* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Document
Duffy antigen system wikipedia , lookup
Immune system wikipedia , lookup
DNA vaccination wikipedia , lookup
Lymphopoiesis wikipedia , lookup
Psychoneuroimmunology wikipedia , lookup
Innate immune system wikipedia , lookup
Adaptive immune system wikipedia , lookup
Monoclonal antibody wikipedia , lookup
Cancer immunotherapy wikipedia , lookup
Molecular mimicry wikipedia , lookup
Adoptive cell transfer wikipedia , lookup
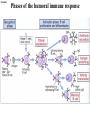
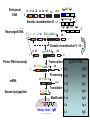
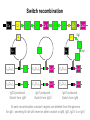
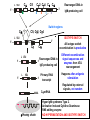
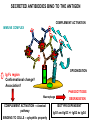
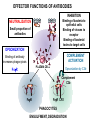
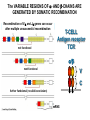
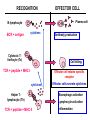
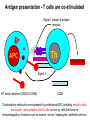
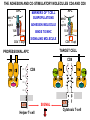
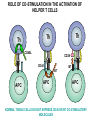
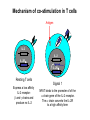
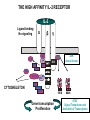
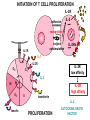
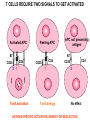
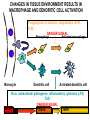
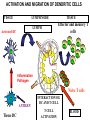
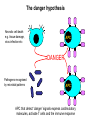
(review) Phases of the humoral immune response ISOTYPE SWITCH Antibody isotype switching Throughout the immune response the specificity of an antibody will be essentially the same (notwithstanding affinity maturation) The effector function of antibodies throughout a response needs to change drastically as the response progresses. Antibodies are able to retain Variable regions whilst exchanging Constant regions that contain the structures that interact with cells. Organisation of the functional human heavy chain C region genes J regions Cm Cd Cg3 Cg1 Ca1 Cg2 Cg4 Ce Ca2 embrionális Embryonal L1 V1 L2 V2 Ln Vn D1 - 12 DNS DNA 5' szomatikus rekombináció Somatic recombination D– D-J kapcsolódás átrendeződött Rearranged DNA DNS L1 V1 Ln Vn Cm Cδ Cg3 CM CD CG3 J1-4 J CA1Cε2 Ca1 CG1 CE2Cg1 CE1 Ca2 CG 2 CG 4 Cε1 CA2 C g1 Cg4 D1D2 J1J2-4 Cm CM Cδ CD 5' 3' szomatikus rekombináció Somatic recombination V -D-J kapcsolódás CM Cδ CD D2J1 J2-4 Cm 3' L1 V1 5' Primerprimer RNA transcript RNS-átirat mRNA mRNS 3’ Transcription transzkripció 5' L1 V1D2 J1 J2-4 Cm CM Cδ CD IgM Cγ1 IgG Cγ2 IgG Cγ3 IgG Modification Cγ4 IgG AAAA transzláció Translation L V DJ Cm C polipeptid Ig ISOTYPES Cµ 3' Processing átalakítás Cm L1 V1 D2 J1 CM naszcens Nascent polypeptide módosítás V C V–D–J NEHÉZL ÁNC (M ) Heavy chain IIgM Cα IgA Cε IgE Switch regions Cm Sm Cd Cg3 Sg3 Cg1 Sg1 Ca1 Sa1 Cg2 Sg2 Cg4 Sg4 Ce Se Ca2 Sa2 • Upstream of C regions are repetitive regions of DNA called switch regions. (The exception is the Cd region that has no switch region). • The Sm consists of 150 repeats of [(GAGCT)n(GGGGGT)] where n is between 3 and 7. • Switching is mechanistically similar in may ways to V(D)J recombination. • Isotype switching does not take place in the bone marrow, however, and it will only occur after B cell activation by antigen and interactions with T cells. Switch recombination Cm Cd Cg3 Cg1 Ca1 Cg2 Cg4 Cd Ce Cd Ca2 Sg3 Cg3 Cg3 Cm Sg1 Cm Cg1 V23D5J4 Cg3 V23D5J4 Ca1 V23D5J4 Ca1 V23D5J4 Cg3 V23D5J4 Ca1 V23D5J4 Ca1 IgG3 produced. Switch from IgM IgA1 produced. Switch from IgG3 IgA1 produced. Switch from IgM At each recombination constant regions are deleted from the genome An IgE - secreting B cell will never be able to switch to IgM, IgD, IgG1-4 or IgA1 Cm L VDJ Cδ Rearranged DNA in Cg2 Cg4 Ce Ca 3' 5' S S S S IgM-producing cell S Switch regions Cm Cδ, Cg2, Cg4 ISOTYPE SWITCH