation by cytokines and
inhibition by neomycin. Clin Exp Immunol 86, 22-9 (1991).
Marsh, K. et al. Indicators of life-threatening malaria in African children. N Engl J
Med 332, 1399-404 (1995).
Kinyanjui, S. M., Conway, D. J., Lanar, D. E. & Marsh, K. IgG antibody responses
to Plasmodium falciparum merozoite antigens in Kenyan children have a short halflife. Malar J 6, 82 (2007).
Salmon, J. E., Edberg, J. C., Brogle, N. L. & Kimberly, R. P. Allelic polymorphisms
of human Fc gamma receptor IIA and Fc gamma receptor IIIB. Independent
mechanisms for differences in human phagocyte function. J Clin Invest 89, 12741281 (1992).
Groux, H. & Gysin, J. Opsonization as an effector mechanism in human protection
against asexual blood stages of Plasmodium falciparum: functional role of IgG
subclasses. Res Immunol 141, 529-542 (1990).
__________________________________________________________________________
Amre Nasr, 2008
55
211.
212.
213.
214.
215.
216.
217.
218.
219.
220.
221.
222.
223.
224.
225.
226.
227.
228.
229.
230.
Rascu, A., Repp, R., Westerdaal, N. A., Kalden, J. R. & van de Winkel, J. G.
Clinical relevance of Fc gamma receptor polymorphisms. Ann N Y Acad Sci 815,
282-95 (1997).
Shi, Y. P. et al. Fcgamma receptor IIa (CD32) polymorphism is associated with
protection of infants against high-density Plasmodium falciparum infection. VII.
Asembo Bay Cohort Project. J Infect Dis 184, 107-111 (2001).
Omi, K. et al. Fcgamma receptor IIA and IIIB polymorphisms are associated with
susceptibility to cerebral malaria. Parasitol Int 51, 361 - 366 (2002).
Cooke, G. S. et al. Association of Fcgamma receptor IIa (CD32) polymorphism with
severe malaria in West Africa. Am J Trop Med Hyg 69, 565-568 (2003).
Bryceson, A. D., Fleming, A. F. & Edington, G. M. Splenomegaly in Northern
Nigeria. Acta Trop 33, 185-214 (1976).
Greenwood, B. M. et al. Ethnic differences in the prevalence of splenomegaly and
malaria in The Gambia. Ann Trop Med Parasitol 81, 345-54 (1987).
Hill, A. V. Genetic susceptibility to malaria and other infectious diseases: from the
MHC to the whole genome. Parasitology 112 Suppl, S75-84 (1996).
Piazza, A. et al. Genetic and population structure of four Sardinian villages. Ann
Hum Genet 49, 47-63 (1985).
Dolo, A. et al. Difference in susceptibility to malaria between two sympatric ethnic
groups in Mali. Am J Trop Med Hyg 72, 243-8 (2005).
Modiano, D. et al. Plasmodium falciparum malaria in sympatric ethnic groups of
Burkina Faso, west Africa. Parassitologia 37, 255-9 (1995).
Modiano, D. et al. Different response to Plasmodium falciparum malaria in west
African sympatric ethnic groups. Proc Natl Acad Sci U S A 93, 13206-11 (1996).
Modiano, D. et al. Humoral response to Plasmodium falciparum Pf155/ring-infected
erythrocyte surface antigen and Pf332 in three sympatric ethnic groups of Burkina
Faso. Am J Trop Med Hyg 58, 220-4 (1998).
Modiano, D. et al. Interethnic differences in the humoral response to non-repetitive
regions of the Plasmodium falciparum circumsporozoite protein. Am J Trop Med
Hyg 61, 663-7 (1999).
Modiano, D. et al. The lower susceptibility to Plasmodium falciparum malaria of
Fulani of Burkina Faso (west Africa) is associated with low frequencies of classic
malaria-resistance genes. Trans R Soc Trop Med Hyg 95, 149-52 (2001).
Pandey, J. P., Astemborski, J. & Thomas, D. L. Epistatic effects of immunoglobulin
GM and KM allotypes on outcome of infection with hepatitis C virus. J Virol 78,
4561-5 (2004).
