* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Last Time… - UW-Madison Department of Physics
Density matrix wikipedia , lookup
Compact Muon Solenoid wikipedia , lookup
Renormalization group wikipedia , lookup
Nuclear structure wikipedia , lookup
Quantum field theory wikipedia , lookup
Elementary particle wikipedia , lookup
Quantum gravity wikipedia , lookup
Quantum tomography wikipedia , lookup
Aharonov–Bohm effect wikipedia , lookup
Matrix mechanics wikipedia , lookup
Canonical quantum gravity wikipedia , lookup
Bell's theorem wikipedia , lookup
Electron scattering wikipedia , lookup
Quantum mechanics wikipedia , lookup
Quantum entanglement wikipedia , lookup
Coherent states wikipedia , lookup
Renormalization wikipedia , lookup
History of quantum field theory wikipedia , lookup
Monte Carlo methods for electron transport wikipedia , lookup
Measurement in quantum mechanics wikipedia , lookup
Quantum chaos wikipedia , lookup
Identical particles wikipedia , lookup
Path integral formulation wikipedia , lookup
Quantum teleportation wikipedia , lookup
Quantum potential wikipedia , lookup
Eigenstate thermalization hypothesis wikipedia , lookup
Wave packet wikipedia , lookup
Symmetry in quantum mechanics wikipedia , lookup
Interpretations of quantum mechanics wikipedia , lookup
Double-slit experiment wikipedia , lookup
EPR paradox wikipedia , lookup
Quantum vacuum thruster wikipedia , lookup
Quantum electrodynamics wikipedia , lookup
Quantum state wikipedia , lookup
Relational approach to quantum physics wikipedia , lookup
Photon polarization wikipedia , lookup
Canonical quantization wikipedia , lookup
Quantum tunnelling wikipedia , lookup
Introduction to quantum mechanics wikipedia , lookup
Relativistic quantum mechanics wikipedia , lookup
Quantum logic wikipedia , lookup
Uncertainty principle wikipedia , lookup
Bohr–Einstein debates wikipedia , lookup
Old quantum theory wikipedia , lookup
Probability amplitude wikipedia , lookup
Theoretical and experimental justification for the Schrödinger equation wikipedia , lookup
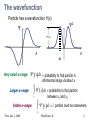
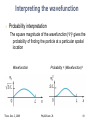
Exam 3 covers Lecture, Readings, Discussion, HW, Lab Exam 3 is tonight, Thurs. Dec. 3, 5:30-7 pm, 145 Birge Magnetic dipoles, dipole moments, and torque Magnetic flux, Faraday effect, Lenz’ law Inductors, inductor circuits Electromagnetic waves: Wavelength, freq, speed E&B fields, intensity, power, radiation pressure Polarization Modern Physics (quantum mechanics) Photons & photoelectric effect Bohr atom: Energy levels, absorbing & emitting photons Uncertainty principle Thurs. Dec. 3, 2009 Phy208 Lect. 26 1 From Last time… De Broglie wavelength Uncertainty principle Wavefunction of a particle Spatial extent of ‘wave packet’ x • x = spatial spread of ‘wave packet’ • Spatial extent decreases as the spread in included wavelengths increases. Thurs. Dec. 3, 2009 Phy208 Lect. 26 3 Wavefunction • Quantify this by giving a physical meaning to the wave that describing the particle. • This wave is called the wavefunction. – Cannot be experimentally measured! • But the square of the wavefunction is a physical quantity. – It’s value at some point in space is the probability of finding the particle there! Thurs. Dec. 3, 2009 Phy208 Lect. 26 4 The wavefunction Particle has a wavefunction (x) 2 2(x) x dx Very small x-range x 2 xdx = probability to find particle in infinitesimal range dx about x x2 Larger x-range Entire x-range xdx = probability to find particle 