L VDJ 3' 5' Ce Ca L VDJ 5' 3' Rearranged DNA in IgE-producing cell L VDJ Primary RNA 3' transcript 5' AAAA e-Heavy chain Ce mRNA All isotype switch recombination is productive Different recombination signal sequences and enzymes from VDJ rearrangement Happens after antigenic stimulation Regulated by external signals, not random Hyper IgM syndrome Type 2. Activation Induced Cytidine Deaminase RNS editing enzyme NO HYPERMUTATION AND ISOTYPE SWITCH SOMATIC HYPERMUTATION MONOCLONAL ANTIBODIES Monoclonal antibodies - product of one B-lymphocyte clone - homogeneous in antigenspecificity, affinity, and isotype - can be found in pathologic condition in humans (the product of a malignant cell clone) - advantages against polyclonal antibodies: antibodies of a given specificity and isotype can be produced in high quantity and assured quality. -therapeutic usage of monoclonals: anti-TNF-α therapy in rheumatology, tumor therapy Possible use of monoclonal antibodies - Identifying cell types Immunohistochemistry Characterization of lymphomas with CD (cluster of differentiation) markers - Isolation of cells Isolation of CD34+ stem cells for autologous/allogeneic transplantation (from peripheral blood!) - Blood group determination (with anti-A, anti-B, and anti-D monoclonals) - Identification of cell surface and intracellular antigens Cell activation state - Targeted chemotherapy CD20+ anti-B-cell monoclonals in non-Hodgkin lymphoma Prevention of organ rejection after transplantation Monoclonal antibodies as drugs? Mouse monoclonal antibodies may elicit an immune response upon administration in human subjects. (see immunogenicity-determining factors!) How can we solve this problem? VL J2 gene product V35 gene product CDR1 CDR2 CDR3 Complementary Determining Region = hypervariable region STRUCTURE OF THE VARIABLE REGION • Hypervariable (HVR) or complimentarity determining regions (CDR) Variability Index HVR3 150 100 HVR2 HVR1 50 FR2 FR1 0 25 FR3 75 50 Amino acid residue • Framework regions (FR) FR4 100 SOMATIC HYPERMUTATION Day 0. Ag Day7 7nap CDR1 CDR2 CDR3 CDR1 CDR2 IgM PRIMARY immune response Day 14 14 nap IgM/IgG Day 14. Ag Day 21 21 nap IgG SECONDARY Immune response Hypervariable regions Plasma cell clones CDR3 1 2 3 4 5 6 7 8 AFFINITY MATURATION 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 ANTIBODY MEDIATED EFFECTOR FUNCTIONS • Neutralization – binding of the antibody inhibits the binding of the pathogen to the cell surface, entry to the cell or multiplication • Opsonization – binding of the antibody triggers complement activation and binding to the cell surface by complement (CR1) and IgG (FcgR) receptors • Cytophylic property - antibody isotypes have distinct complement activating and FcR binding activity SECRETED ANTIBODIES BIND TO THE ANTIGEN COMPLEMENT ACTIVATION IMMUNE COMPLEX OPSONIZATION Ig Fc region Conformational change? Association? FcR CR PHAGOCYTOSIS Macrophage COMPLEMENT ACTIVATION – classical pathway BINDING TO CELLS – cytophilic property DEGRADATION ISOTYPE DEPENDENT IgG1 and IgG3 >> IgG2 és IgG4 ANTIBODY MEDIATED EFFECTOR FUNCTIONS SPECIFIC ANTIBODY Bacterial toxin Bacterium in the interstitium Bacterium in the plasma Toxin receptor Neutralization Opsonization Complement activation COMPLEMENT Internalization Phagocytosis Phagocytosis and lysis EFFECTOR FUNCTIONS OF ANTIBODIES INHIBITION Binding of bacteria to epithelial cells Binding of viruses to receptor Binding of bacterial toxins to target cells NEUTRALIZATION Small proportion of antibodies OPSONIZATION COMPLEMENT ACTIVATION Binding of antibody increases phagocytosis PLAZMA