Pandey, J. P. & French, M. A. GM phenotypes influence the concentrations of the
four subclasses of immunoglobulin G in normal human serum. Hum Immunol 51,
99-102 (1996).
Bolad, A. et al. Distinct interethnic differences in immunoglobulin G class/subclass
and immunoglobulin M antibody responses to malaria antigens but not in
immunoglobulin G responses to nonmalarial antigens in sympatric tribes living in
West Africa. Scand J Immunol 61, 380-6 (2005).
Steinberg, S. G. & Cook, C. E. The distribution of human immunoglobulin
allotypes. Oxford University Press (1981).
Morahan, G., Berek, C. & Miller, J. F. An idiotypic determinant formed by both
immunoglobulin constant and variable regions. Nature 301, 720-2 (1983).
Torres, M., May, R., Scharff, M. D. & Casadevall, A. Variable-region-identical
antibodies differing in isotype demonstrate differences in fine specificity and
idiotype. J Immunol 174, 2132-42 (2005).
__________________________________________________________________________
56
231.
232.
233.
234.
235.
236.
237.
238.
239.
240.
241.
242.
243.
244.
245.
246.
247.
248.
Atherton, A., Armour, K. L., Bell, S., Minson, A. C. & Clark, M. R. The herpes
simplex virus type 1 Fc receptor discriminates between IgG1 allotypes. Eur J
Immunol 30, 2540-7 (2000).
Bang, R. et al. Analysis of binding sites in human C-reactive protein for
Fc{gamma}RI, Fc{gamma}RIIA, and C1q by site-directed mutagenesis. J Biol
Chem 280, 25095-102 (2005).
Stein, M. P. et al. C-reactive protein binding to FcgammaRIIa on human monocytes
and neutrophils is allele-specific. J Clin Invest 105, 369-76 (2000).
Abrams, E. T. et al. Malaria during pregnancy and foetal haematological status in
Blantyre, Malawi. Malar J 4, 39 (2005).
Hurt, N., Smith, T., Teuscher, T. & Tanner, M. Do high levels of C-reactive protein
in Tanzanian children indicate malaria morbidity. Clin Diagn Lab Immunol 1, 43744 (1994).
Hurt, N. et al. Evaluation of C-reactive protein and haptoglobin as malaria episode
markers in an area of high transmission in Africa. Trans R Soc Trop Med Hyg 88,
182-6 (1994).
Imrie, H. et al. Low prevalence of an acute phase response in asymptomatic children
from a malaria-endemic area of Papua New Guinea. Am J Trop Med Hyg 76, 280-4
(2007).
Ansar, W., Bandyopadhyay, S. M., Chowdhury, S., Habib, S. H. & Mandal, C. Role
of C-reactive protein in complement-mediated hemolysis in Malaria. Glycoconj J
23, 233-40 (2006).
Pied, S. et al. C-reactive protein protects against preerythrocytic stages of malaria.
Infect Immun 57, 278-82 (1989).
Marnell, L., Mold, C. & Du Clos, T. W. C-reactive protein: ligands, receptors and
role in inflammation. Clin Immunol 117, 104-11 (2005).
Carlson, C. S. et al. Polymorphisms within the C-reactive protein (CRP) promoter
region are associated with plasma CRP levels. Am J Hum Genet 77, 64-77 (2005).
MacGregor, A. J., Gallimore, J. R., Spector, T. D. & Pepys, M. B. Genetic effects on
baseline values of C-reactive protein and serum amyloid a protein: a comparison of
monozygotic and dizygotic twins. Clin Chem 50, 130-4 (2004).
Pankow, J. S. et al. Familial and genetic determinants of systemic markers of
inflammation: the NHLBI family heart study. Atherosclerosis 154, 681-9 (2001).
Retterstol, L., Eikvar, L. & Berg, K. A twin study of C-Reactive Protein compared
to other risk factors for coronary heart disease. Atherosclerosis 169, 279-82 (2003).
