* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Slide 1
Molecular neuroscience wikipedia , lookup
Central pattern generator wikipedia , lookup
Feature detection (nervous system) wikipedia , lookup
Neuroplasticity wikipedia , lookup
Endocannabinoid system wikipedia , lookup
Synaptic gating wikipedia , lookup
Neuroanatomy wikipedia , lookup
Neuropsychopharmacology wikipedia , lookup
Circumventricular organs wikipedia , lookup
Stimulus (physiology) wikipedia , lookup
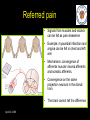
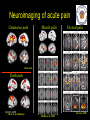
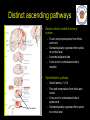
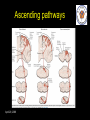
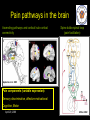
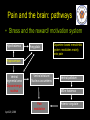
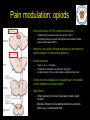
The perception of pain From Ch. 24 “Principles of Neural Science”, 4th Ed. Kandel et al April 29, 2009 Somatic sensations • Somatic sensation = bodily sensation – Pain is a submodality of somatic sensation – Pain and nociception (conscious vs. peripheral) • Pain sensation is the most salient sensation – – – – – Pricking Burning Aching Stinging Soreness • Pain is a warning of actual or potential injury and damage • Pain depends on the psychological state – The same stimulus can result in different responses under similar conditions and in different individuals (soldiers, athlete, etc.) April 29, 2009 Nociceptors Mechanical nociceptor • Information about stimuli that can damage tissue are conveyed by nociceptors • Chemicals are released from traumatized tissue – E.g. Substance P, histamine, and bradykinin • 3 classes of nociceptors – Mechnical: pinch, punctate, squeeze – Thermal: above 45 deg or below 5 deg Polymodal: mechanical, thermal, chemical April 29, 2009 Somatic receptor types 1 2 3 4 April 29, 2009 Afferent fibers Different size and conduction velocity of axons Compound AP = sum of all activated nerves Spike amplitude is proportional to fiber diameter -Large fibers conduct faster than small/ thin fibers because the internal resistance to current flow is low and nodes of Ranvier are spaced further apart - Myelination sheets increase conduction velocity April 29, 2009 Nociceptive afferents Compound Action Potential DRG Spinal dorsal horn First pain: Sharp and pricking, faster A-delta fibers Second pain, burning and dull, slower C-fibers April 29, 2009 Blocking each nerve blocks the sensation Spinal dorsal horn neurons • 2 overall types of interneurons: – – • Lamina I and II – – • Direct input from A-beta and A-delta. Direct/ indirect from C-fibers. Convergence of visceral afferents. WDR interneurons projecting to brain stem and thalamus. Lamina VI – • Direct input from A-beta. Nonnoxious input. Topographically organised receptive field Lamina V – • Direct input from mainly A-delta and C fibers. NS and WDR interneurons Lamina III and IV – • Nociceptive specific: responds exclusively to noxious stimuli Wide dynamic range neurons: graded response to non-noxious and noxious stimuli Direct input from A-alpha (nonnoxious) from joints and muscle Lamina VII and VIII – Respond to noxious input. Polysynaptic. Bilateral response April 29, 2009 Neurotransmitters • Fast synaptic potentials – Glutamate (amino acid) – Efficient reuptake of amino acids – Range: postsynaptic neurons in vicinity • Slow synaptic potentials – Neuropeptides e.g. Substance P – No reuptake mechanisms – Range: diffusion, many neurons, unlocalized nature of pain • Neuropeptides – Released and increased in persistent pain conditions – Enhances and prolong the actions of glutamate – Application of substance P produces signs of inflammation e.g. heat, redness, and swelling April 29, 2009 Peripheral activation and sensitization April 29, 2009 Chronic pain • Chronic pain appears to serve no useful purpose – Abnormal pain states – Nociceptive and neuropathic • Nociceptive pain – Direct activation of nociceptors – Tissue damage or inflammation • Neuropathic pain – Direct injury to the nerves – Peripheral or central – Burning or electrical