CELL FcgR Opsonization by C3b Complement C3b FcgR FcgR CR1 PHAGOCYTES ENGULFMENT, DEGRADATION T – CELLS PROMOTE B – CELL DIFFERENTIATION T-CELL ANTIGEN CYTOKINES B -CELL PLASMA CELL ISOTYPE SWITCH AND AFFINITY MATURATION OCCURS IN COLLABORATION WITH T – CELLS ONLY HOW T – CELLS RECOGNIZE ANTIGENS? T CELL ACTIVATION B- AND T-CELL RECEPTORS SHARE BASIC STRUCTURE mIg H mIg L a TCR T cell receptor TCR V TCRa C TCR = a + The variable region of the -chain is generated by gene rearrangements of the V – D – J gene segments analogous to the generation of IgH diversity The variable region of the a-chain is generated by the recombination of V and J analogous to IgL T-CELL Single binding C site No somatic mutation The VARIABLE REGIONS OF a- AND -CHAINS ARE GENERATED BY SOMATIC RECOMBINATION Recombination of Va and Ja genes can occur after multiple unsuccessful recombination not functional T-CELL Antigen receptor TCR a/ next funcional V C further funkcional (no allelic exclusion) mRNS CHARACTERISTICS OF T-CELL ANTIGEN RECOGNITION 1. The TCR is not able to interact directly with soluble or cell-bound antigen 2. T-cell activation can be induced by antigen in the presence of acessory cells, only 3. T-cells recognize virus-infected cells ACCESSORY CELL ANTIGEN BINDING NO INTERACTION V T-CELL ACTIVATION C Antigen receptor B-CELL a/ T-CELL T CELL RECEPTOR MEDIATED SIGNALING Multisubunit Immune Recognition Receptors APC MIRR MHC Antigen Antigen TCR BCR αβ s V s s sV s C s s sC ss P P D/E X7 D/E X2 YXXL/I X7 YXXL/I ITAM Immunoreceptor Tyrosine-based Activation Motif ACTIVATION CD3 CD3 εδ s s s s εγ s s s s ζζ s s (review) Phases of T cell response (review) BCR signaling (review) TCR signaling Intracellular (cytoplasmatic) Ca2+ concentration increase The intracytoplasmatic Ca2+ increase can be monitored by fluorescent indicator dye. The intensity or the specificity of fluorescence increase paralelly with the Ca2+ concentration. Kinetic measurements connected to signaling (1.) Homogenous population – single cell property measured one after the other - timing Detected in Detected in single cell homogenous population – measured cell by cell Indicator – detection of intracellular Ca2+ signal fluorescent Ca2+ indicators Fluo-3 or Indo-1 Kinetic measurements connected to signaling (2.) Fluo-3 AM – excitable by blue light Indo-1 AM – excitable by UV light These indicator dyes connected with apolar groups (e.g. acetoxy-methylester: AM) – go across the cell membrane. In the cell, estherases cleveage these groups - the fluorochrome became polar – catched in the cell for example – ic Ca2+ signal in a single cell antigen presentation by B cell to T cell (click) THE IMMUNOLOGICAL SYNAPSE APC T APC T CELL THE INTERACTION OF T CELLS AND ANTIGEN PRESENTING CELLS interaction recognition 1 2 3 4 5 6 stabilization 7 8 separation Negulescu P.A. et. al. Immunity 4: 421-430, 1996 THE IMMUNOLOGICAL SYNAPSE ANTIGEN PRESENTING CELL ICAM-1 B7 CD48 CD4 CD2 CD28 adaptor LFA-1 SIGNALING COMPLEX T CELL ICAM – Intercellular Adhesion Molecule ACTIVATED T CELL RECOGNITION EFFECTOR CELL Plasma cell B-lymphocyte BCR + antigen cytokines Cytotoxic Tlimfocyte (Tc) Antibody production Cell killing TCR + peptide + MHC-I Effector cell retains specific receptor cytokines Helper Tlymphocyte (Th) TCR + peptide + MHC-II Effector cells secrete cytokines Macrophage activation Lymphocyte activation Inflammation RECOGNITION OF EXOGENOUS AND ENDOGENOUS ANTIGENES BY T-LYMPHOCYTES Tc Th Exogenous Ag Endogenous Ag Peptides of endogenous proteins (virus, tumor) bind to class I MHC molecules Peptides of exogenous