Creasey, A. et al. Eleven years of malaria surveillance in a Sudanese village
highlights unexpected variation in individual disease susceptibility and outbreak
severity. Parasitology 129, 263-71 (2004).
Theander, T. G. Unstable malaria in Sudan: the influence of the dry season. Malaria
in areas of unstable and seasonal transmission. Lessons from Daraweesh. Trans R
Soc Trop Med Hyg 92, 589-92 (1998).
Ogoe, B. M. et al. Studies on the allotypic variants of IgG receptors Fc gamma RIIA
and Fc gamma IIIb and their association with severe clinical malaria amonge
Ghanaian children. The Third MIM Pan-African Malaria Conference Abstract No:
166, 116- 116. (2002).
Ouma, C. et al. Association of FCgamma receptor IIA (CD32) polymorphism with
malarial anemia and high-density parasitemia in infants and young children. Am J
Trop Med Hyg 74, 573-7 (2006).
__________________________________________________________________________
Amre Nasr, 2008
57
249.
250.
251.
252.
253.
254.
255.
256.
257.
258.
259.
260.
261.
Modiano, D. et al. HLA class I in three West African ethnic groups: genetic
distances from sub-Saharan and Caucasoid populations. Tissue Antigens 57, 128-37
(2001).
Sanders, L. A. et al. Fc gamma receptor IIa (CD32) heterogeneity in patients with
recurrent bacterial respiratory tract infections. J Infect Dis 170, 854-861 (1994).
Tebo, A. E., Kremsner, P. G. & Luty, A. J. Fc gamma receptor-mediated
phagocytosis of Plasmodium falciparum-infected erythrocytes in vitro. Clin Exp
Immunol 130, 300-306 (2002).
Jafarshad, A. et al. A novel antibody-dependent cellular cytotoxicity mechanism
involved in defense against malaria requires costimulation of FcgammaRII and
FcgammaRIII. J Immunol 178, 3099 - 3106 (2007).
Ferreira, M. U., Kimura, E. A. S. & Katzin, A. M. The IgG-subclass distribution of
naturally acquired antibodies to Plasmodium falciparum, in relation to malaria
exposure and severity. Ann Trop Med Par 92, 245 - 56 (1998).
Hamad, A. A. et al. Chronic Plasmodium falciparum infections in an area of low
intensity malaria transmission in the Sudan. Parasitology 120 ( Pt 5), 447-56 (2000).
Hamad, A. A. et al. A marked seasonality of malaria transmission in two rural sites
in eastern Sudan. Acta Trop 83, 71-82 (2002).
Taylor, R. R., Smith, D. B., Robinson, V. J., McBride, J. S. & Riley, E. M. Human
antibody response to Plasmodium falciparum merozoite surface protein 2 is
serogroup specific and predominantly of the immunoglobulin G3 subclass. Infect
Immun 63, 4382-8 (1995).
Bredius, R. G. et al. Phagocytosis of Staphylococcus aureus and Haemophilus
influenzae type B opsonized with polyclonal human IgG1 and IgG2 antibodies.
Functional hFc gamma RIIa polymorphism to IgG2. J Immunol 151, 1463-1472
(1993).
Parren, P. W. et al. On the interaction of IgG subclasses with the low affinity Fc
gamma RIIa (CD32) on human monocytes, neutrophils, and platelets. Analysis of a
functional polymorphism to human IgG2. J Clin Invest 90, 1537-1546 (1992).
Sanders, L. A. et al. Human immunoglobulin G (IgG) Fc receptor IIA (CD32)
polymorphism and IgG2-mediated bacterial phagocytosis by neutrophils. Infect
Immun 63, 73-81 (1995).
Norris, C. F. et al. Relationship between Fc receptor IIA polymorphism and
infection in children with sickle cell disease. J Pediatr 128, 813-9 (1996).
Kovacs, A. et al. A novel common single nucleotide polymorphism in the promoter
region of the C-reactive protein gene associated with the plasma concentration of Creactive protein. Atherosclerosis 178, 193-8 (2005).
__________________________________________________________________________
58