2 x1 between x1 and x2 2 x dx 1 particle must be somewhere Thurs. Dec. 3, 2009 Phy208 Lect. 26 5 Probability Probability 0.5 P(heads)=0.5 P(tails)=0.5 0.0 Heads Tails Probability P heads P tails 1 0.5 P(1)=1/6 P(2)=1/6 1/6 0.0 etc 1 2 3 4 5 6 P 1 P 2 P 3 P 4 P 5 P 6 1 Thurs. Dec. 3, 2009 Phy208 Lect. 26 6 Discrete vs continuous 6-sided die, unequal prob 0.5 1/6 0.0 Loaded die Infinite-sided die, all numbers between 1 and 6 “Continuous” probability distribution 1 2 3 4 5 6 0.5 P(x) Probability 6 P(x)dx 1 0.0 0 1 2 3 4 x 1 Thurs. Dec. 3, 2009 1/6 Phy208 Lect. 26 5 6 7 Example wavefunction What is P(-2<x<-1)? 1 2 xdx = fractional area under curve -2 < x < -1 2 2 = 1/8 total area -> P = 0.125 What is (0)? 2 xdx = entire area under 2 curve = 1 = (1/2)(base)(height)=22(0)=1 0 1/ 2 Thurs. Dec. 3, 2009 Phy208 Lect. 26 8 Wavefunctions Each quantum state has different wavefunction Wavefunction shape determined by physical characteristics of system. Different quantum mechanical systems Pendulum (harmonic oscillator) Hydrogen atom Particle in a box Each has differently shaped wavefunctions Thurs. Dec. 3, 2009 Phy208 Lect. 26 9 Quantum ‘Particle in a box’ Particle confined to a fixed region of space e.g. ball in a tube- ball moves only along length L L Classically, ball bounces back and forth in tube. A classical ‘state’ of the ball. State indexed by speed, momentum=(mass)x(speed), or kinetic energy. Classical: any momentum, energy is possible. Quantum: momenta, energy are quantized Thurs. Dec. 3, 2009 Phy208 Lect. 26 10 Classical vs Quantum Classical: particle bounces back and forth. Sometimes velocity is to left, sometimes to right L Quantum mechanics: Particle represented by wave: p = mv = h / Different motions: waves traveling left and right Quantum wavefunction: superposition of both at same time Thurs. Dec. 3, 2009 Phy208 Lect. 26 11 Quantum version Quantum state is both velocities at the same time Superposition waves is standing wave, made equally of Wave traveling right ( p = +h/ ) Wave traveling left ( p = - h/ ) Determined by standing wave condition L=n(/2) : 2L One halfwavelength L Quantum wave function: superposition of both motions. Thurs. Dec. 3, 2009 momentum h h p 2L Phy208 Lect. 26 2 2 x sin x L 12 Different quantum states p = mv = h / Different speeds correspond to different subject to standing wave condition integer number of half-wavelengths fit in the tube. 2L /n 2L n=1 One halfwavelength n=2 L Two halfwavelengths Thurs. Dec. 3, 2009 2 2 sin n x Wavefunction: x L momentum h h p po 2L n=1 n=2 Phy208 Lect. 26 momentum h h p 2 po L 13 Particle in box question A particle in a box has a mass m. Its energy is all kinetic = p2/2m. Just saw that momentum in state n is npo. It’s energy levels A. are equally spaced everywhere B. get farther apart at higher energy C. get closer together at higher energy. Thurs. Dec. 3, 2009 Phy208 Lect. 26 14 Particle in box energy levels Quantized momentum h h p n npo 2L Energy = kinetic 2 2 npo p E n2Eo 2m 2m Or Quantized Energy Energy n=5 n=4 En n2Eo n=3 n=quantum number n=2 n=1 Thurs. Dec. 3, 2009 Phy208 Lect. 26 15 Zero-point motion Lowest energy is not zero Confined quantum particle cannot be at rest Always some motion Consequency of uncertainty principle xp /2 p cannot be zero p not exactly known p cannot be exactly zero Thurs. Dec. 3, 2009 Phy208 Lect. 26 16 Question A particle is in a particular quantum state in a box of length L. The box is now squeezed to a shorter length, L/2. The particle remains