sensation April 29, 2009 Chronic pain • Spontaneous ongoing pain – Pain of variable intensity and duration – Spontaneous discharges in periphery and centrally • Referred pain – Pain in a location distant from the source. Could be explained by viscero-somatic convergence in lamina V • Hyperalgesia – Increased pain sensitivity • Allodynia – Non-painful input becomes painful e.g. touch on sun burned skin • Allodynia and hyperalgesia only exist during stimulation • Alterations in biochemical properties and excitability of dorsal horn neurons can induce spontaneous pain, hyperalgesia and allodynia April 29, 2009 Referred pain • Signals from muscles and viscera can be felt as pain elsewhere • Example: myocardial infarction and angina can be felt in chest and left arm • Mechanism: convergence of afferents muscle/ viscera afferents and somatic afferents. • Convergence on the same projection neurons in the dorsal horn • The brain cannot tell the difference April 29, 2009 Hyperalgesia • Peripheral sensitization: – – • Central sensitization: – – – – – – • Increased spontaneous activity Hyperexcitability of spinal dorsal horn neurons Wind-up: progressive increased response = amplification (depends on glutamate acting on NMDA receptors) Prolonged after-discharges to afferent input Expansion of peripheral receptive fields of central neurons Can be induced by repetitive firing of nociceptive afferents Primary hyperalgesia – – • Increased nociceptor sensitivity Increased spontaneous activity Hyperalgesia in damaged area (within 5-10mm) Peripheral sensitization Secondary hyperalgesia: – – Hyperalgesia in surrounding undamaged tissue (10-20mm). Peripheral and central sensitization April 29, 2009 Clinical hyperalgesia Myofascial pain patients (PTS) vs. normal controls (CTR) Myofascial trigger points are hyperalgesic contractures in the muscle Pressure pain thresholds P<0.001 800 600 400 200 IMES stimulus-response curves 6 Pain Intensity Pressure [kPa] 1000 PTS 5 CTR 4 P<0.001 3 2 0 CTR PTS 6 10 14 Stimulus Intensity [mA] Niddam et al. 2008 April 29, 2009 Pain and the brain • Pain is a subjective conscious experience. Pain does not exist without the brain • CNS inhibitory or facilitatory mechanisms are remarkable efficient in decreasing or amplifying the pain experience • Changes in CNS contributes to chronic pain (reorganization: biochemical, atrophy, functions) • A better understanding of endogenous pain modulatory systems may lead to new mechanism-based therapies and drug targets April 29, 2009 Pain and the brain: modulation • Factors that can influence the pain experience – Top-down brain processes • • • • • Memories (previous experience) Emotion Cognition (attention/ distraction) Mood (depression, anxiety) Context (stress, anticipation/ expectation, placebo) – Endogenous pain control systems – Other factors • Genes • Pathological factors (structure, transmitters, receptors, transporters etc.) • Age, gender April 29, 2009 Acute vs. chronic pain • Acute pain characteristics – Activation of peripheral receptors under normal conditions – Sensation of pain closely related to the duration of the stimulus • Chronic pain characteristics – Spontaneous ongoing pain • Peripheral sensitization (spontaneous resting activity and hyperexcitable receptors) • Central sensitization (prolonged peripheral input) – Lowered pain threshold (Hyperalgesia) – Non-nociceptive input becomes painful (allodynia) – Functional and structural changes in PNS and CNS • • • • • April 29, 2009 Segmental expansion of receptive fields De novo synthesis of membrane proteins Spouting of spinal terminals of afferent fibers Formation of new synaptic contacts Altered balance in descending influences Acute vs. chronic pain • It is important to differentiate between: – Acute and chronic pain states • Different time horizons engage different emotional coping strategies • Chronic pain becomes maladaptive and is highly co-morbid with mood and anxiety disorders • Chronic pain induces CNS changes – Ongoing