proteins (toxin, bacteria, allergen) bind to class II MHC molecules Antigen presentation - T cells are co-stimulated Signal 1 antigen & antigen receptor Th APC ACTIVATION Signal 2 B7 family members (CD80 & CD86) CD28 Costimulatory molecules are expressed by professional APC including dendritic cells, monocytes, macrophages, and B cells, but not by cells that have no immunoregulatory functions such as muscle, nerves, hepatocytes, epithelial cells etc. THE ADHESION AND CO-STIMULATORY MOLECULES CD4 AND CD8 APC MARKERS OF T CELL SUBPOPULATIONS MHCII + peptid ADHESION MOLECULE TCR CD4 APC MHCI + peptid TCR BINDS TO MHC p56lck p56lck SIGNALING MOLECULE + CD4 T-sejt CD8 CD8+ T-sejt TARGET CELL PROFESSIONAL APC CD8 a1 s s a2 V4 a2 s s s s α β Lck Helper T-cell SIGNAL Lck a b T-cell Cytotoxic a1 V3 2m s s a3 V2 CD4 s s a3 s s 2m V1 2 1 a2 a1 ROLE OF CO-STIMULATION IN THE ACTIVATION OF HELPER T CELLS Th Th Th CD40L CD28 CD40 B7 B7 APC APC APC NORMAL TISSUE CELLS DO NOT EXPRESS CD40 OR B7 CO-STIMULATORY MOLECULES Mechanism of co-stimulation in T cells Antigen 1 IL-2 IL-2 IL-2Ra IL-2Ra Resting T cells Express a low affinity IL-2 receptor and g chains and produce no IL-2 Signal 1 NFAT binds to the promoter of of the a chain gene of the IL-2 receptor. The a chain converts the IL-2R to a high affinity form Mechanism of co-stimulation in T cells Costimulation Antigen Signal 2 1 2 Activates AP-1 and NFk-B to increase IL2 gene transcription by 3 fold Stabilises and thus increases the half-life of IL-2 mRNA by 20-30 fold IL-2 IL-2Ra IL-2 production increased by 100 fold overall Immunosuppressive drugs illustrate the importance of IL-2 in immune responses Cyclosporin & FK506 inhibit IL-2 by disrupting TcR signalling Rapamycin inhibits IL-2R signalling THE HIGH AFFINITY IL-2 RECEPTOR IL-2 Ligand binding No signaling α β γ JAK Janus kinase CYTOSKELETON Gene transcription Proliferation STAT Signal Transducer and Activatior of Transcription INITIATION OF T CELL PROLIFERATION IL-2R adhesion IL-2 recognition IL-2Rα costimulation IL-1R IL-1 IL-2R G0 IL-2R low affinity G1 IL-2 M G2 IL-2R high affinity S transferrin IL-2 insulin PROLIFERATION AUTOCRINE GROTH FACTOR CO-STIMULATION IS ESSENTIAL FOR PRIMING OF NAIVE T LYMPHOCYTES The antigen-specific and the co-stimulatory signals have to be induced in concert to induce T lymphocyte activation The antigen-specific and co-stimulatory signals can be delivered simultaneously by professional antigen presenting cells, only The antigen-specific and the co-stimulatory singnals has to be delivered by the same professional antigen presenting cell T CELLS REQUIRE TWO SIGNALS TO GET ACTIVATED Activated APC Resting APC B7 CD28 APC not presenting antigen B7 CD4 2 1 T-cell activation CD4 CD28 1 T-cell anergy CD4 CD28 2 No effect ANTIGEN SPECIFIC ACTIVATION, ANERGY OR NEGLECTION Anergy Antigen Naïve T cell 1 Signal 1 only IL-2 IL-2Ra Epithelial cell Self peptide epitopes presented by a non-classical APC e.g. an epithelial cell The T cell is unable to produce IL-2 and therefore is unable to proliferate or be clonally selected. Unlike immunosupressive drugs that inhibit ALL specificities of T cell, Signal 1 in the absence of signal 2 causes T cell unresponsiveness to a specific antigen PROFESSIONAL ANTIGEN PRESENTING CELLS Express MHC class I and class II molecules Express co-stimulatory molecules (B7, CD40) Take up extracellular antigens B cells – soluble proteins, toxins (ADAPTIVE) Macrophages – extracellular pathogens (bacteria, yeast) INNATE – particles Dendritic cells – viruses, apoptotic cells CHARACTERISTICS OF PROFESSIONAL ANTIGEN