in the same quantum state. The energy of the particle is now A. 2 times bigger B. 2 times smaller C. 4 times bigger D. 4 times smaller E. unchanged Thurs. Dec. 3, 2009 Phy208 Lect. 26 17 Quantum dot: particle in 3D box CdSe quantum dots dispersed in hexane (Bawendi group, MIT) Color from photon absorption Decreasing particle size Determined by energylevel spacing Energy level spacing increases as particle size decreases. i.e 2 E n 1 E n Thurs. Dec. 3, 2009 n 1 h 2 n 2 h 2 8mL2 8mL2 Phy208 Lect. 26 18 Interpreting the wavefunction Probability interpretation The square magnitude of the wavefunction ||2 gives the probability of finding the particle at a particular spatial location Wavefunction Thurs. Dec. 3, 2009 Probability = (Wavefunction)2 Phy208 Lect. 26 19 Higher energy wave functions L n p n=3 h 3 2L E Probability 2 h 32 8mL2 n=2 2 h 2L h2 2 8mL2 h 2L h2 8mL2 n=1 Wavefunction Thurs. Dec. 3, 2009 2 Phy208 Lect. 26 20 Probability of finding electron Classically, equally likely to find particle anywhere QM - true on average for high n Zeroes in the probability! Purely quantum, interference effect Thurs. Dec. 3, 2009 Phy208 Lect. 26 21 Quantum Corral D. Eigler (IBM) 48 Iron atoms assembled into a circular ring. The ripples inside the ring reflect the electron quantum states of a circular ring (interference effects). Thurs. Dec. 3, 2009 Phy208 Lect. 26 22 Particle in box again: 2 dimensions Motion in x direction Motion in y direction Same velocity (energy), but details of motion are different. Thurs. Dec. 3, 2009 Phy208 Lect. 26 23 Quantum Wave Functions Probability (2D) Wavefunction Ground state: same wavelength (longest) in both x and y Need two quantum #’s, one for x-motion one for y-motion Use a pair (nx, ny) Ground state: (1,1) Probability = (Wavefunction)2 One-dimensional (1D) case Thurs. Dec. 3, 2009 Phy208 Lect. 26 24 2D excited states (nx, ny) = (2,1) (nx, ny) = (1,2) These have exactly the same energy, but the probabilities look different. The different states correspond to ball bouncing in x or in y direction. Thurs. Dec. 3, 2009 Phy208 Lect. 26 25 Particle in a box What quantum state could this be? A. nx=2, ny=2 B. nx=3, ny=2 C. nx=1, ny=2 Thurs. Dec. 3, 2009 Phy208 Lect. 26 26 Next higher energy state Ball has same bouncing motion in x and in y. Is higher energy than state with motion only in x or only in y. (nx, ny) = (2,2) Thurs. Dec. 3, 2009 Phy208 Lect. 26 27 Three dimensions Object can have different velocity (hence wavelength) in x, y, or z directions. Need three quantum numbers to label state (nx, ny , nz) labels each quantum state (a triplet of integers) Each point in three-dimensional space has a probability associated with it. Not enough dimensions to plot probability But can plot a surface of constant probability. Thurs. Dec. 3, 2009 Phy208 Lect. 26 28 Particle in 3D box Ground state surface of constant probability (nx, ny, nz)=(1,1,1) 2D case Thurs. Dec. 3, 2009 Phy208 Lect. 26 29 (121) (112) (211) All these states have the same energy, but different probabilities Thurs. Dec. 3, 2009 Phy208 Lect. 26 30 (222) (221) Thurs. Dec. 3, 2009 Phy208 Lect. 26 31 3-D particle in box: summary Three quantum numbers (nx,ny,nz) label each state nx,y,z=1, 2, 3 … (integers starting at 1) Each state has different motion in x, y, z Quantum numbers determine px h n nx x h 2L Momentum in each direction: e.g. 2 2 p p p Energy: E y z E o nx2 ny2 nz2 2m 2m 2m 2 x Some quantum states have same energy Thurs. Dec. 3, 2009 Phy208 Lect. 26 32