spontaneous chronic pain vs. perturbations of chronic pain (allodynia/ hyperalgesia) • Passive vs. active coping => medial vs. lateral brain regions? April 29, 2009 Neuroimaging of acute pain Cutaneous pain Muscle pain Visceral pain PFC PFC ACC PFC ACC PCC PPC PPC Insula PCC PCC Chen et al. Tooth pain Insula Thalamus Insula Vermis Amygdala April 29, 2009 Lin et al. (preliminary) Vermis Lu et al., 2004 Niddam et al., 2002 Distinct ascending pathways • Dorsal column-medial lemniscal system – Touch and proprioception from limbs and trunk – Somtatotopically organized from spinal to cortical level – Ascends ipsilateral side – Cross over to contralateral side in medulla • Contralateral April 29, 2009 Spinothalamic pathway – Spinal lamina I, V-VII – Pain and temperature from limbs and trunks – Cross over to contralateral side in spinal cord – Somtatotopically organized from spinal to cortical level Ascending pathways • 5 major ascending pathways – Spinothalamic: axons of nociceptive specific and WDR neurons from laminae I and V-VII; contralateral projection, ascends in anterolateral white matter – Spinoreticular: neurons in laminae VII and VIII; anterolateral ascend – Spinomesencephalic: neurons in laminae I and V; anterolateral ascend to PAG, and spinoparabrachial tract to PB, amygdala; pain affect – Cervicothalamic: arises from lateral cervical nucleus; laminae III and IV; some projects via the dorsal column to cuneate and gracile nuclei (large fiber pathway) – Spinohypothalamic; laminae I, V, VIII; autonomic control • Thalamic nuclei – Lateral nuclear group: spinothalamic tract, NS and WDR, laminae I and V, small receptive fields, encoding location of injury – Medial nuclear group: spinoreticulothalamic tract, laminae VII and VIII April 29, 2009 Ascending pathways April 29, 2009 Pain pathways in the brain Ascending pathways and cortical/ sub-cortical connectivity Spino-bulbo-spinal loop (pain facilitation) Apkarian et al. 2005 Pain components (variable expression): Sensory-discriminative, affective-motivational Cognitive, Motor April 29, 2009 Millan 2002 Pain and the brain: pathways • Stress and the reward/ motivation system Hypothalamus Amygdala Dopamine based mesolimbic system modulates mainly tonic pain Hippocampus Ventral tegmental area Dopoaminergic nucleus April 29, 2009 Ventral striatum/ Nucleus accumbens Ventral pallidum MDm thalamus Pain modulation Anterior cingulate Motivation and emotions April 29, 2009 Borsook 2007, EJP Pain modulation: large fibers The gate control hypothesis • The balance of activity in small- and largediameter fibers is important in pain transmission/ determines the pain intensity • The gate control theory involves 4 types of neurons in the dorsal horn of the spinal cord – – – – • Large-diameter fibers – • Inhibits interneuron and increases pain transmission/ Opens the pain-gate Observation: in absence of conduction in A-ά/ A-β fibers pain perception is abnormal – April 29, 2009 Excites interneuron and decrease pain transmission/ Closes the pain-gate Small-diameter fibers – • Large-diameter afferent (non-nociceptive) Small-diameter afferent (nociceptive) Inhibitory interneurons (spontaneously active) Projection neurons Pin prick, pinch, ice cold produces burning pain Pain modulation: opiods • Direct stimulation of PAG produces analgesia – – • Moprhine (an opioid) induced analgesia via endogenous opioid receptors in descending pathway • Opioid receptors – – – Types: -, δ-, κ-, nociceptin Transmitters: enkephalins, β-endorphin, dynorphin Location (mainly): PAG, ventral medulla, superficial dorsal horn • Stress-induced analgesia of escapable pain is mediated via the endogenous opioid system • Side effects – – April 29, 2009 Inhibits firing of nociceptive neurons in lamina I and V Descending pathway recruited: PAG excites rostroventral medulla/ nucleus raphe magnus (5HT) Other regions not involved in pain also contains opioid receptor Minimize diffusion by local administration can avoid side effects e.g. in cerebrospinal fluid