PRESENTING CELLS Macrophage Ag uptake phagocytosis +++ Dendritic cell phagocytosis +++ virus infection ++++ B - lymphocyte Ag-specific mIg ++++ MHC expression induced +/+++ bacteria, cytokine constitutive ++++ constitutive +++ immature/mature +++/++++ activation ++++ Pesented Ag particulate Ag intra/extracellular pathogens protein virus protein, allergen apoptotic cell soluble protein toxin Co-stimulation induced +/++ constitutive ++++ éretlen/érett +++/++++ induced +/+++ Localization lymphoid tissue connective tissue body cavities evenly lymphoid tissue connective tissue epithelium immature – tissue mature – T cell area Lymph node lymphoid tissue peripheral blood follicles ORIGIN AND DIFFERENTIATION OF DENDRITIC CELLS BONE MARROW MYELOID CMP CD34+ CLA+ LYMPHOID ? CD34+ STEM CELL CLP CD34+ CLABLOOD CD11c+ CD1a+ CD11c+ CD1a- CD14+ CD4+ IFNα Monocyte/preDC1 Langerhans/LC Interstitialis Macrophage DC1 TISSUE preDC2 DC2/plasmocitoid CHANGES IN TISSUE ENVIRONMENT RESULTS IN MACROPHAGE AND DENDRITIC CELL ACTIVATION Phagocytosis of bacteria, degradation (LPS, TLR) DANGER SIGNAL Macrophage Monocyte Dendritic cell Activated macrophage Activated dendritic cell Virus, extracellular pathogenes, inflammatory cytokines (LPS, TLR) DANGER SIGNAL BLOOD TISSUE LYMPH FUNCTIONS OF DENDRITIC CELLS Peptides MH C Protein En zym MHC I es MHC II MHC II Naíve T cell HIV-1 DC-SIGN FcgRII D Naíve T cell Exo som es Measles CD91 HSP+peptide CD14 Toll aVb5 Toll aVb3 PS-R CD36 CR N B7 aVb3 aVb5 ICAM-1 CD1a Glycolipid FcgRII AKTIVÁCIÓ IDC TISSUE Antigen uptake C G SI MR CD46 MR ABCA1 I MHC II PS-R CD40 MDR CCR7 SLAM MDC LYMPHATIC TISSUES Antigen processing and presentation MHC expression Costimulatory molecules ACTIVATION AND MIGRATION OF DENDRITIC CELLS TISSUE LYMPH NODE LYMPH Activated DC TISSUE Effector and memory T cells Inflammation Pathogen Naive T cells ANTIGEN Tissue DC INTERACTION OG DC AND T CELL T CELL ACTIVATION BLOOD DENDRITIC CELL T LYMPHOCITE INTERACTION IN THE LYMPHOID ORGANS Activated dendritic cells in the lymphatic tissues act as antigen presenting cells Tight contact with specific T cells The danger hypothesis & co-stimulation Full expression of T cell function and self tolerance depends upon when and where co-stimulatory molecules are expressed. Cell containing only self antigens No danger Apoptotic cell death. A natural, often useful cell death. No danger APC APC Innocuous challenge to the immune system fails to activate APC and fails to activate the immune system Fuchs & Matzinger 1995 The danger hypothesis Necrotic cell death e.g. tissue damage, virus infection etc APC DANGER Pathogens recognised by microbial patterns APC APC that detect ‘danger’ signals express costimulatory molecules, activate T cells and the immune response Some implications of the danger hypothesis • There is no window for tolerance induction in neonates • Neonatal T cells are not intrinsically tolerisable but the neonatal environment predisposes to tolerance • Antigens induce tolerance or immunity depending upon the ability of the immune system to sense them as ‘dangererous’, and not by sensing whether they are self or ‘non-self’. • Apoptosis, the ‘non-dangerous’ death of self cells may prevent autoimmunity when old or surplus cells are disposed of. • Suggests that tolerance is the default pathway of the immune system on encountering antigens. • Explains why immunisations require adjuvants to stimulate cues of danger such as cytokines or costimulatory molecule expression. Doesn’t exclude self-nonself discrimination, but is very hard to enequivocally disprove experimentally