* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Solute
Stoichiometry wikipedia , lookup
Inductively coupled plasma mass spectrometry wikipedia , lookup
Gas chromatography–mass spectrometry wikipedia , lookup
Spinodal decomposition wikipedia , lookup
Rutherford backscattering spectrometry wikipedia , lookup
Chemical Corps wikipedia , lookup
X-ray fluorescence wikipedia , lookup
Strengthening mechanisms of materials wikipedia , lookup
Chemical potential wikipedia , lookup
Abundance of the chemical elements wikipedia , lookup
Safety data sheet wikipedia , lookup
Electron configuration wikipedia , lookup
Registration, Evaluation, Authorisation and Restriction of Chemicals wikipedia , lookup
Metallic bonding wikipedia , lookup
Periodic table wikipedia , lookup
Solvent models wikipedia , lookup
Gas chromatography wikipedia , lookup
Chemical bond wikipedia , lookup
Condensed matter physics wikipedia , lookup
Molecular dynamics wikipedia , lookup
Liquid–liquid extraction wikipedia , lookup
Chemical element wikipedia , lookup
Implicit solvation wikipedia , lookup
Crystallization wikipedia , lookup
Chemical thermodynamics wikipedia , lookup
Vapor–liquid equilibrium wikipedia , lookup
Atomic nucleus wikipedia , lookup
Extended periodic table wikipedia , lookup
History of chemistry wikipedia , lookup
History of molecular theory wikipedia , lookup
IUPAC nomenclature of inorganic chemistry 2005 wikipedia , lookup
Chemistry: A Volatile History wikipedia , lookup
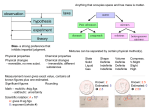
CP Chemistry Chapter 2 Matter and Change • Matter – anything that has mass and takes up space • Mass- amount of matter an object contains • Materials – differ in type of matter they are composed of Properties of matter – physical and chemical Physical property – quality or condition of a substance that can be observed or measured without changing the substance’s composition Examples of Physical Properties Color Odor Hardness Density Solubility Melting point, boiling point, freezing point Physical state – solid, liquid, gas States of matter • Solid (s) definite shape and volume • Liquid (l) definite volume, no definite shape • Gas (g) no definite shape, no definite volume • Plasma – we don’t deal with Phase Differences Solid – definite volume and shape; particles packed in fixed positions. Liquid – definite volume but indefinite shape; particles close together but not in fixed positions Gas – neither definite volume nor definite shape; particles are at great distances from one another Plasma – high temperature, ionized phase of matter as found on the sun. Gas and vapor not the same Gas – normally exists that way at room temperature Vapor – gas state of a substance that is normally a liquid or solid at room temperature Chemical Property • The ability of a substance to undergo chemical reactions and to form new substances • Rusting, burning, fermenting, exploding, rotting, decomposing • Iron and sulfur example Changes in matter • Physical change - A change which alters a material without changing its composition • Cutting, grinding, melting, boiling, freezing, dissolving Chemical Change • Change which results in one or more new substances being formed • The new substance has different chemical properties!!! • Examples • Paper burns, iron rusts, silver tarnishes • All physical and chemical changes involve a change in ENERGY • Energy is the ability to do work or produce heat Indications that a chemical reaction has occurred • Energy released or absorbed • Color change • Odor released • Production of a gas • Irreversibility • Formation of a solid (precipitate) Mixtures Physical blend of two or more substances Composition can vary 2 types: Heterogeneous Homogeneous Heterogeneous Mixture Not uniform in composition Has 2 or more phases Phase – any part of a system with uniform composition and properties Dirt, salad, paper, rocky road ice cream Homogeneous Mixture Has a completely uniform composition All components are evenly distributed Consists of a single phase Called a SOLUTION Solutions contain a SOLVENT and one or more SOLVENTS SOLVENT – substance that the solute is dissolved in. Is usually the substance present in the largest amount SOLUTE – the substance that dissolves in the solvent to make the solution Solution – what do you think of? • Can be gas, liquid, or solid!!!!! • Can you think of any examples? Solutions Gas solution AIR Solute – oxygen, carbon dioxide, other gases Solvent – nitrogen gas Liquid solution Rubbing alcohol Solute – isopropyl alcohol (liquid) Solvent – water (liquid) Another Liquid solution Saltwater Solute – salt (solid) Solvent – water (liquid) Solid solution – any alloy BRASS Solute – zinc (solid) Solvent – copper (solid) STAINLESS STEEL Solute – chromium, nickel (solids) Solvent – iron (solid) Separating mixtures Use PHYSICAL methods (no chemical changes involved) Magnet, sifting, Liquid mixtures – distillation Matter made up of Atoms Made up of protons, neutrons, and electrons Protons – have a positive charge Neutrons – no charge Electrons – negative charge In a neutral atom, #protons = #electrons Protons and neutrons – packed in a tiny NUCLEUS Electrons – move around the nucleus in an “electron cloud” Chemical bond is formed when two atoms share or transfer electrons Substances Can be elements or compounds Element – substances made up of only one kind of atom Every element has a unique atomic number Atomic number – number of protons in the nucleus Atom is the smallest particle of an element that has the properties of that element Are at least 111 officially named elements About 90 are naturally occurring Chemical symbol is used to represent each element Has one or two letters, first letter is always capitalized Compound – 2 or more elements that have been CHEMICALLY combined. Can only be separated by CHEMICAL means – chemical reaction must take place Periodic Table Gives names and symbols of all the elements In order of increasing ATOMIC NUMBER Two main divisions: Metals – left of “step” Nonmetals – right of “step” Most of the elements that touch the step are called METALLOIDS or SEMIMETALS (exception is Al (aluminum). It is a METAL Metals Shiny solids Exception is Hg (Mercury) is a LIQUID Good conductors of heat and electricity Malleable ductile Nonmetals Gases or dull solids (not shiny) Exception is Br (Bromine) is a liquid Poor conductors of heat and electricity Metalloids (Semimetals) Some properties of both metals and nonmetals Good as computer components (only conduct electricity when heated) 3 categories Monatomic (most elements) – exist as single atoms. Fe, Al, S Diatomic – exist in pairs when not bonded with other elements Are 7 – easy to remember! H, N, O, F, Cl, Br, I H2, N2, O2, F2, Br2, I2 Polyatomic A few elements exist as groups of 3 or more atoms O3 P4 Number written to the right and below the line Shows the number of atoms of that element in that element or compound H2, N2, O2, F2, Cl2, Br2, I2 P4 O3 Consist of two or more kinds of atoms that are chemically bonded H2O, NaCl, NaHCO3 (baking soda) Used to represent compounds Is a group of symbols that shows the number and kinds of elements in that compound Shows the DEFINITE makeup of the compound NaCl 1 Na 1 Cl H2O 2H 1O C12H22O11 (sucrose) (table sugar) 12 C atoms 22 H atoms 11 O atoms Smallest unit of sucrose has a total of 45 atoms Parenthesis means you have 2 groups of NH4 So……. Name and number of types of atoms…. N 2 H 8 C 1 O 3 Ca 5 P 3 O 12 F 1 To separate a compound to its elements, the compound must be CHEMICALLY changed Forming and separating compounds require CHEMICAL reactions to occur Matter substance element mixture compound heterogeneous homogeneous